Clear Sky Science · en
Bioinspired charge reservoir enables efficient CO2 photoreduction with H2O via tungsten valence oscillation
Turning Air and Water into Fuel
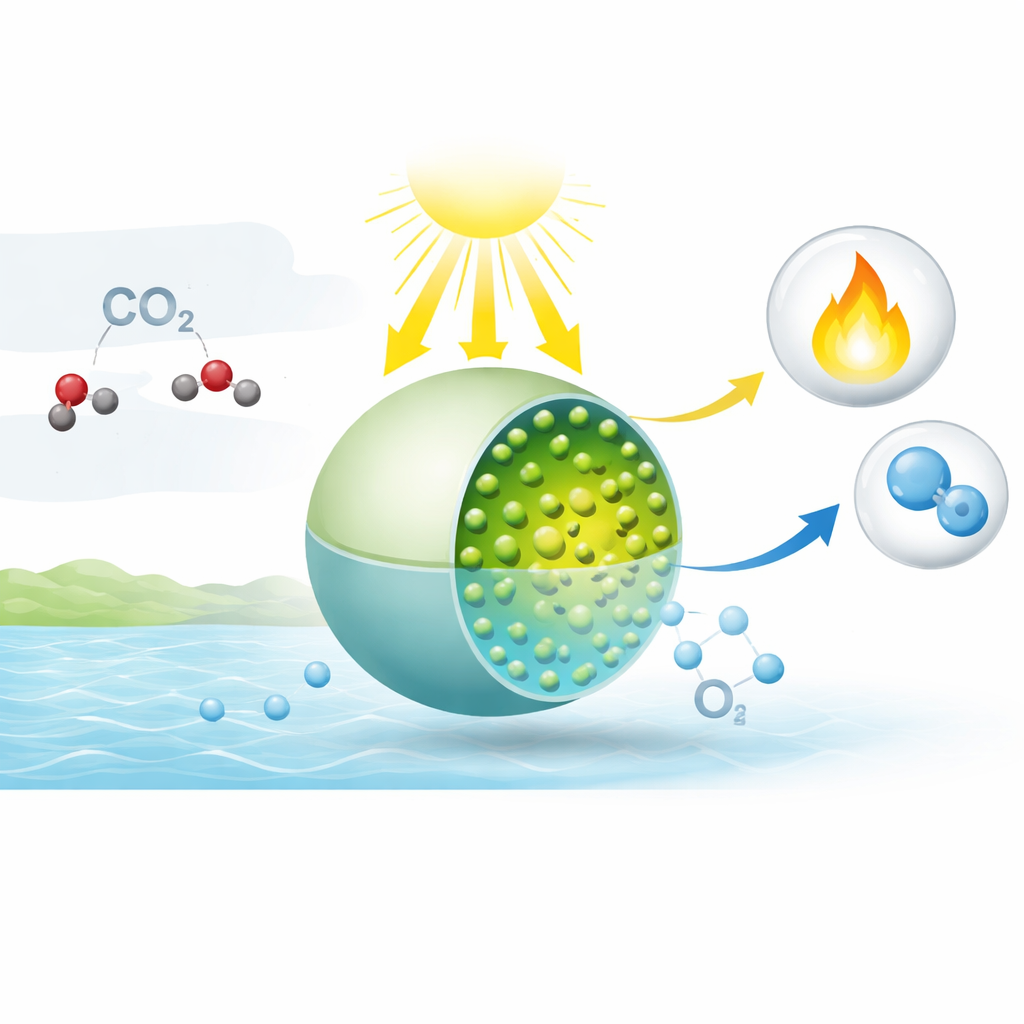
Burning fossil fuels adds carbon dioxide to the air, warming the planet and wasting the sun’s free energy. This study explores a different path: using sunlight to turn carbon dioxide and water directly into useful fuels, much like plants do during photosynthesis. The researchers borrow a clever trick from nature’s playbook to manage fleeting electrical charges, making this sunlight-driven chemistry work more efficiently and without relying on wasteful additives.
Lessons Borrowed from Green Leaves
In natural photosynthesis, two light-harvesting units in plant cells share the workload. One splits water, releasing oxygen and freeing electrons; the other uses those electrons to turn carbon dioxide into energy-rich molecules. Crucially, plants use a small carrier molecule, plastoquinone, to temporarily hold and shuttle electrons so they do not vanish before they can do useful work. The team behind this paper set out to build an artificial version of that temporary storage system, so that water splitting and carbon dioxide conversion could each proceed at their own pace while still remaining tightly linked.
