Clear Sky Science · en
Coacervate droplets as pH-regionalized protocells
Why tiny droplets matter for life’s chemistry
Inside our cells, countless chemical reactions must run at just the right speed and under just the right conditions to keep us alive. One of the most important of these conditions is acidity, often described by pH. Yet some of the busiest “workplaces” in the cell, called membrane-less organelles, have no surrounding membrane or pumps to set their pH. This study explores how simple, droplet-like structures made from short peptides can mimic these organelles, create their own tiny pH zones, and control complex reactions like copying DNA and making proteins—offering clues to both modern cell biology and how the earliest protocells on Earth might have functioned.
Hidden pH zones inside the cell’s control center
The authors begin with the nucleolus, a large droplet-like compartment inside the cell nucleus that helps build ribosomes, the protein factories of the cell. Using a fluorescent dye that changes color with pH, they measured acidity inside the nucleolus and in the surrounding nucleoplasm in several cell types. They found that the nucleolus is consistently slightly more acidic than its surroundings, revealing a built-in pH difference across this invisible boundary. When they treated cells with drugs that disturb nucleolar activity or structure, this pH contrast shrank or disappeared, linking the local acidity not to membrane pumps but to the very existence and health of the droplet-like structure itself.
Building synthetic droplets that split acidity
To study this effect under controlled conditions, the team built an artificial system 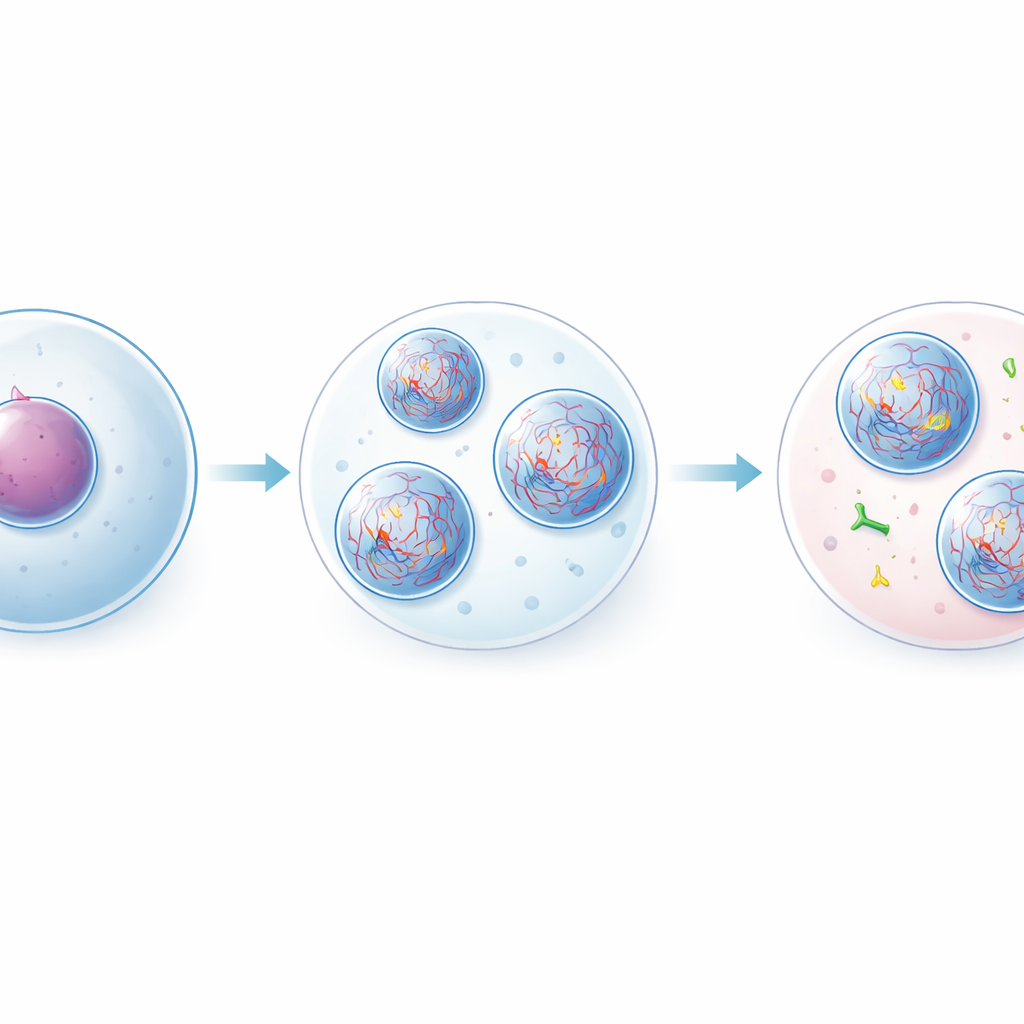
Turning droplets into working mini-reactors
Next, the researchers loaded these peptide droplets with real enzymes that naturally change pH as they work. One enzyme, glucose oxidase, converts sugar to an acid, shifting its environment toward lower pH. Another, urease, breaks down urea to produce basic products that raise pH. The enzymes spontaneously concentrated inside the droplets because of attractive charge-based interactions with the peptide chains. When their substrates were added, the interior of the droplets shifted its pH more strongly than the surrounding solution, and this tuning range could be widened or narrowed by adjusting droplet composition and salt content. Despite the crowded interior, the enzymes remained active, though their speed and apparent affinity for substrates differed from those in plain solution, reflecting the special microenvironment inside each droplet.
