Clear Sky Science · en
Microbiota-induced EI24 improves homeostasis but impedes function of alveolar macrophages via metabolic regulation
Why tiny lung guardians matter
Every breath you take brings not only oxygen into your lungs, but also dust, microbes, and other intruders. Patrolling this frontier are alveolar macrophages—specialized immune cells that swallow debris and help fend off infections and cancer. This study uncovers a surprising way in which our everyday microbes "train" these cells: a molecule called EI24, switched on by the microbiota, keeps lung sentinels stable and quiet, but at the cost of dulling their ability to fight viruses and tumors. Understanding this trade-off could open new avenues for safer, more effective immunotherapies.
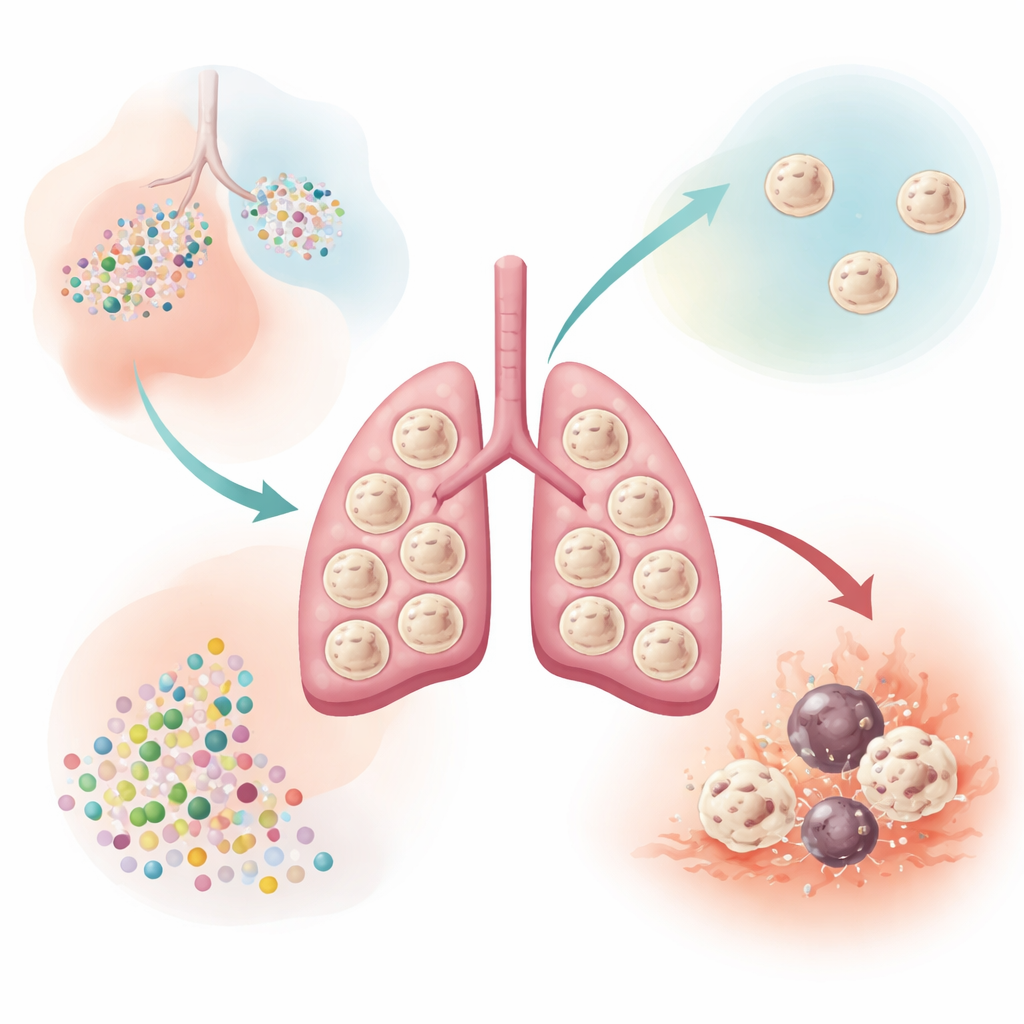
Meet the lung’s live-in cleaners
Alveolar macrophages sit in the tiny air sacs of the lungs, where gas exchange occurs. They live there for years, renewing themselves and quietly clearing out surfactant, dead cells, and stray particles while avoiding unnecessary inflammation that would damage the delicate tissue. The authors focused on a protein called EI24, previously linked to cell stress responses and cellular recycling, but not well understood in lung immunity. They discovered that among many types of tissue macrophages in the body, alveolar macrophages express especially high levels of EI24, hinting that this protein might be crucial for life on the lung’s exposed surface.