A Tiny Battery Hidden in a Mineral Particle
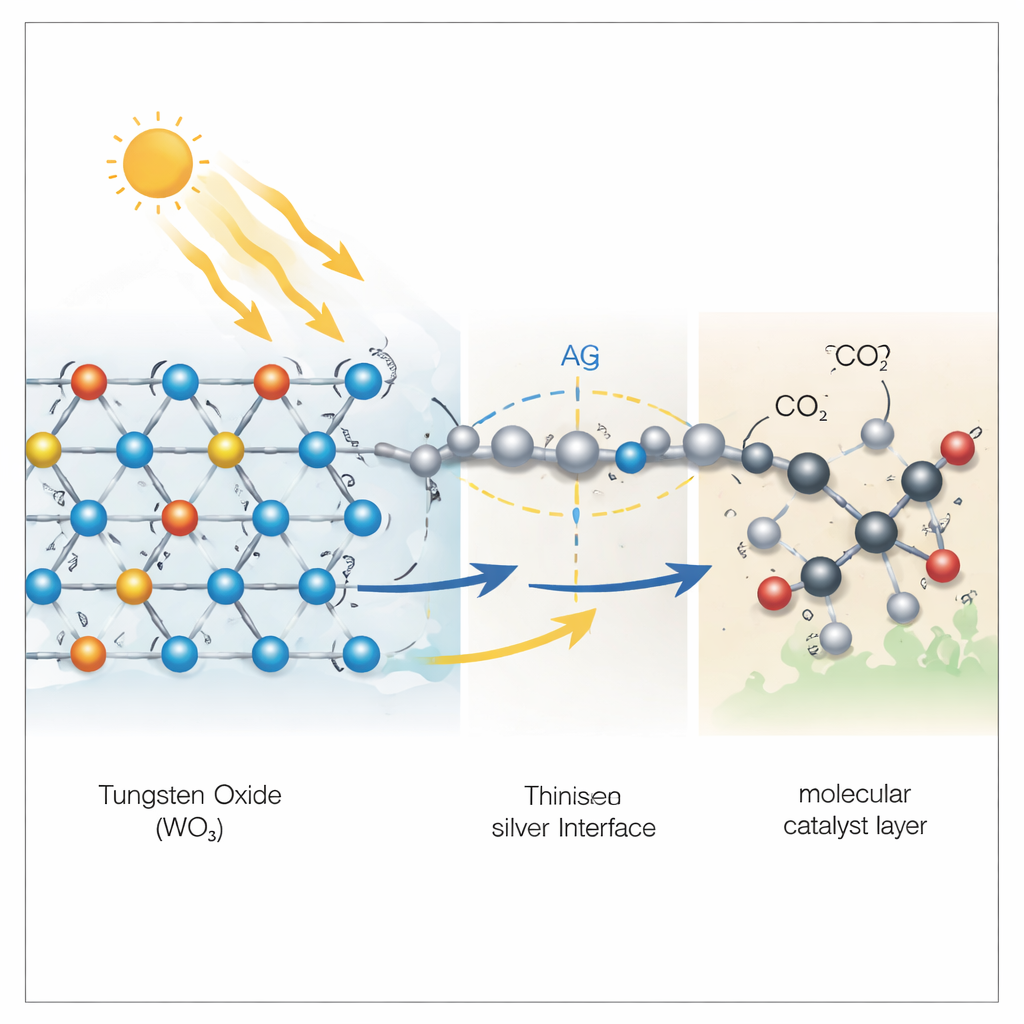
The researchers designed a material based on tungsten trioxide, a yellow mineral-like solid, decorated with single atoms of silver. Under light, tungsten atoms in this solid can flip between two charge states, acting like tiny rechargeable sites that soak up extra electrons and release them later. In this design, the silver-modified tungsten trioxide (called Ag/WO3) behaves like a miniature charge reservoir, much like plastoquinone in plants. Experiments showed that when the material is lit, it stores long-lived electrons inside its structure and can later pass them on to other substances that need them to drive chemical reactions.
Helping Catalysts Do the Hard Work
On its own, Ag/WO3 does not turn carbon dioxide into fuel very efficiently. The breakthrough comes when it is paired with “active components” that specialize in the carbon chemistry, such as a cobalt-containing dye-like molecule (cobalt phthalocyanine), a polymeric material called carbon nitride, or copper oxide. These partners are good at turning carbon dioxide into carbon monoxide or methane but tend to lose efficiency because their electrons and holes quickly cancel each other out. When they are coupled with Ag/WO3, the stored electrons in the tungsten material selectively remove the unwanted positive charges (holes) from the active component. This keeps a high density of useful electrons at the sites where carbon dioxide is reduced, dramatically boosting the rate of fuel-forming reactions.
Big Performance Jump and Everyday Sunlight
The most striking example is the combination of cobalt phthalocyanine with Ag/WO3. In pure water and under simulated sunlight, this hybrid produces carbon monoxide at a rate about 100 times higher than cobalt phthalocyanine alone, rivaling systems that need added organic “sacrificial” chemicals to mop up holes. Similar performance boosts were seen when pairing Ag/WO3 with carbon nitride or copper oxide, and the approach worked not only in a lab lamp setup but also outdoors under real sunlight. Careful measurements of how light-induced charges move and recombine confirmed that the tungsten–silver support repeatedly “charges” and “discharges,” stabilizing electrons and feeding them into the reaction just when and where they are needed.
A Versatile Blueprint for Sunlight Fuels
To a non-specialist, the main message is that the authors have built a tiny, rechargeable “buffer” for electrons that lets a wide range of catalysts turn carbon dioxide and water into fuel more efficiently, without burning through disposable helper chemicals. By separating the roles—one material dedicated to splitting water and storing charge, and another focused on reshaping carbon dioxide—the system becomes both more flexible and more robust. This bioinspired strategy offers a general blueprint for future solar-fuel devices that could one day convert sunlight, air and water into carbon-neutral fuels on a meaningful scale.
Citation: Huang, Y., Shi, X., Zhang, H. et al. Bioinspired charge reservoir enables efficient CO2 photoreduction with H2O via tungsten valence oscillation. Nat Commun 17, 2204 (2026). https://doi.org/10.1038/s41467-026-68991-3
Keywords: artificial photosynthesis, CO2 reduction, solar fuel, photocatalyst, tungsten oxide