Clear Sky Science · en
Extracellular matrix mediates circulating tumor cell clustering in triple-negative breast cancer metastasis
How Rogue Breast Cancer Cells Team Up in the Bloodstream
When breast cancer spreads, it is often because loose cancer cells escape into the bloodstream and seed tumors in distant organs. This study asks a deceptively simple question with big implications: how do the most aggressive breast cancer cells, which have lost their usual “Velcro” for sticking together, still manage to travel as tight-knit clusters—and why does that make them so deadly?
A Sticky Sugar Coat That Replaces Classic Cell Glue
The authors focus on triple-negative breast cancer (TNBC), an especially aggressive form that often sheds circulating tumor cells (CTCs) into the blood. In many cancers, clusters of CTCs are far more likely to form metastases than lone cells. Typically, such clusters are held together by epithelial adherens junctions—special protein complexes that act like molecular rivets between neighboring cells. But TNBC cells frequently lose these junction proteins, raising a paradox: without the usual glue, what keeps their clusters intact? By mining large cancer gene databases, the researchers found that TNBC tumors are enriched for genes involved in the cell’s outer scaffolding, known as the extracellular matrix. One standout gene, HAS2, makes an enzyme that produces hyaluronan (HA), a long, sugary polymer that coats cells. High HAS2 and HA levels were strongly linked to aggressive, metastatic tumor features.
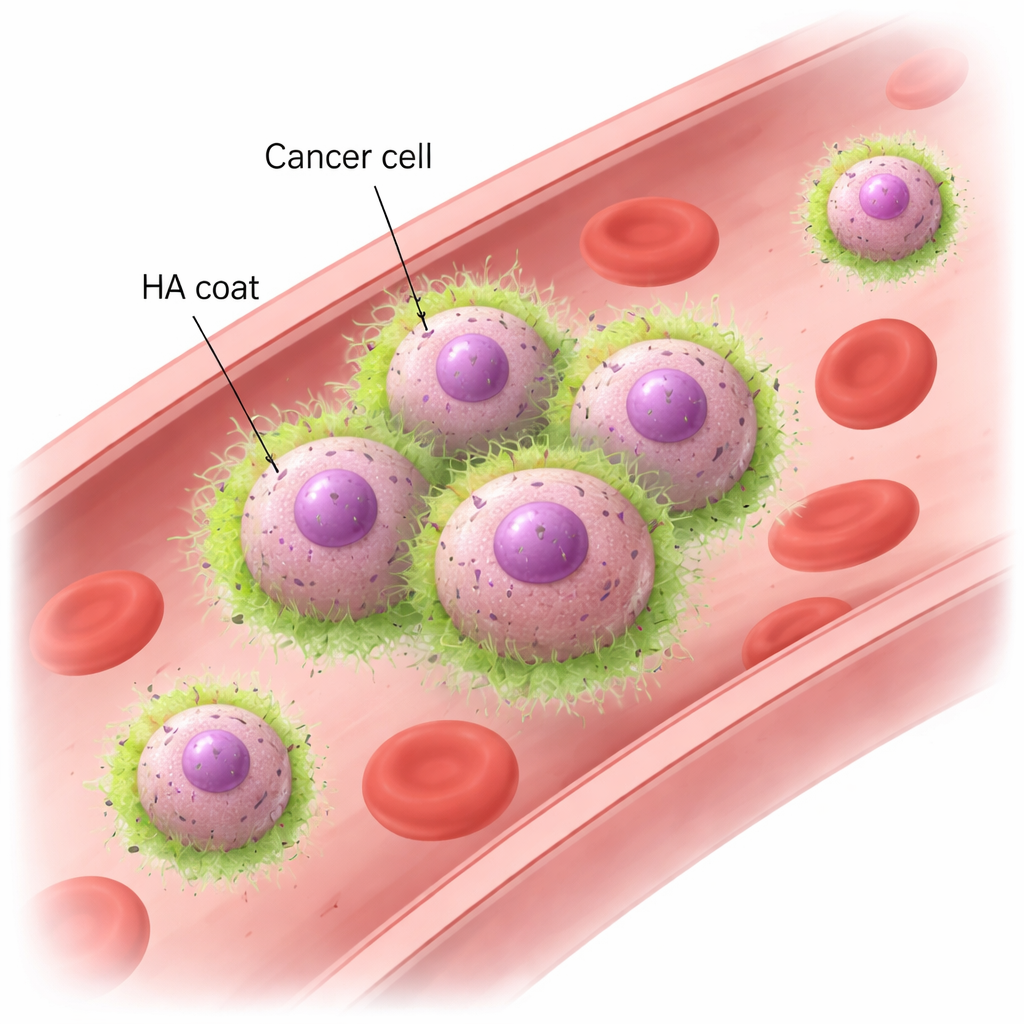
Hyaluronan and CD44: A New Way for Cells to Hold Hands
Using mouse models and human breast cancer cell lines, the team showed that metastatic TNBC cells rapidly form clusters when exposed to swirling fluid that mimics blood flow. These clusters remain intact even under forces similar to those in real blood vessels, whereas less aggressive breast cancer cells fall apart and drift as singles. Microscopy revealed