Stability versus strength in lung defenders
To see what EI24 actually does, the researchers bred mice lacking this protein specifically in macrophages. These animals developed normally and produced normal numbers of young macrophage precursors in the lung, but as they matured, about half of their alveolar macrophages disappeared. The surviving cells showed clear signs of increased cell death driven by a classic death enzyme, caspase-3. Yet these fewer remaining cells were not weak: they displayed a more "on-duty" profile, with higher uptake of bacteria in lab tests and stronger production of inflammatory molecules when stimulated. Genetic and chromatin analyses showed that hundreds of genes involved in antigen presentation, cell killing, and inflammatory responses were turned up, while regulatory pathways that normally keep activation in check were loosened.
Revved-up metabolism with a hidden cost
Digging deeper, the team found that EI24-deficient macrophages had rewired their metabolism. Instead of relying mostly on a calm, energy-efficient state, they showed higher levels of both sugar-burning (glycolysis) and mitochondrial energy production. These boosted energy pathways helped fuel stronger inflammatory responses and more vigorous swallowing of microbes and tumor cells. But this metabolic overdrive also increased reactive oxygen by-products inside mitochondria, which in turn activated caspase-3 and drove the cells toward programmed death. Blocking these metabolic pathways or mopping up the reactive molecules reduced both the excess inflammation and the tendency to die, tying together the cell’s energy use, killing power, and lifespan.
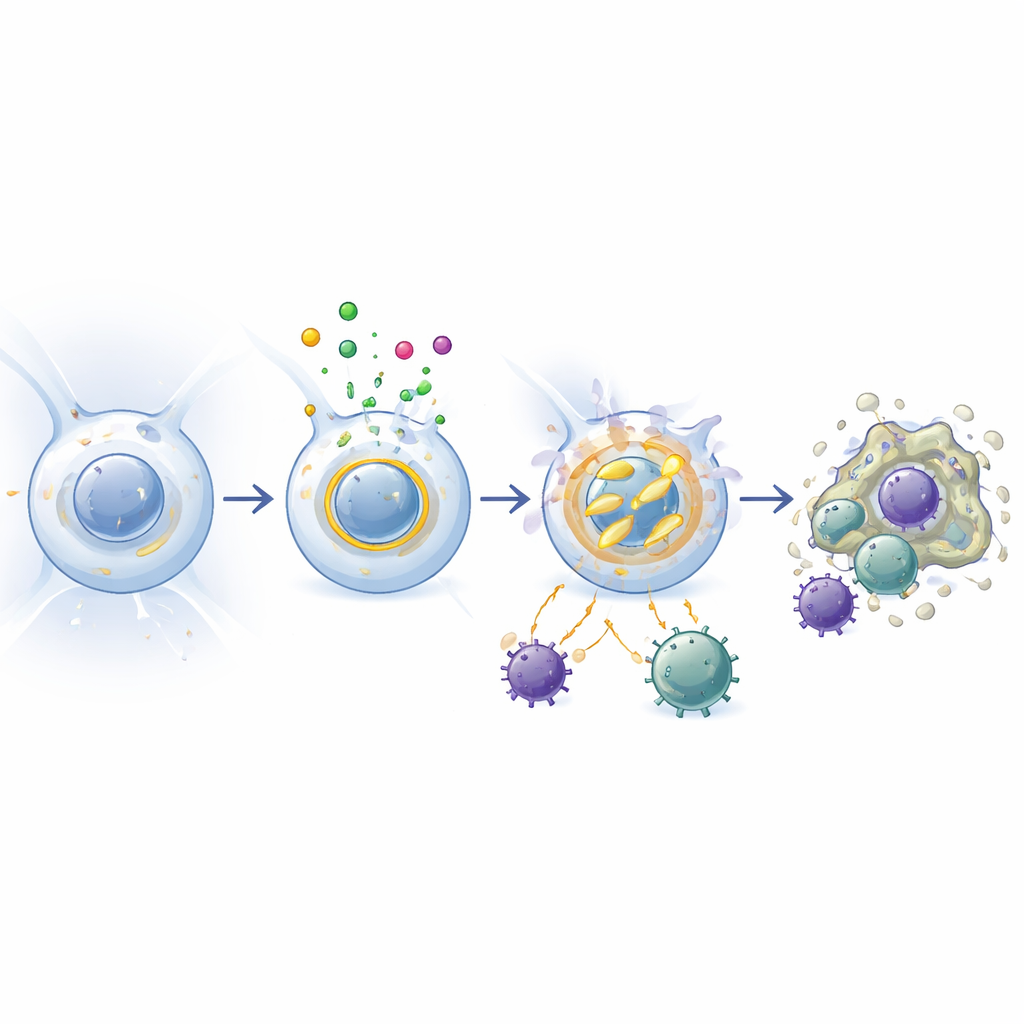
How friendly microbes set the dial
The lung is not a sealed chamber; it constantly encounters harmless and beneficial microbes from the air and from the gut. The researchers compared normal mice with germ-free animals that were raised without any microbes. In germ-free mice, alveolar macrophages made much less EI24, and removing EI24 had little impact on their numbers or behavior. When these germ-free mice were later exposed to normal microbes, their lung macrophages ramped up