Clear Sky Science · en
Covalent modification of a glutamic acid inspired by HaloTag technology
Turning Protein “Anchors” into Drug Targets
Many modern cancer drugs work by latching onto proteins inside our cells. But some of the most important proteins have no easy “handles” for medicines to grab. This study introduces a clever chemistry trick, inspired by a popular lab tool called HaloTag, to latch onto a normally hard‑to‑reach spot on a protein that helps control cancer‑relevant growth signals. The work could open new routes to drugs that shut down tumour‑driving pathways in a more durable way.
Why Most Covalent Drugs Aim for the Same Spot
In recent years, so‑called targeted covalent drugs have become an exciting class of medicines. They carry a mildly reactive chemical group that forms a permanent bond to a specific amino acid on a protein, locking the drug in place. Almost all of these drugs go after cysteine, a relatively rare but highly reactive amino acid. By contrast, two other amino acids, aspartate and glutamate, are far more common and often crucial for a protein’s shape and function, but their acidic “carboxylate” groups are much less reactive in the watery environment of a cell. That makes them difficult to modify selectively, and only a few successful examples of glutamate‑ or aspartate‑targeting covalent drugs existed before this work.
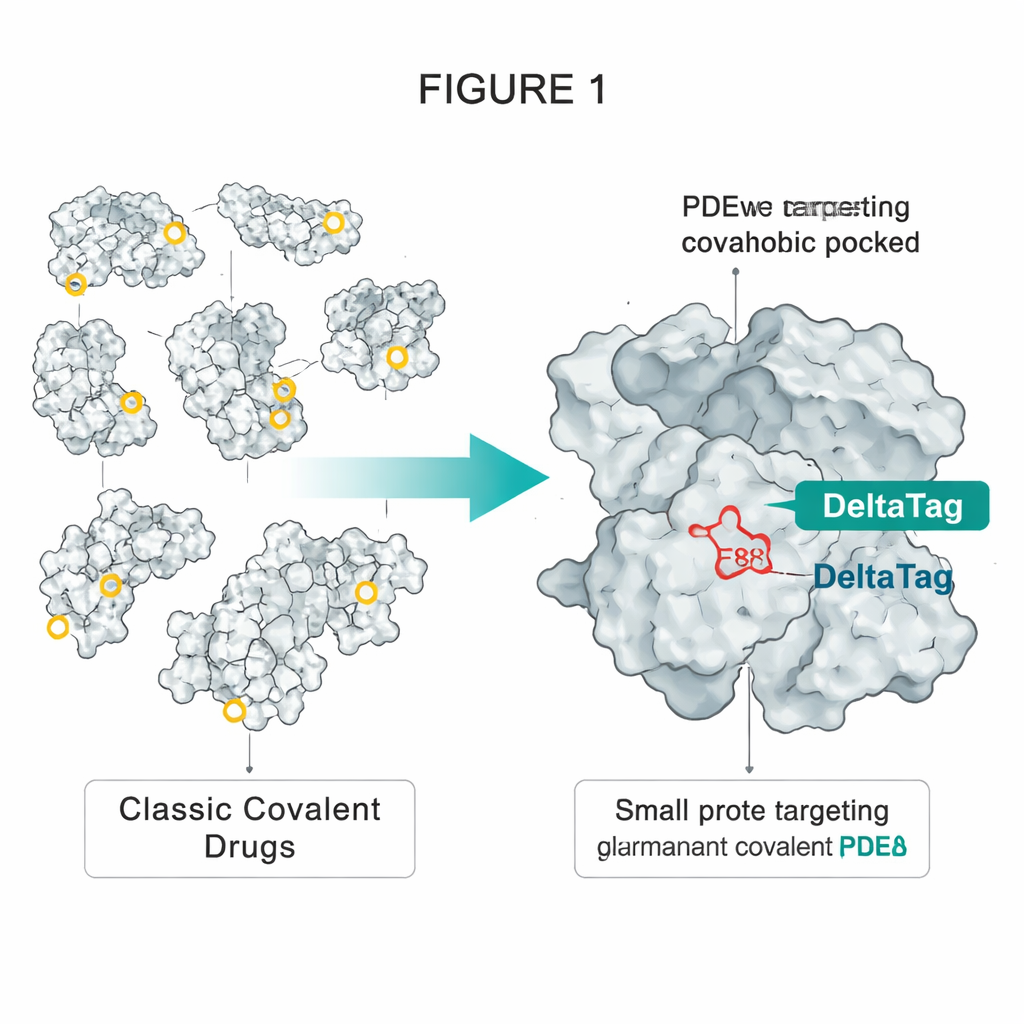
Borrowing a Trick from HaloTag Technology
The authors took inspiration from HaloTag, a widely used engineered protein that can be permanently tagged with fluorescent dyes. In HaloTag, a specially placed aspartate deep inside a greasy pocket reacts with a simple chloroalkane chain on the dye, forming a stable ester bond. The team noticed that another protein, PDEδ, has a somewhat similar hydrophobic pocket that contains a single