that the metastatic cells are wrapped in an HA “coat,” which becomes concentrated exactly where cells touch each other. Removing HA with enzymes, or dialing down HAS2, caused the cells to lose their ability to cluster under flow, even though they could still sediment together when left undisturbed. The key partner on the cell side is CD44, a common surface receptor that binds HA. Cells lacking CD44 could no longer keep HA on their surface or form clusters. Conversely, non-clustering cells engineered to produce both HAS2 and CD44 gained a robust ability to cluster. These experiments showed that HA interacting with CD44 is both necessary and sufficient to make cancer cells stick together in fast-moving blood.
Tiny Protrusions and Mechanical Locks Build Strong Clusters
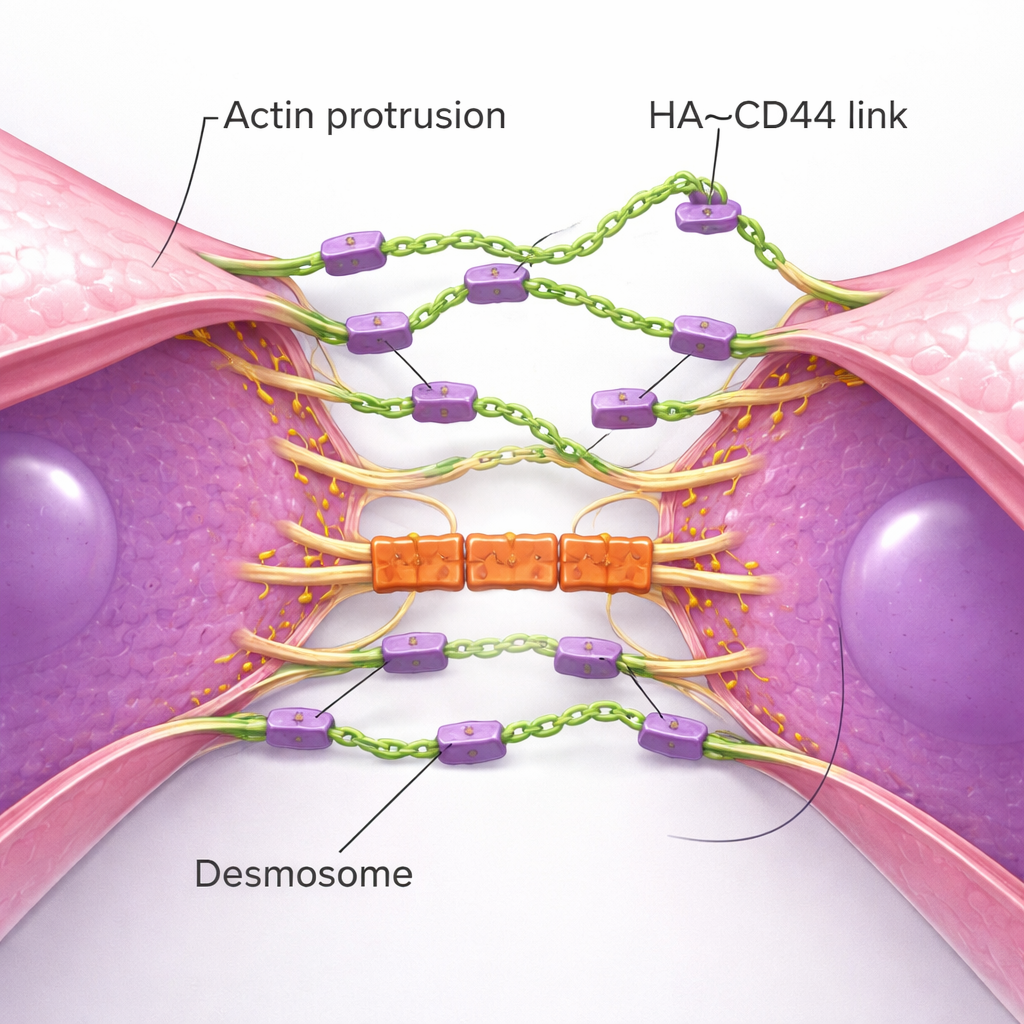
High-resolution imaging uncovered the physical choreography of how clusters form. Single TNBC cells extend numerous thin, actin-based membrane protrusions—like feelers—studded with CD44 and tipped with HA. When two cells drift close, these feelers from each cell meet first, forming initial HA–CD44 bridges. Over time, the protrusions slide alongside one another, pulling whole cell bodies closer until broad areas of the membranes lie nearly side by side. This process echoes how normal epithelial cells build adherens junctions, but here it is driven by a flexible sugar coat instead of rigid protein chains. As the contact zone matures, another set of structures joins in: desmosomes, robust junctions that link cell membranes to internal scaffolding. Desmosomal proteins accumulate only after HA-based contact is established and act as mechanical “locks” that help clusters resist the ripping forces of blood flow. When desmosome components were reduced, clusters formed but broke apart at higher shear stress.
Why Clustering Matters for Survival and Metastasis