EI24. Experiments using microbial sensor pathways showed that signals detected by Toll-like receptors 2 and 4—molecular "doorbells" for bacterial components—were responsible for this increase. In effect, the microbiota pushes alveolar macrophages into a more stable, less reactive state by turning up EI24, helping maintain calm in an environment that would otherwise see constant immune alarms.
Turning down EI24 to boost therapy
Despite having fewer macrophages, mice lacking EI24 in these cells were better protected against severe influenza infection and experimental lung metastases from melanoma. They cleared virus more efficiently, produced more antiviral interferons in the airspaces, and their macrophages more readily engulfed tumor cells. Importantly, these animals did not develop chronic lung damage or loss of lung function, suggesting that selectively removing EI24 can enhance defense without obvious harm in the short to medium term. The team also showed that bone marrow–derived macrophages engineered to lack EI24 were more effective when transferred into other mice, outperforming normal macrophages in limiting both viral infection and tumor spread in the lungs.
What this means for future treatments
For a general reader, the central message is that our resident microbes help keep lung immune cells alive and calm by turning on EI24—but this same safety mechanism can blunt the cells’ ability to fight infections and cancer. By carefully dialing down EI24, scientists can push macrophages into a more energetic, aggressive state that better eliminates viruses and tumor cells, while still preserving overall lung health in experimental animals. This work suggests that targeting EI24, or the metabolic circuits it controls, could one day make macrophage-based therapies more potent allies against respiratory infections and metastatic cancer.
Citation: Huang, Y., Su, M., Zhang, Y. et al. Microbiota-induced EI24 improves homeostasis but impedes function of alveolar macrophages via metabolic regulation. Nat Commun 17, 2227 (2026). https://doi.org/10.1038/s41467-026-69000-3
Keywords: alveolar macrophages, lung immunity, microbiota, macrophage metabolism, cancer and viral defense