accessible glutamate, called E88. PDEδ ferries lipid‑modified signaling proteins such as the small GTPase Rheb through the cell, helping them reach membranes where growth‑promoting complexes like mTORC1 are activated. Earlier non‑covalent inhibitors of PDEδ could block this shuttle, but their effect was limited because another protein, Arl2, can pry them out of the pocket over time.
Designing DeltaTag to Lock onto Glutamate
To overcome this “kick‑out” problem, the researchers started from a known high‑affinity PDEδ blocker and re‑engineered one of its side chains to carry a haloalkane “warhead” similar to those used in HaloTag ligands. Through several rounds of structural tweaking guided by protein crystal structures, they arrived at a compound called DeltaTag. Its key feature is a phenethyl bromide group positioned so that, once the molecule nestles into PDEδ’s lipid pocket, the bromide is precisely aligned to react with E88. Biophysical measurements and high‑resolution X‑ray structures confirmed that DeltaTag forms a covalent ester link specifically with this glutamate and that other potentially more reactive amino acids on PDEδ remain untouched. The compound is reactive enough to label the protein efficiently, yet stable enough in water and in the presence of the cell’s main sulphur‑based antioxidant, glutathione, to avoid widespread, indiscriminate damage.
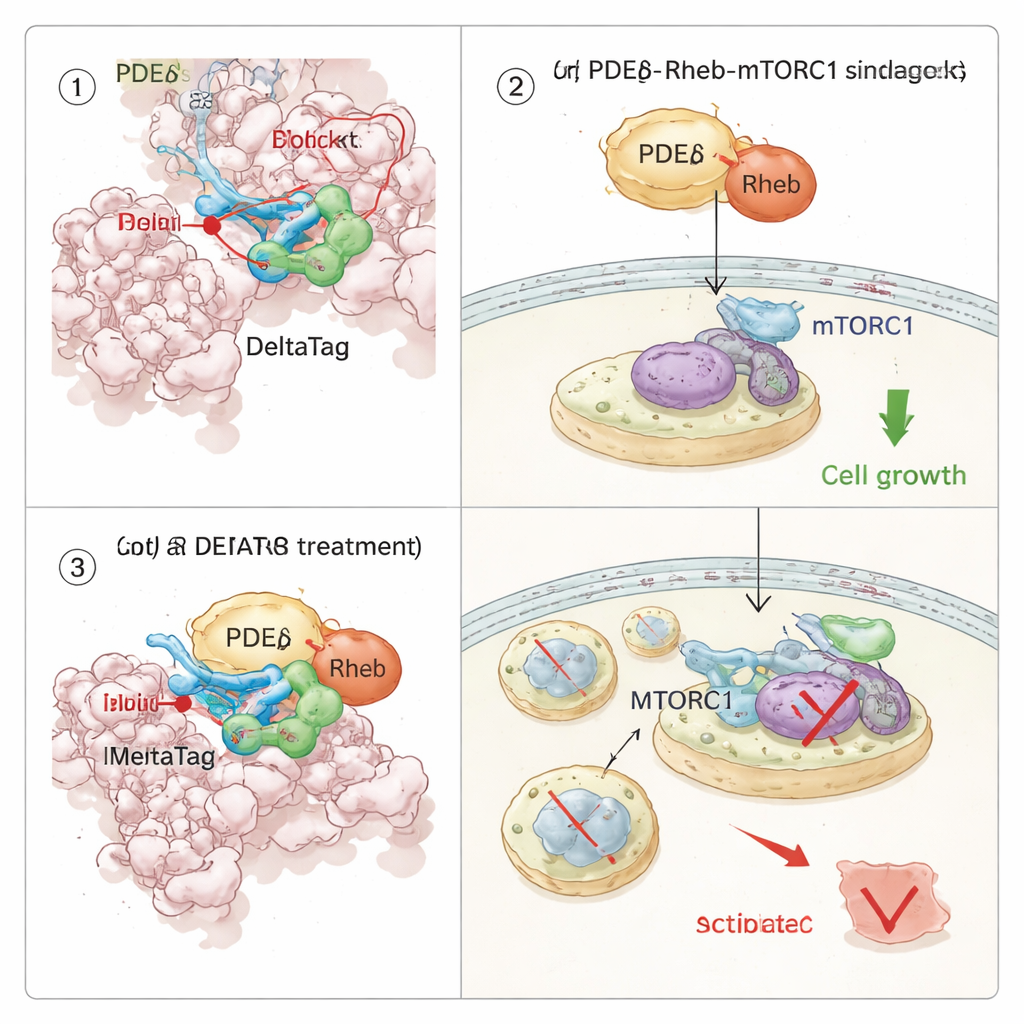
Re‑wiring a Growth Pathway Inside Cancer Cells
Because PDEδ chaperones Rheb, and Rheb in turn switches on the mTORC1 growth pathway, the team asked how covalent inhibition of PDEδ would ripple through cell signalling. Using thermal stability profiling across thousands of proteins, they showed that DeltaTag binds selectively to PDEδ in cell extracts and perturbs proteins linked to the Rheb–mTOR axis. In live‑cell