Programming reaction cascades with local acidity
With controllable pH zones in hand 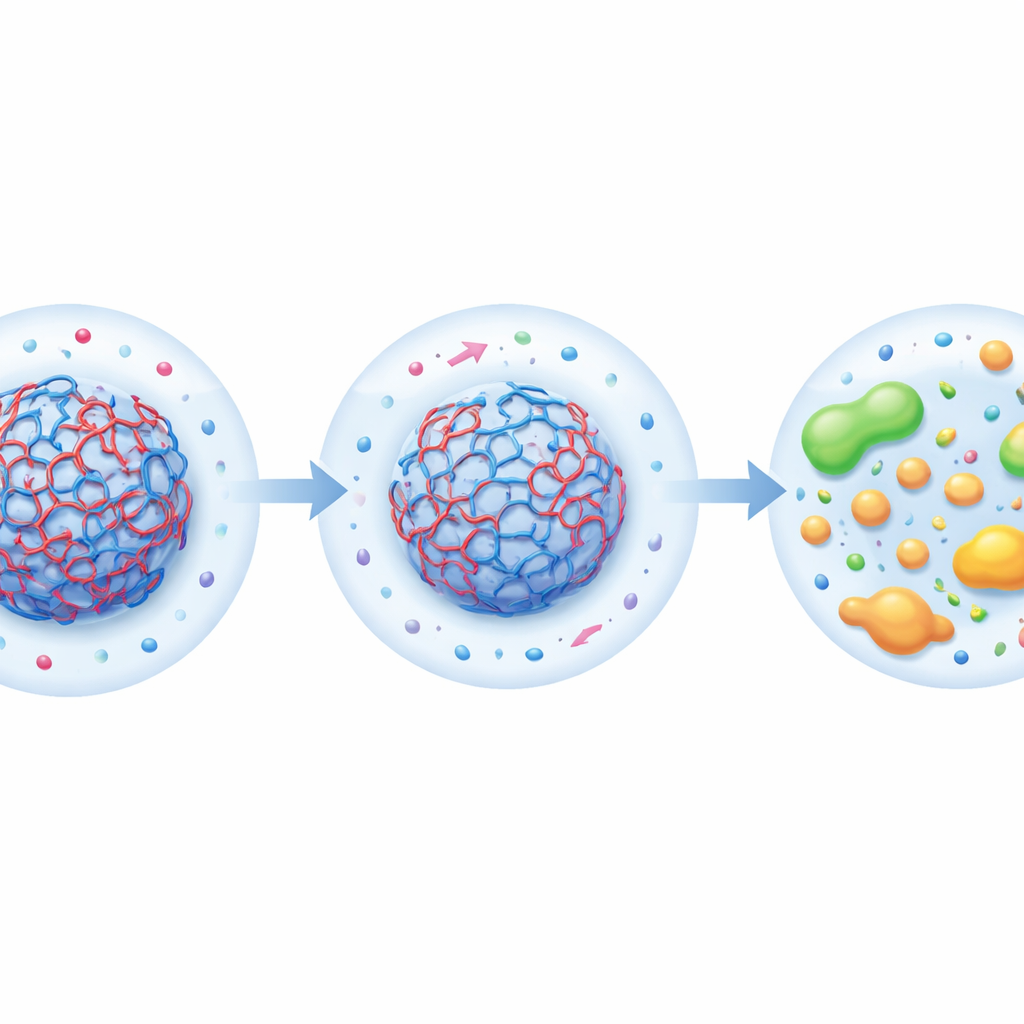
What this means for cells and protocells
Taken together, the work shows that droplets formed by phase separation can naturally create and maintain small but meaningful pH differences—without membranes, pumps, or elaborate machinery. In living cells, similar condensates may use this principle to fine-tune which reactions happen where and when, helping organize metabolism and gene control in space. In the context of early life, such coacervate droplets act as plausible protocells, offering sheltered environments where key reactions like copying genetic material and making simple proteins could be guided by local chemistry alone. By demonstrating precise pH control and complex reaction chains in these minimal systems, the study points toward both a deeper understanding of modern cellular organization and new tools for synthetic biology that harness phase-separated droplets as programmable, pH-tuned microreactors.
Citation: Wang, C., Fang, Z., Zhang, L. et al. Coacervate droplets as pH-regionalized protocells. Nat Commun 17, 2252 (2026). https://doi.org/10.1038/s41467-026-68980-6
Keywords: membraneless organelles, phase separation, coacervate droplets, pH regulation, protocells