The HA-based clustering system turned out to be more than just a way to stay together; it directly boosted survival. Under prolonged simulated blood flow, HA-rich clusters showed much lower levels of damaging reactive oxygen species and far less cell death than HA-depleted single cells. In mouse models, TNBC cells lacking HAS2 produced far fewer CTCs, formed only tiny clusters, and generated dramatically fewer lung metastases—even though their primary tumors grew just as well. When pre-formed clusters of control and HAS2-deficient cells were injected into mice, the HA-poor cells were much less likely to successfully colonize the lungs. Moreover, HA-coated CTCs could “recruit” other cells—both cancer cells that lacked HA and normal immune cells such as neutrophils—into mixed clusters, provided those partners expressed CD44. Patient blood samples from women with metastatic TNBC showed the same pattern: CTCs often carried an HA coat, with HA enriched at contact points between tumor cells and between tumor and non-tumor cells.
What This Means for Patients and Future Therapies
To a non-specialist, the main takeaway is that some of the deadliest breast cancer cells travel not as loners but as tiny, cooperative teams held together by a sugary outer coat and its receptor, rather than by the classic cell–cell “rivets” found in normal tissues. This HA–CD44-based mechanism helps tumor cells survive harsh blood flow, join forces with each other and with supportive blood cells, and more efficiently seed new tumors in distant organs. Targeting HA production (for example, by inhibiting HAS2) or blocking its interaction with CD44 could, in principle, weaken these clusters and make it harder for TNBC to metastasize, offering a new angle for future anti-metastatic treatments.
Citation: Bobkov, G.O., Patel, K.J., Lege, B.M. et al. Extracellular matrix mediates circulating tumor cell clustering in triple-negative breast cancer metastasis. Nat Commun 17, 1352 (2026). https://doi.org/10.1038/s41467-026-69007-w
Keywords: triple-negative breast cancer, circulating tumor cells, hyaluronan, CD44, metastasis