Clear Sky Science · en
Stepwise hydrogen spillover–engineered synergistic sites enable near-quantitative conversion of waste PET to p-xylene
Turning Trash Bottles into Valuable Fuel
Plastic bottles and polyester clothes are convenient in daily life, but they leave behind mountains of stubborn waste. This study describes a new way to transform one of our most common plastics, polyethylene terephthalate (PET), almost completely into a single, high-value chemical called p-xylene, which is used to make fresh polyester and other products. In other words, the work points to a route for turning used packaging and textiles back into premium raw material, while cutting both costs and carbon emissions.
The Problem with Everyday Plastics
Modern society produces billions of tons of plastic, much of which ends up in landfills, rivers, and oceans. PET, the plastic in drink bottles, food containers, films, and many fabrics, makes up a large share of this waste. It is tough and chemically resilient, which is good for products but bad for recycling. Existing methods can break PET down, yet they often give a mix of different chemicals rather than one clean product, making purification difficult and expensive. Industry, however, needs extremely pure p-xylene as a cornerstone ingredient for new polyester fibers, solvents, and some specialized chemicals.
A Catalyst that Guides the Reaction
The researchers designed a solid catalyst made from copper and cobalt on an oxygen-containing support, referred to as CuCo/CoOx. In the presence of hydrogen gas and a suitable liquid solvent, this material drives PET to break apart and re-form into p-xylene with a yield above 99.9%—essentially quantitative. That performance is far better than that of simpler copper or cobalt catalysts, and even surpasses systems based on precious metals like platinum and ruthenium. The process works at moderate temperature and pressure, and the catalyst can be reused several times without losing activity, making it more realistic for industrial deployment.
How the Invisible Handoff of Hydrogen Works
At the heart of the catalyst’s success is a subtle phenomenon called stepwise hydrogen spillover. When the catalyst is heated under hydrogen, copper sites are reduced first and start to split hydrogen molecules into reactive atoms. These atoms move, or “spill over,” onto nearby cobalt oxide regions, helping convert part of the cobalt into metallic form. Once these particular cobalt sites are formed—especially those with a specific crystal structure—they become even better at splitting hydrogen, driving a second wave of spillover across the surface. This sequence creates a high density of special boundary regions where metallic cobalt touches cobalt oxide, and where missing oxygen atoms leave tiny vacancies. Experiments and computer simulations show that these interfaces are exceptionally good at both activating hydrogen and weakening the strong carbon–oxygen bonds in PET.
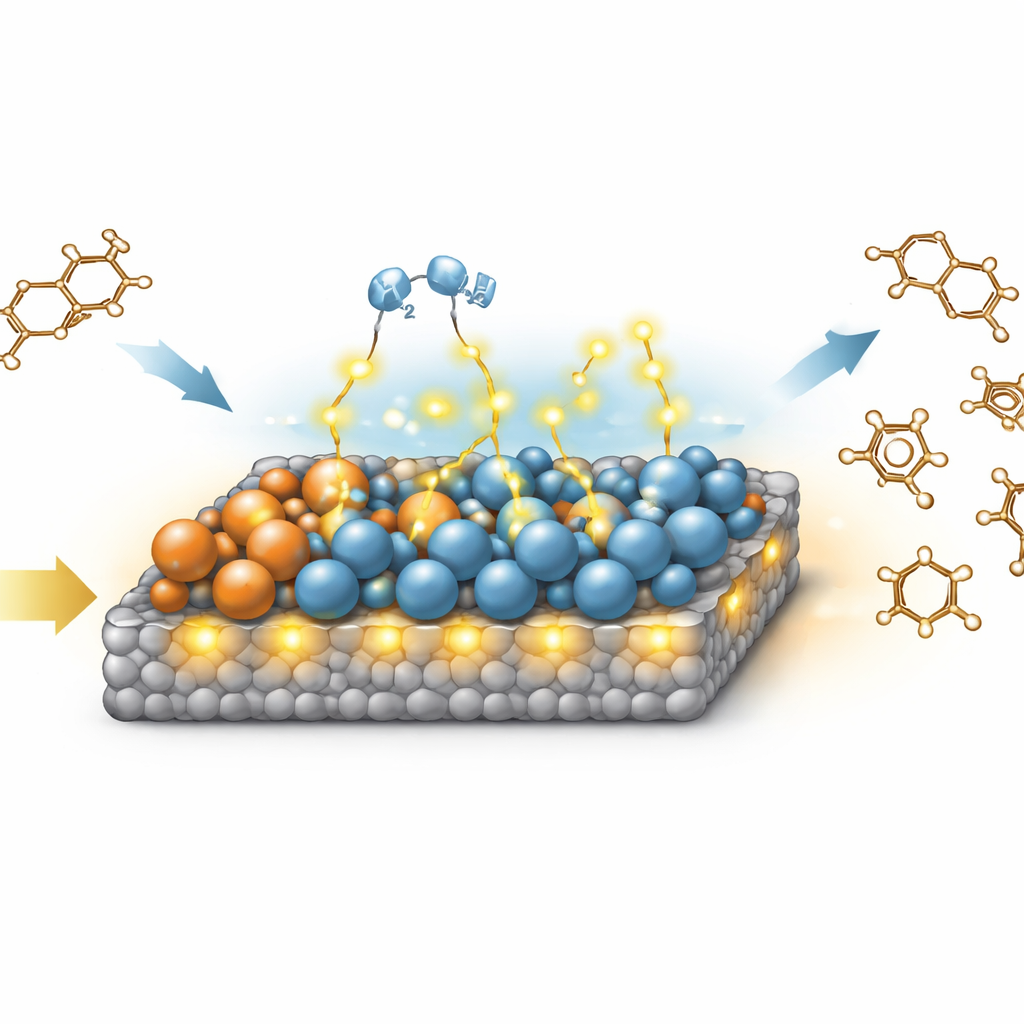
From Plastic Chains to Simple Rings
To follow what happens to PET itself, the team examined intermediate molecules produced under milder conditions. They found that the long PET chains first break into smaller pieces containing a benzene ring with short side arms. These fragments then undergo a series of hydrogen-driven trimming steps on the catalyst surface: first the ester links are cleaved, then the oxygen-containing groups are gradually removed. Along the way, fleeting aldehyde-like species appear, as detected by infrared spectroscopy, before finally giving p-xylene, a simple aromatic ring with two identical side groups. Importantly, the catalyst’s surface not only makes these steps fast; it also holds the starting material strongly while allowing the final p-xylene to leave easily, preventing the reaction from stalling or overreacting.
Real Waste, Real Benefits
The new catalyst is not limited to pure laboratory samples. It can handle more than two dozen real PET-based wastes, including bottles, cups, films, fabrics, and mixed plastic streams that contain other polymers and common additives. In most cases it still converts PET to p-xylene with near-perfect selectivity. An economic and environmental assessment suggests that using waste PET instead of oil-based feedstocks could cut the carbon footprint of p-xylene production by about one-third, while also lowering costs and more than doubling profit margins per kilogram of product. In simple terms, this approach turns used plastic from a growing environmental liability into a valuable chemical resource, offering a promising path toward a more circular and climate-friendly plastics economy.
Citation: Ni, W., Ran, H., Wang, R. et al. Stepwise hydrogen spillover–engineered synergistic sites enable near-quantitative conversion of waste PET to p-xylene. Nat Commun 17, 2128 (2026). https://doi.org/10.1038/s41467-026-68990-4
Keywords: plastic upcycling, PET recycling, heterogeneous catalysis, p-xylene production, hydrogen spillover