Clear Sky Science · en
Chemical staining for fundamental studies and optimization of binders in Li-ion battery negative electrodes
Seeing the Hidden Glue Inside Batteries
Lithium-ion batteries power our phones, cars and soon entire neighborhoods, yet a crucial ingredient inside them is almost invisible: the binder, a thin polymer “glue” that holds particles together. This paper shows a new way to chemically “stain” that hidden glue so it lights up under an electron microscope. By finally seeing where the binder really sits, the authors show how to make longer-lasting, faster-charging batteries and improve factory processes that currently rely on trial and error.
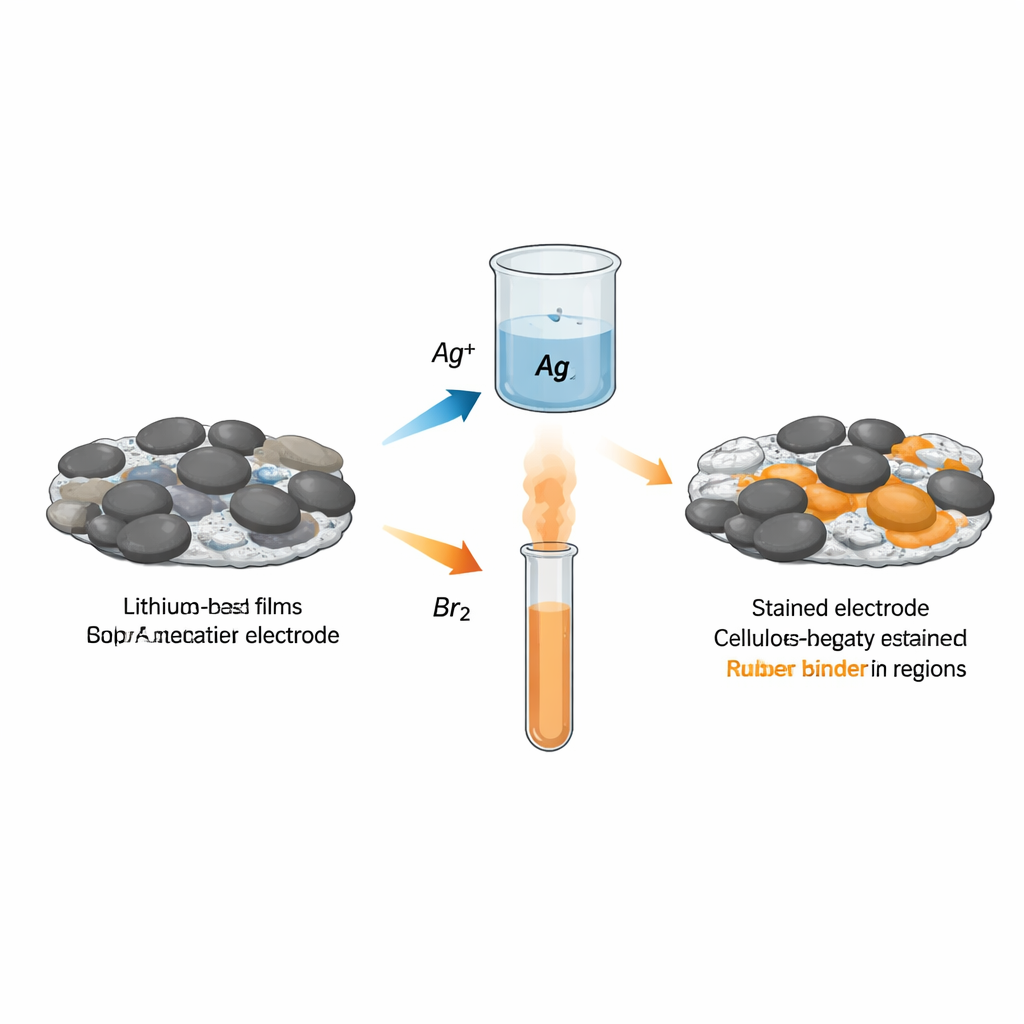
Why Binder Placement Matters
In a typical negative electrode, over 95% of the volume is active material such as graphite; less than 5% is binder and conductive carbon that provide mechanical strength and electrical pathways. Despite its tiny fraction, the binder’s spatial distribution strongly affects how well particles stick to each other and to the metal current collector, how easily electrons and ions move, and how stable the surface layers are during cycling. Until now, mapping where water-based binders go inside commercial graphite and graphite–silicon electrodes has been extremely difficult, so engineers mostly changed binder chemistry rather than its placement.
Making the Invisible Binder Light Up
The authors introduce two simple chemical stains tailored to the most widely used water-processable binders: carboxymethyl cellulose (CMC) and styrene–butadiene rubber (SBR). Immersing an electrode in a silver nitrate solution causes silver ions to latch selectively onto acidic groups in CMC, while exposing it to bromine vapor adds bromine atoms to the carbon–carbon double bonds in SBR. These added silver or bromine atoms are heavy enough to stand out in backscattered electron images and can be precisely measured by X‑ray spectroscopy. Tests on pure binder films and mixed electrodes confirm that silver tracks CMC and bromine tracks SBR with good specificity and sensitivity over realistic binder contents.
Revealing Hidden Films and Fragile Structures
With the stained electrodes in hand, the team used advanced electron imaging to explore binder organization at multiple scales. On the micrometer level, they identified distinct types of binder-rich clusters: some dominated by conductive carbon and CMC that help electrons percolate, and others richer in rubbery SBR that contribute elasticity. On the nanometer scale, the silver stain revealed an ultra-thin, roughly 10–15 nanometer CMC film that conformally coats graphite particles in fresh, uncompressed electrodes. This continuous coating had long been theorized but rarely seen directly. Strikingly, industrially relevant calendering (the hot rolling step used to densify electrodes) shattered this fragile film into scattered patches, leaving large areas of graphite bare in both lab-made and commercial samples. That patchiness likely alters where ions can enter, where protective surface layers form, and where damaging lithium plating may begin.
