Clear Sky Science · en
Nonporous hydrophobic organic crystals for carbon dioxide capture via chain-melting phase transition
Why this matters for everyday life
Cutting carbon dioxide (CO₂) emissions is central to slowing climate change, but current capture technologies are often energy-hungry, costly, and complex. This study introduces a surprisingly simple solid material that can soak up CO₂ from smokestacks under realistic conditions and then let it go again with only a gentle heating step. By acting almost like a reversible "solid sponge" that works even in humid air, these crystals point toward more affordable, practical systems for cleaning up industrial exhaust.
A new kind of solid CO₂ sponge
The researchers focused on a family of small organic molecules derived from a common chemical called monoethanolamine, widely used today in liquid CO₂ scrubbers. By attaching a medium-length oily chain—ten carbon atoms long—they created a compound called C10-MEA that forms soft, needle-like crystals. Unlike conventional capture materials that depend on permanent pores and large internal surface areas, these crystals are initially nonporous and repel water. Yet when exposed to CO₂, they undergo a rapid solid-to-solid transformation that allows gas to move through and react, capturing CO₂ without the material ever turning into a liquid.
How CO₂ reshapes the solid
When C10-MEA crystals meet CO₂, heat released by the chemical reaction locally loosens and "melts" the long side chains, a phenomenon known as chain melting. This temporary softening lets CO₂ diffuse into the solid and form a tightly bound structure called an ammonium carbamate, in which each CO₂ molecule is paired with two amine groups from the host material. Advanced techniques—including X-ray powder diffraction, electron diffraction, infrared and Raman spectroscopy, and solid-state NMR—reveal that the crystals reorganize from a simple layered packing into a more intricate basket-weave network. In this new arrangement, a dense web of hydrogen bonds and cooperative interactions between the oily chains stabilizes the CO₂-rich solid, locking in a high capture capacity of about 2.5 millimoles of CO₂ per gram of material.
Efficient capture, gentle release
