Clear Sky Science · en
Escherichia coli promotes colorectal cancer metastasis by maintaining enhancer-promoter loops through releasing neutrophil extracellular traps
Gut Germs and Cancer’s Secret Highway
Colorectal cancer often kills not because of the original tumor in the bowel, but because cancer cells travel to the liver and settle there. This study uncovers an unexpected accomplice in that journey: a common gut bacterium, Escherichia coli. By following the chain of events from bacteria in the intestine to aggressive tumors in the liver, the researchers reveal how microbes, immune cells, and the DNA architecture inside cancer cells can work together to fuel metastasis—and suggest new ways to break this chain.
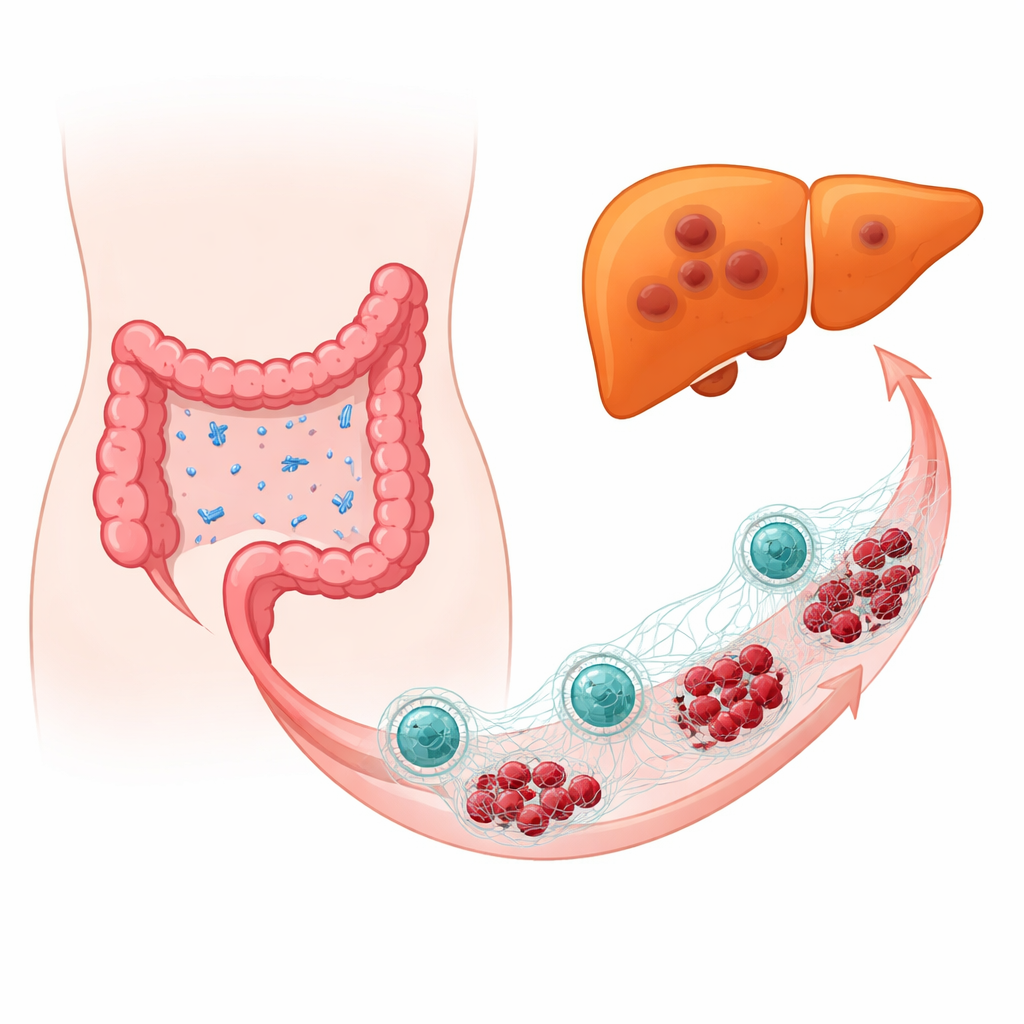
When Friendly Bacteria Turn Hostile
The team examined tumor samples from people with colorectal cancer, comparing those whose cancer had spread to the liver with those whose had not. Using high-resolution DNA-based profiling of microbes, they found that E. coli was much more abundant in cancers that had already seeded the liver. In mouse models and in mice with humanized immune systems, deliberately adding E. coli near colorectal tumors made liver metastases larger and more frequent, and pushed tumor cells toward a more mobile, invasive state. Even when most of the normal gut flora were removed with antibiotics and only E. coli was reintroduced, the bacteria still boosted tumor growth and spread, implicating E. coli itself rather than general microbiome disruption.
Immune Nets That Help Cancer Escape
Neutrophils—front-line immune cells that normally trap and kill microbes—emerged as crucial go-betweens. In response to E. coli, neutrophils released web-like structures called extracellular traps, made of DNA and toxic proteins. These nets are meant to capture bacteria, but in this setting they unintentionally protected and empowered cancer cells. When the researchers broke down the nets with enzymes or blocked key enzymes needed to form them, liver metastases shrank and tumors became less aggressive. Depleting neutrophils had a similar protective effect, pointing to a paradox: the very immune response that fights infection was being hijacked to help cancer spread.
From Electrical Signals to Gene Switches
How could sticky DNA nets outside a tumor cell change what happens deep inside its nucleus? The answer lay in calcium, a simple ion that acts as an electrical signal in cells. The study showed that neutrophil nets increased the level of a channel protein, TRPC1, on colorectal cancer cells. TRPC1 let extra calcium rush into the cell, which in turn activated a calcium-sensitive regulator called NFATC3 and created a self-reinforcing loop that kept TRPC1 levels high. This calcium surge encouraged two small proteins, S100A8 and S100A9, to latch together and then bind to a major growth-control protein, STAT3. The trio moved into the nucleus, where they did something especially striking: they helped bend and loop the DNA so that distant regulatory regions and gene start sites touched, physically stabilizing “enhancer–promoter loops” that turn certain genes up.