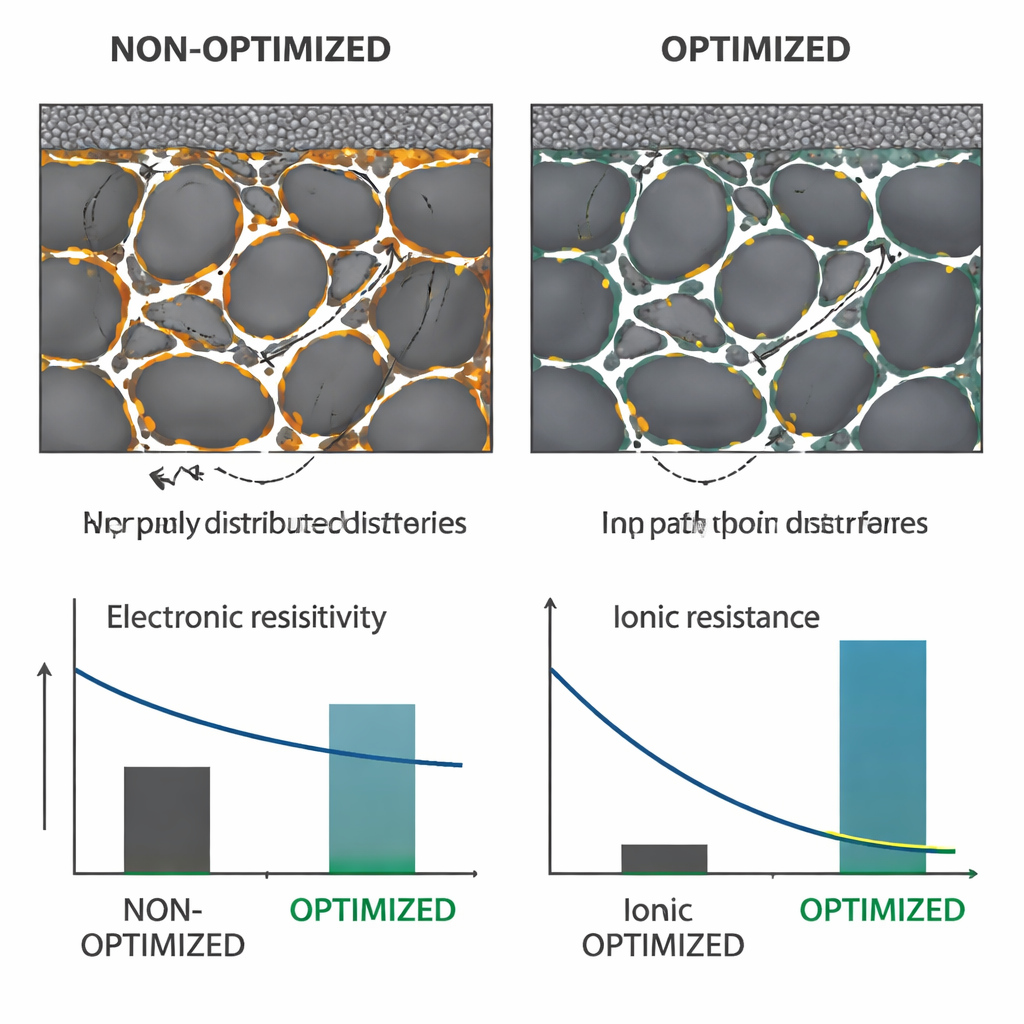
Turning Images into Better Manufacturing
Because the stained binders are now measurable, the authors could link microstructure to performance and process choices. By tweaking how the slurry is mixed—specifically, starting with a more concentrated CMC solution—they greatly reduced the formation of large carbon–binder clusters without changing the overall recipe. This led to a measurable 14% drop in electronic resistivity of the graphite coating. In a second study, they used staining to track binder migration during rapid high‑temperature drying, a key bottleneck in industrial coating lines. A simple “phase inversion” step—briefly dipping the wet coating into acetone before drying—drove more binder toward the current collector instead of the top surface. The resulting electrodes bent without cracking, adhered better, and showed about 40% lower ionic resistance through their pores, all without changing thickness, porosity, or composition.
Limits, Opportunities, and What It Means for Batteries
The staining method is not universal: highly reactive materials such as nano‑silicon or lithium iron phosphate can interfere with the chemistry, and silver or bromine must be applied to test samples, not live cells. Even so, the approach works well for the dominant water-based binders in graphite and many silicon-containing anodes, using only modest lab equipment. For non-specialists, the key takeaway is that how the “glue” inside an electrode is arranged—down to tens of nanometers—can significantly influence power, lifetime, and safety. By giving manufacturers a clear picture of binder location, this work opens practical routes to faster drying, better mechanical robustness, and more uniform current distribution, ultimately helping create more reliable and efficient lithium-ion batteries.
Citation: Zankowski, S.P., Wheeler, S., Barthelay, T. et al. Chemical staining for fundamental studies and optimization of binders in Li-ion battery negative electrodes. Nat Commun 17, 1438 (2026). https://doi.org/10.1038/s41467-026-69002-1
Keywords: lithium-ion batteries, electrode binders, graphite anodes, electron microscopy, battery manufacturing