In performance tests, C10-MEA stood out among related compounds with slightly shorter or longer chains. It captured CO₂ quickly, reaching full loading within minutes even at low gas concentrations and modest temperatures. The process behaves like chemisorption—forming actual chemical bonds—yet the energy needed to reverse it is surprisingly small, comparable to that of materials that merely hold gases physically. Once the CO₂-rich crystal is formed, a modest temperature increase of only about 30 °C is enough to trigger desorption. Remarkably, the authors show that pure CO₂ itself can be used as the gas that strips out the captured CO₂ at around 65 °C and normal pressure, delivering an undiluted stream suitable for compression and storage.
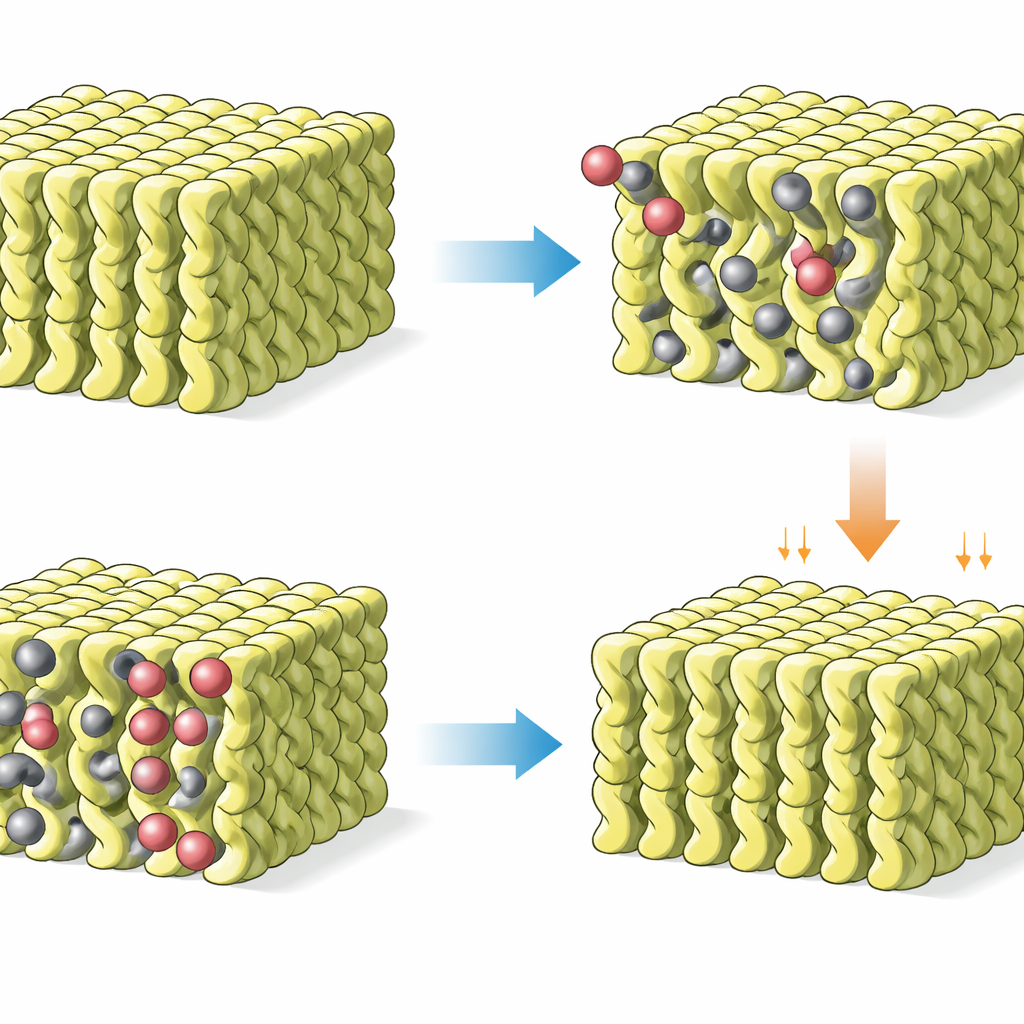
Robust under real-world conditions
For any capture medium to be practical at power plants or factories, it must tolerate water, oxygen, and repeated cycling. The hydrophobic nature of C10-MEA crystals makes them resistant to water uptake: under fully humid CO₂, they still form the same solid CO₂ adduct instead of turning into a water-swollen gel. Under humid nitrogen, by contrast, the starting crystals do absorb water and become gel-like, showing that the presence of CO₂ effectively protects the structure. Thermogravimetric and spectroscopic studies confirm that CO₂ is the primary species released on heating, and that the material remains stable under flue-gas-like mixtures containing air, moderate CO₂ levels, and high humidity. In continuous tests, the crystals completed hundreds of absorption–desorption cycles at constant temperature with only about a one-percent loss in capacity, underscoring their durability.
What this means for the future of carbon capture
By combining high CO₂ capacity, resistance to water, and low-energy release in a single, easily made organic solid, this work outlines a new blueprint for capture materials. Instead of relying on permanent pores or energy-intensive liquid solvents, the crystals exploit a reversible phase change—chain melting and recrystallization—to move between CO₂-free and CO₂-rich states. Because these materials can provide concentrated CO₂ streams using relatively mild heating, their estimated energy cost is lower than many existing options. If scaled successfully, such responsive solids could make industrial carbon capture more economical and flexible, helping to decarbonize large emission sources without demanding radical changes to how power and manufacturing plants operate.
Citation: Petrović, A., Lima, R.J.d.S., Hadaf, G.B. et al. Nonporous hydrophobic organic crystals for carbon dioxide capture via chain-melting phase transition. Nat Commun 17, 2293 (2026). https://doi.org/10.1038/s41467-026-69006-x
Keywords: carbon capture, solid sorbents, phase-change materials, chemisorption, hydrophobic crystals