imaging experiments, DeltaTag caused Rheb to relocate from a diffuse pattern in the cell interior to more clustered internal membranes, consistent with disruption of its normal shuttling. Global phosphoproteomics and focused Western blotting revealed that mTORC1 activity, tracked by phosphorylation of a ribosomal protein called S6, dropped over time, while compensatory signals in related pathways rose. Importantly, in several human cancer cell lines driven by mutant KRAS and dependent on strong mTOR signalling, DeltaTag slowed cell proliferation more effectively than closely related reversible inhibitors, and its impact was weaker in cells lacking PDEδ, supporting that its main action is on‑target.
Opening a Door to New Kinds of Drug Targets
This work shows that it is possible to build small molecules that form a permanent, highly specific bond to a single glutamate buried in a greasy pocket on a protein, using a relatively simple haloalkane chemistry borrowed from HaloTag. In the model system PDEδ, that covalent link yields a more durable block of a cancer‑relevant growth pathway than earlier reversible drugs. More broadly, the same design logic could be applied to other proteins that have hydrophobic cavities but lack the usual “reactive” amino acids, as long as they present a strategically placed glutamate or aspartate. With further optimisation, glutamate‑targeting covalent inhibitors like DeltaTag may become useful tools for chemical biology and, ultimately, leads for new therapies that tackle currently hard‑to‑drug proteins.
Citation: Zhang, R., Liu, J., Gasper, R. et al. Covalent modification of a glutamic acid inspired by HaloTag technology. Nat Commun 17, 1257 (2026). https://doi.org/10.1038/s41467-026-68999-9
Keywords: covalent inhibitors, glutamate targeting, PDEδ, mTOR signalling, cancer drug design