Clear Sky Science · en
A molecularly defined basalo-prefrontal-thalamic circuit regulates sensory and affective dimensions of pain in male mice
Why this brain circuit matters for pain
Pain is more than a sharp sting or dull ache; it also carries an emotional punch and can reshape our mood and behavior for years. Many chronic pain patients get little relief from current drugs and often face serious side effects. This study in male mice uncovers a precisely defined brain circuit that helps the brain dial pain up or down, both in terms of how strong it feels and how upsetting it is. By mapping and controlling this pathway, the work points toward new, more targeted ways to ease chronic pain without relying solely on traditional painkillers.
A hidden highway from thinking brain to pain hub
The researchers focused on the medial prefrontal cortex, a front‑of‑the‑brain region involved in decision‑making, emotion and pain control, and on the thalamus, a deep relay center that shapes both the intensity and unpleasantness of pain. They discovered that a specific group of cells in the medial prefrontal cortex, marked by a gene called Foxp2, forms a major output pathway to the thalamus. These cells sit mainly in the deepest cortical layer and send long fibers to particular thalamic nuclei, while largely ignoring other typical targets of the prefrontal cortex. Using tracing methods and whole‑brain mapping, the team showed that these Foxp2‑marked neurons strongly connect to three thalamic regions known to influence pain processing: the parataenial nucleus, the mediodorsal thalamus, and the ventromedial thalamus. 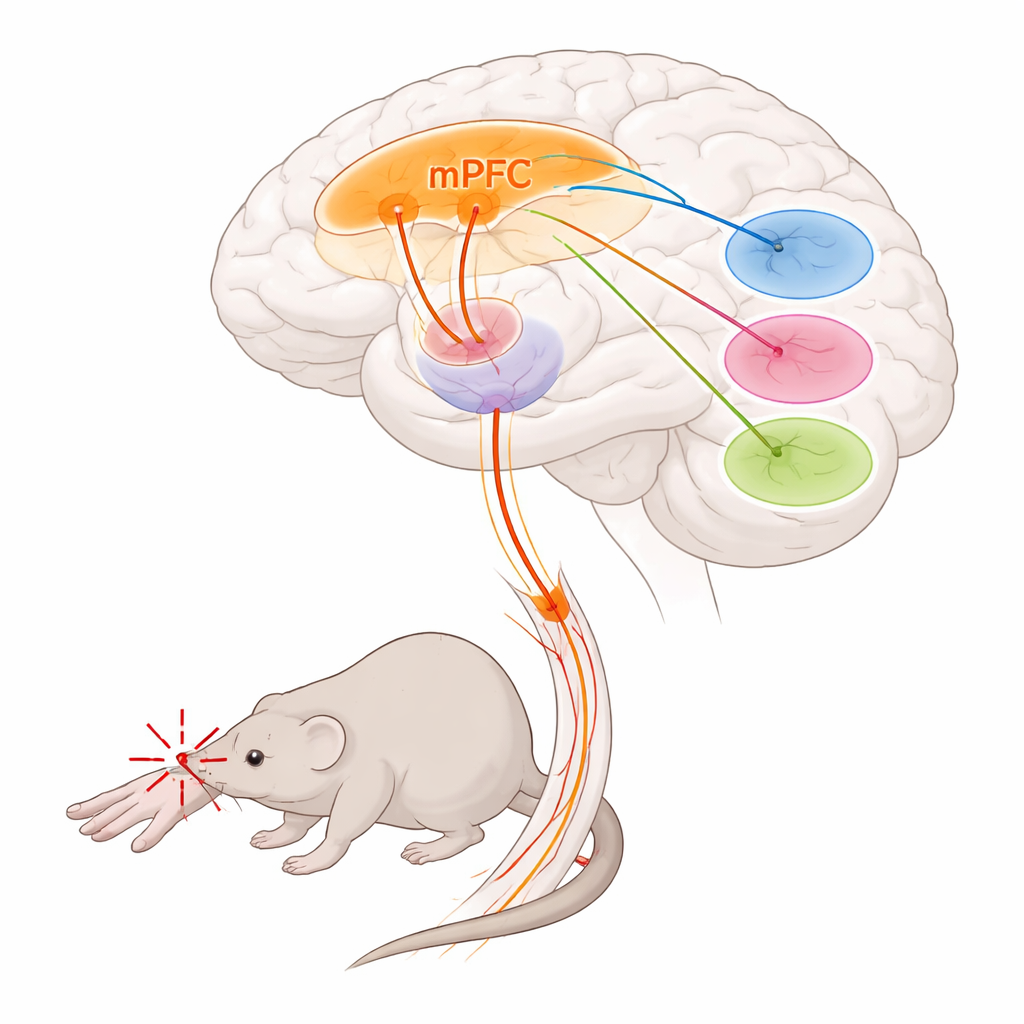
When pain hits, a key control center goes quiet
