Clear Sky Science · en
Engineering synthetic cells with intramembrane domains possessing distinct bilayer asymmetries
Why Tiny Synthetic Bubbles Matter
Every living cell is wrapped in a membrane that behaves like a smart, flexible skin. This skin is not uniform: the inner and outer sides, and even different patches along its surface, can have very different makeups and shapes. Those differences help cells send signals, bind proteins, and even divide. Yet it has been remarkably hard to build simple, controllable model membranes that capture this complexity. This study introduces a practical way to make cell-sized “synthetic cells” whose membranes are asymmetric on both sides and laterally patterned into distinct regions—opening a route to more realistic cell mimics and to studying how membrane patterns drive budding and division. 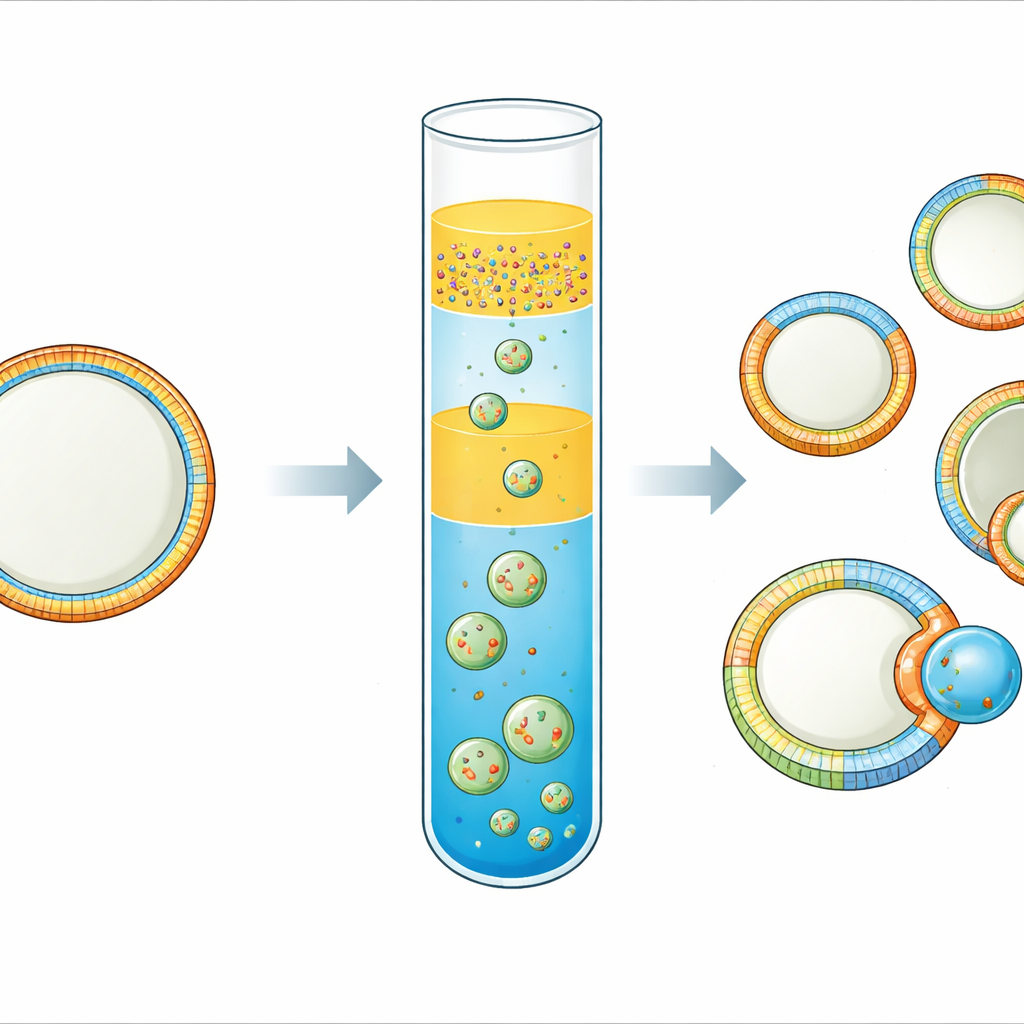
Building Better Cell Mimics
Real cell membranes are asymmetric: the inner side is rich in negatively charged lipids that recruit signaling proteins, while the outer side carries different lipids that mediate cell–cell recognition. Many lab methods can make giant vesicles—hollow spheres of fat-like molecules—but their two leaflets usually end up the same, and the inside is hard to access. Other approaches that do create asymmetry often require complex devices, special lipids, or leave the vesicles stuck to a surface. The authors set out to create free-standing, cell-sized vesicles that combine high encapsulation of biomolecules with robust, tunable asymmetry more like that of a living cell.