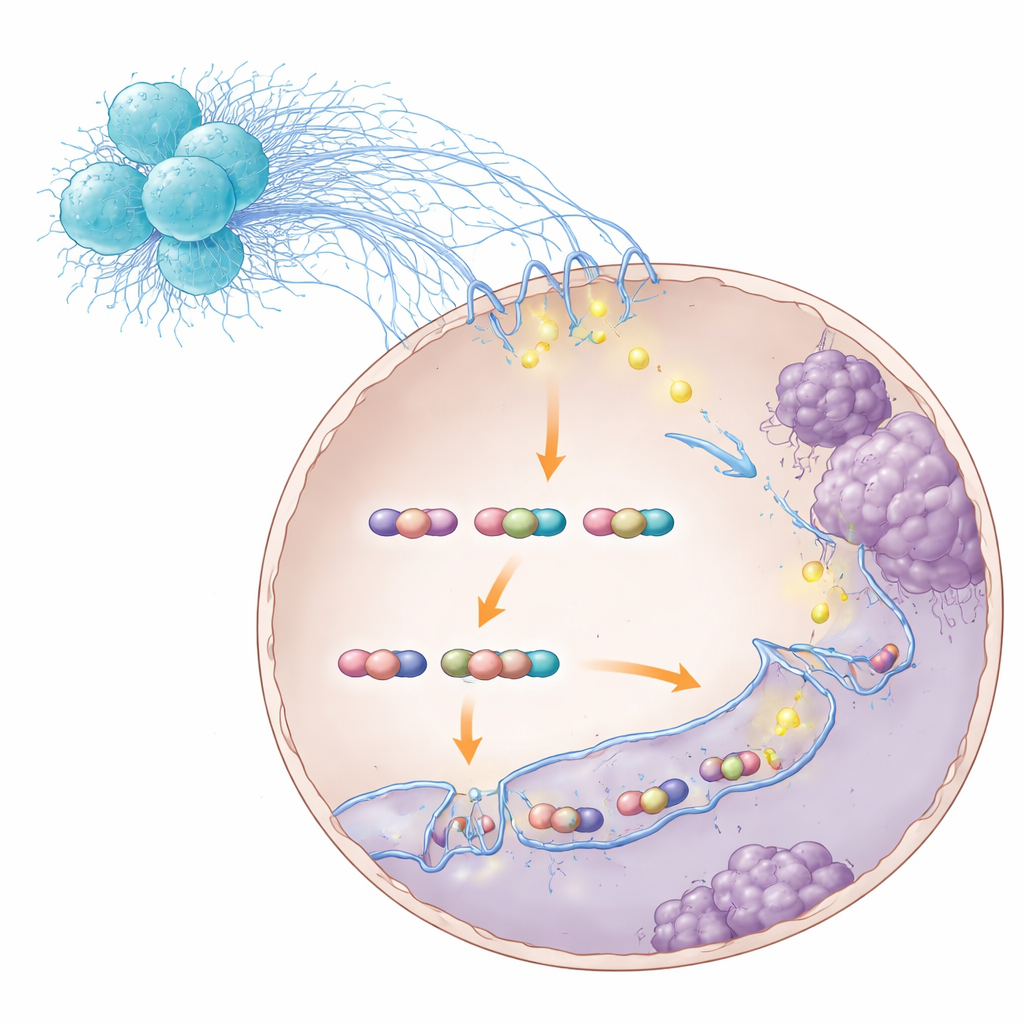
Rewiring Cancer Cells for Metastasis
By mapping the three-dimensional contacts along the genome, the researchers found that the STAT3–S100A8/9 complex promoted loops that favored genes involved in cell movement and invasion. One standout gene was TNS1, which drives cells to loosen their attachments and migrate more easily. When the STAT3–S100A8/9 complex was disrupted, many of these loops weakened or vanished, TNS1 levels dropped, and cancer cells lost some of their ability to invade and seed the liver. Importantly, this disruption did not stop STAT3 from touching DNA altogether—it specifically destabilized the looping architecture that supercharged metastatic programs. In animal models, blocking different steps in this chain—from the bacterial signaling molecule RIPK2 in neutrophils, to S100A8/9, to STAT3 itself—reduced liver metastasis and improved survival.
Breaking the Chain Between Bugs and Tumors
For non-specialists, the key message is that a common gut bacterium can help colorectal cancer spread by first provoking immune cells to cast DNA nets, and then using those nets to drive a calcium-powered relay that rewires how cancer genes are switched on. Rather than relying on broad antibiotics, which can damage beneficial microbes and blunt cancer immunotherapy, the work points to more precise drug targets: bacterial sensors like RIPK2, the TRPC1–calcium–S100A8/9 cascade, and the STAT3-driven DNA loops that lock metastasis-related genes into high gear. Interrupting this chain at one or more points could offer a new strategy to keep colorectal cancer from turning a local problem in the gut into a deadly disease in the liver.
Citation: Pan, B., Yao, Y., Zhang, Z. et al. Escherichia coli promotes colorectal cancer metastasis by maintaining enhancer-promoter loops through releasing neutrophil extracellular traps. Nat Commun 17, 2290 (2026). https://doi.org/10.1038/s41467-026-69005-y
Keywords: colorectal cancer metastasis, gut microbiome, neutrophil extracellular traps, STAT3 signaling, epigenetic gene regulation