To see how this pathway behaves during pain, the scientists implanted tiny lenses above the medial prefrontal cortex and recorded the activity of individual Foxp2‑marked neurons in awake mice. During brief mechanical pinches, chemical pain induced by formalin, and long‑lasting inflammatory pain, these cells consistently quieted down: most showed fewer activity bursts during painful episodes and in the days following inflammatory injury. This shutdown was not subtle; in some conditions more than a third to nearly half of the cells became strongly less active. The finding suggests that acute and chronic pain states may, in part, reflect the silencing of a natural prefrontal braking system that would otherwise help keep pain in check.
Switching the circuit on and off reshapes pain
The team then asked whether this quieting was just a side effect of pain or a driver of it. Using genetic tools, they either blocked the output of Foxp2‑marked neurons long‑term or briefly boosted their activity. Persistent inactivation made mice more sensitive to touch and heat, indicating that this pathway normally restrains incoming pain signals. In contrast, short‑term activation raised the threshold for painful stimuli, reduced late‑phase licking in the formalin test (a measure of ongoing, inflammatory pain), and made mice prefer environments where the circuit had been turned on, signaling relief from the emotional burden of pain. By shining light on specific branches of the pathway, the researchers showed that projections to the parataenial thalamus reduce both the physical sensitivity and coping responses, while projections to mediodorsal and ventromedial thalamus mainly tune the emotional and behavioral dimensions. Together, these results reveal a finely split wiring scheme in which different branches of the same prefrontal population control different facets of the pain experience.
Chemical tuning: a cholinergic boost for pain relief
What controls these prefrontal pain‑relief neurons? Tracing experiments uncovered strong input from a cholinergic region in the basal forebrain called the horizontal diagonal band. Many of the cells sending signals from this area onto Foxp2‑marked neurons use acetylcholine, a messenger involved in attention