A Three-Layer Recipe for Asymmetric Membranes
The team refined an “inverted emulsion” technique, in which tiny water droplets coated with lipids are driven through an oil–water interface to form vesicles. Their key innovation is a triple-layer oil setup. At the bottom sits an oil containing the lipids destined for the outer leaflet of the membrane. Above this, they place a thin spacer oil that has no lipids, and on top of that an emulsion of water droplets surrounded by a second oil that carries the lipids for the inner leaflet. During a brief centrifugation step, the droplets fall through the spacer and bottom layer, picking up a second lipid coat and closing into vesicles. Choosing two oils of different density and including the spacer sharply limits mixing between the lipid pools, so the inner and outer leaflets retain distinct compositions.
Testing That the Two Sides Stay Different
To verify that they had truly different leaflets, the researchers used both chemical and protein-based readouts. In one set of tests, they added a fluorescent lipid to only one leaflet and then exposed vesicles to a chemical in the outside solution that quenches the fluorescence but cannot cross the membrane. When the fluorescent lipid was on the outer side, the signal dropped almost completely; when it was on the inner side, it hardly changed, showing that most labeled lipids stayed where intended. In a second set of experiments, they placed lipids that bind specific proteins—such as biotin-tagged lipids for streptavidin or special headgroups for His-tagged green fluorescent protein—on only one leaflet. Proteins added to the opposite compartment (inside or outside) bound only when their matching lipid was on the same side of the membrane, confirming both strong asymmetry and preserved protein function.
Creating Rafts and Watching Membranes Bud
Real cell membranes also contain “rafts”: tiny patches enriched in certain lipids like cholesterol and sphingomyelin, which can cluster proteins and influence shape. Achieving such phase separation with the inverted emulsion method had been difficult because cholesterol tended to stay dissolved in oil rather than join the membrane. By using squalene in the bottom oil layer, which holds cholesterol less strongly than standard mineral oil, the authors could reproducibly form vesicles whose membranes split into coexisting liquid-ordered and liquid-disordered regions, much like rafts. They then combined this lateral patterning with leaflet asymmetry: one domain carried an inner-leaflet ligand binding streptavidin inside, while another domain carried glycolipids that bound cholera toxin outside. 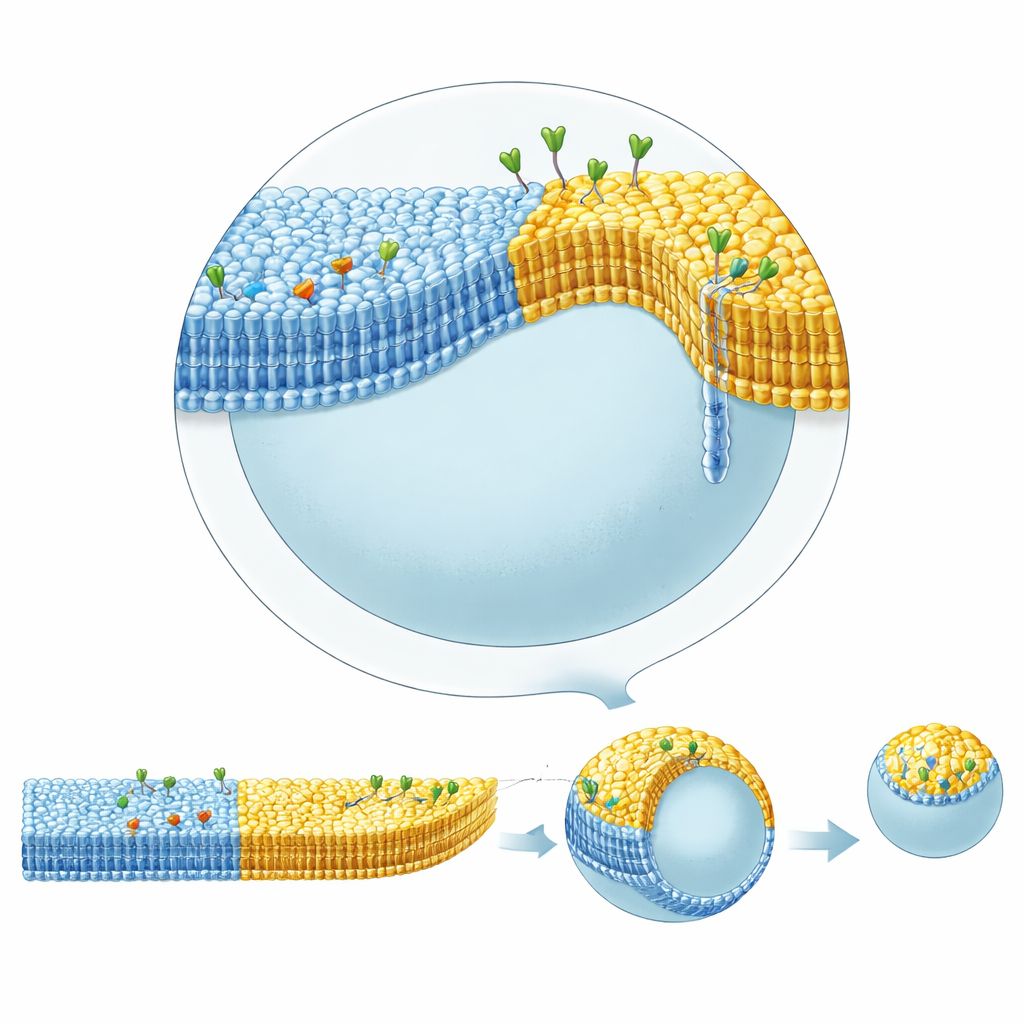
