Clear Sky Science · en
Multicomponent synergistic immobilization via in-situ and dynamic exchange strategies for constructing hierarchical biopharmaceuticals
Why Building Smarter Medicines Matters
Many of today’s most powerful medicines are large, fragile biological molecules such as proteins and enzymes. They can fight cancer, kill harmful bacteria, and speed up healing, but they are easily damaged and hard to deliver to the right place in the body. This paper describes a new way to “pack” several of these delicate components together inside a protective, sponge-like material, so they can work in sequence with more punch and fewer side effects.
Turning a Crystal into a Molecular Apartment Building
The researchers start with a special porous material made from metal ions and small organic building blocks. It behaves a bit like a tiny, flexible apartment building full of rooms and hallways. They show that this framework, called a zeolitic pyrimidine framework, can be grown around proteins so that the proteins end up protected inside the crystal. This “in-situ” trapping does not crush or deactivate the proteins, and careful imaging confirms they are evenly distributed throughout the interior, much like tenants living on every floor of a building.
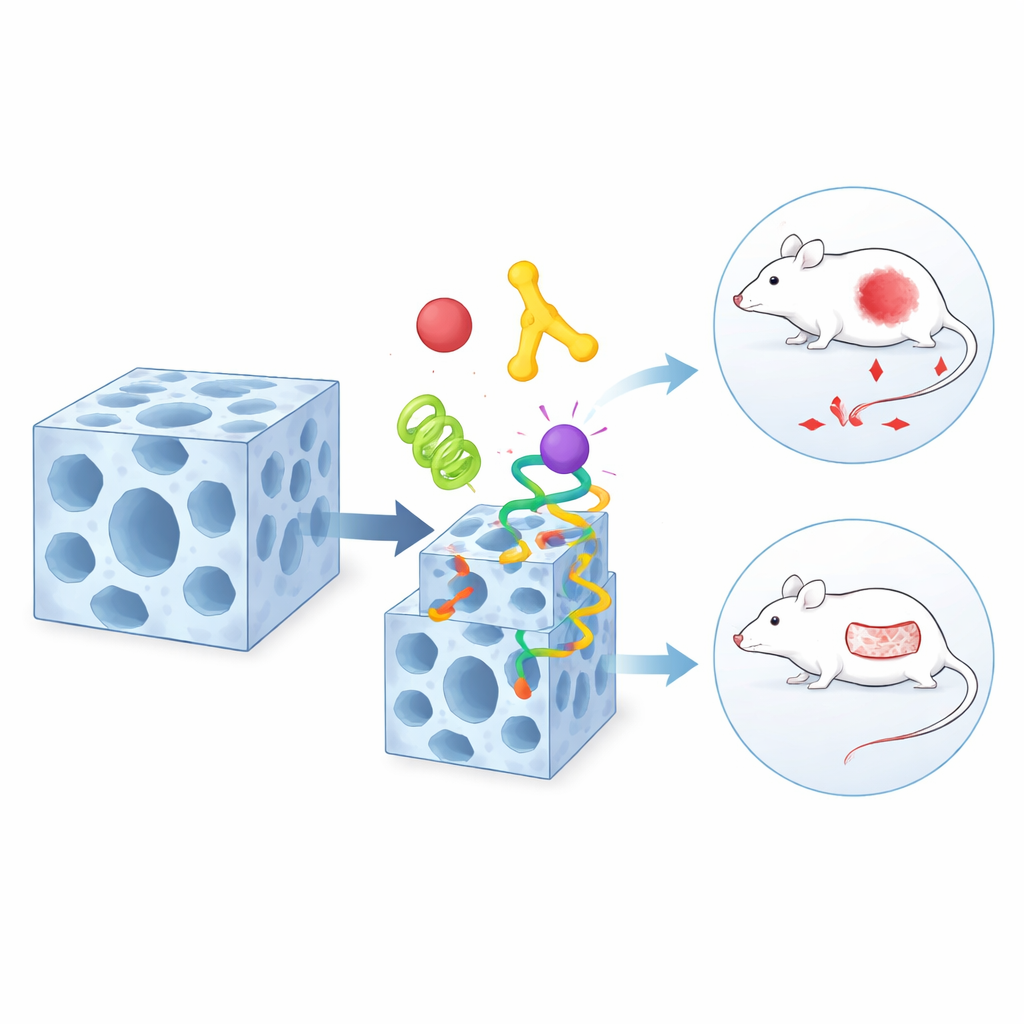
A Shape-Shifting Host for Surface Decoration
The same material has a surprising second trick: it can reversibly change its internal structure when exposed to certain solvents or water. During this gentle shape-shift, some of the chemical connections in the framework loosen and then re-form. The team discovered that particular chemical groups on protein surfaces—such as those found in certain amino acids—can take advantage of this moment of weakness. They temporarily compete with the framework’s original building blocks and slip into newly formed “defects” near the surface. As a result, a layer of proteins becomes anchored mainly on the outside of the crystal, while the inner ones remain in place. This dynamic exchange process lets the scientists decide which components live deep inside and which decorate the outer shell.
Designing Enzyme Assembly Lines
With both the inner and outer spaces under control, the authors build tiny enzymatic “assembly lines.” In one example, an enzyme that turns sugar into an aggressive by-product is locked inside the framework, while a second enzyme that safely breaks down that by-product is fixed near the surface. This inside–outside arrangement makes the reaction run faster and more efficiently than more traditional core–shell designs or random mixtures of enzymes. Because the outer enzymes are easier for molecules to reach, and the harmful intermediate is quickly removed, the system remains active over many cycles and better tolerates heat, harsh conditions, and repeated use.
Smart Anti-Cancer and Anti-Bacterial Packages
The real power of this approach appears when it is applied to medicine. The team combines a tumor-targeting protein, a glucose-eating enzyme, and a standard chemotherapy drug in a single, layered particle. The targeting component on the surface guides the particle to cancer cells, the enzyme drains their energy supply by consuming local sugar, and the small-molecule drug delivers an additional lethal blow. In mice with human tumor implants, this three-part package shrinks tumors far more effectively than any of the components alone or more simply arranged mixtures. In a separate design, the authors tailor the framework for infected skin wounds by placing an antibacterial peptide and a tissue-remodeling enzyme in different regions of the same carrier. In rat models, these particles sharply reduce infection and speed closure of difficult wounds compared with conventional treatments.

How This Work Moves Medicine Forward
To a non-specialist, the key message is that this study turns a single porous crystal into a programmable home for many kinds of therapeutic molecules. By deciding who lives inside, who resides on the surface, and in what ratio, the researchers can orchestrate stepwise treatments—first targeting, then attack, then repair—within one tiny object. This strategy not only protects fragile biological drugs but also lets them help one another, leading to stronger, more precise anti-cancer and anti-bacterial therapies. The same design principles could be adapted to many future “smart medicines” that need multiple components to work together in the right place at the right time.
Citation: Yang, M., Meng, X., Shi, R. et al. Multicomponent synergistic immobilization via in-situ and dynamic exchange strategies for constructing hierarchical biopharmaceuticals. Nat Commun 17, 2208 (2026). https://doi.org/10.1038/s41467-026-69009-8
Keywords: metal-organic frameworks, enzyme immobilization, cancer nanomedicine, wound healing, biopharmaceutical delivery