and arousal. Activating this cholinergic projection reduced mechanical sensitivity and coping behaviors and eased inflammatory hypersensitivity, though it did not clearly shift the negative emotional tone of pain. On the receiving side, Foxp2‑marked neurons were found to be enriched for a particular acetylcholine receptor type, known as the α4β2 nicotinic receptor. Directly delivering a selective α4β2 agonist into the medial prefrontal cortex made mice less sensitive to painful heat and touch, even in chronic inflammatory states, and boosted activity in Foxp2‑marked neurons. Blocking these neurons removed the drug’s benefit, showing that this receptor relieves pain specifically by exciting this cell type. 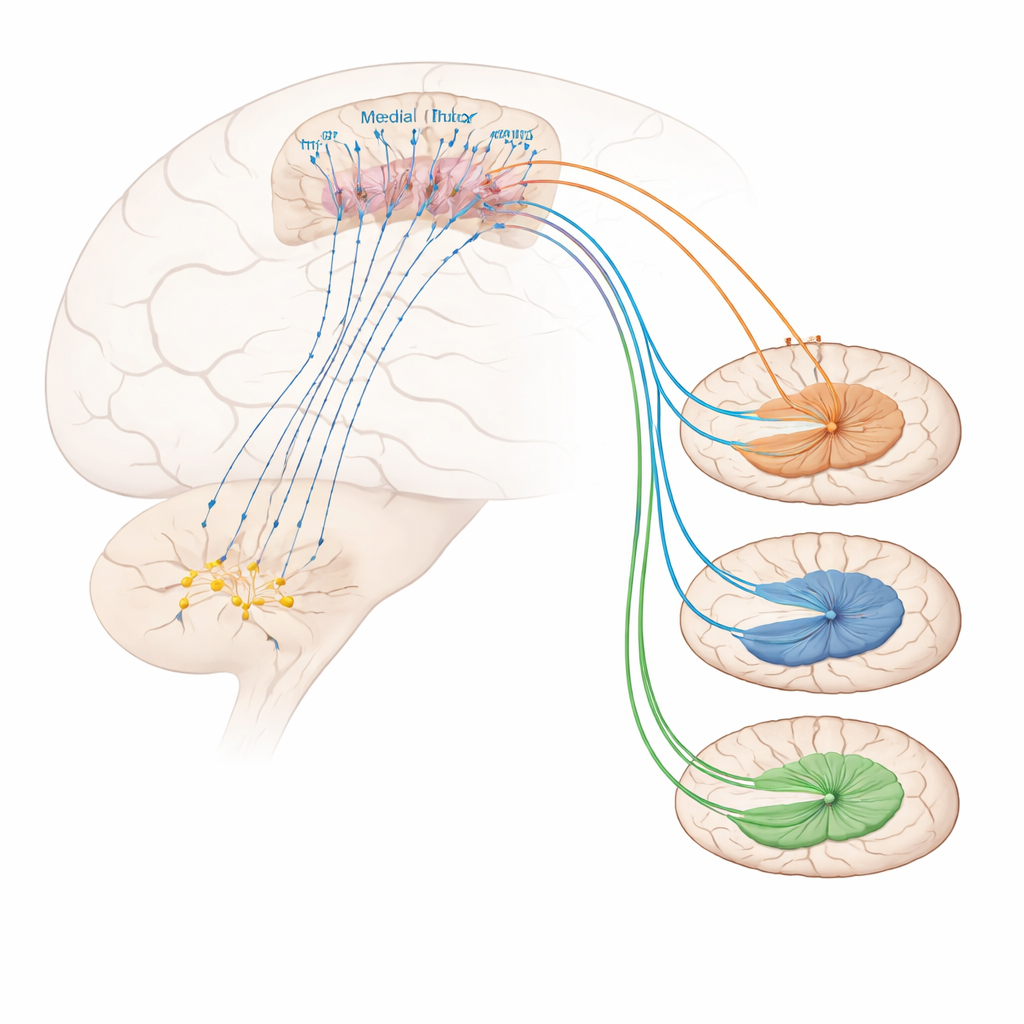
What this means for future pain treatment
In everyday terms, the study identifies a precise three‑step pathway: a cholinergic “starter” region in the basal forebrain, a specialized set of control cells in the medial prefrontal cortex, and distinct relay hubs in the thalamus that separately govern how strong pain feels, how bad it feels emotionally, and how we react to it. Pain appears to turn down this circuit; reactivating it, either by direct stimulation or by targeting its acetylcholine receptors, lessens both the sting and the distress of inflammatory pain in male mice. While much work remains to translate these findings to humans and to other forms of chronic pain, the results highlight a promising strategy: instead of numbing all sensation, future treatments might restore or fine‑tune the brain’s own pain‑relief circuits for more effective and possibly safer long‑term therapy.
Citation: Xie, G., Liu, Y., Qi, X. et al. A molecularly defined basalo-prefrontal-thalamic circuit regulates sensory and affective dimensions of pain in male mice. Nat Commun 17, 2134 (2026). https://doi.org/10.1038/s41467-026-69001-2
Keywords: chronic pain, medial prefrontal cortex, thalamus, cholinergic signaling, nicotinic receptors