How Proteins and Patterns Drive Shape Changes
These complex vesicles displayed striking shape dynamics. When cholera toxin bound to glycolipids in the ordered domain on the outer side, that domain bulged outward into a curved bud connected by a narrow neck, and under slight osmotic deflation could pinch off entirely as a daughter vesicle. This budding did not require energy-carrying molecules like ATP or specialized division machinery; it arose purely from the interplay of lipid composition, domain boundaries, and asymmetric protein binding. By increasing the amount of streptavidin bound on the inner side of the disordered domain, the team could counteract the outward bending of the ordered domain and gradually restore a more spherical shape. Detailed geometric analysis of vesicle contours showed that the curvature of the membrane jumps at the boundary between domains, and that the narrow necks closing during budding must withstand forces of at least tens of piconewtons—quantifying how domain structure and protein crowding can mechanically sculpt membranes.
What This Means for Future Synthetic Cells
In everyday terms, the authors have developed a recipe for making soft, cell-sized bubbles whose skins are patterned and two-sided in much the same way as real cell membranes. Because the method is relatively simple, works with many different lipids, and allows proteins and other large molecules to be trapped inside, it paves the way for building more lifelike artificial cells from the bottom up. Such synthetic cells could help scientists dissect how membrane patterns control signaling, traffic, and division, and they may eventually be harnessed as smart drug carriers that change shape or split on cue inside the body.
Citation: Yandrapalli, N., Seemann, T., Lipowsky, R. et al. Engineering synthetic cells with intramembrane domains possessing distinct bilayer asymmetries. Nat Commun 17, 2620 (2026). https://doi.org/10.1038/s41467-026-68997-x
Keywords: synthetic cells, lipid membranes, membrane asymmetry, phase separation, membrane budding