Clear Sky Science · en
VSIG10L is a major determinant of esophageal homeostasis and inherited predisposition to Barrett’s esophagus
Why this matters for people with heartburn
Chronic heartburn is common, but only a small fraction of people go on to develop Barrett’s esophagus, a condition that can lead to esophageal cancer. This study asks a simple but important question: why are some families much more prone to Barrett’s esophagus than others? By tracing a single gene and watching how it shapes the lining of the esophagus in humans and mice, the researchers uncover a missing link between inherited risk, reflux damage, and the body’s ability to keep the esophagus healthy.
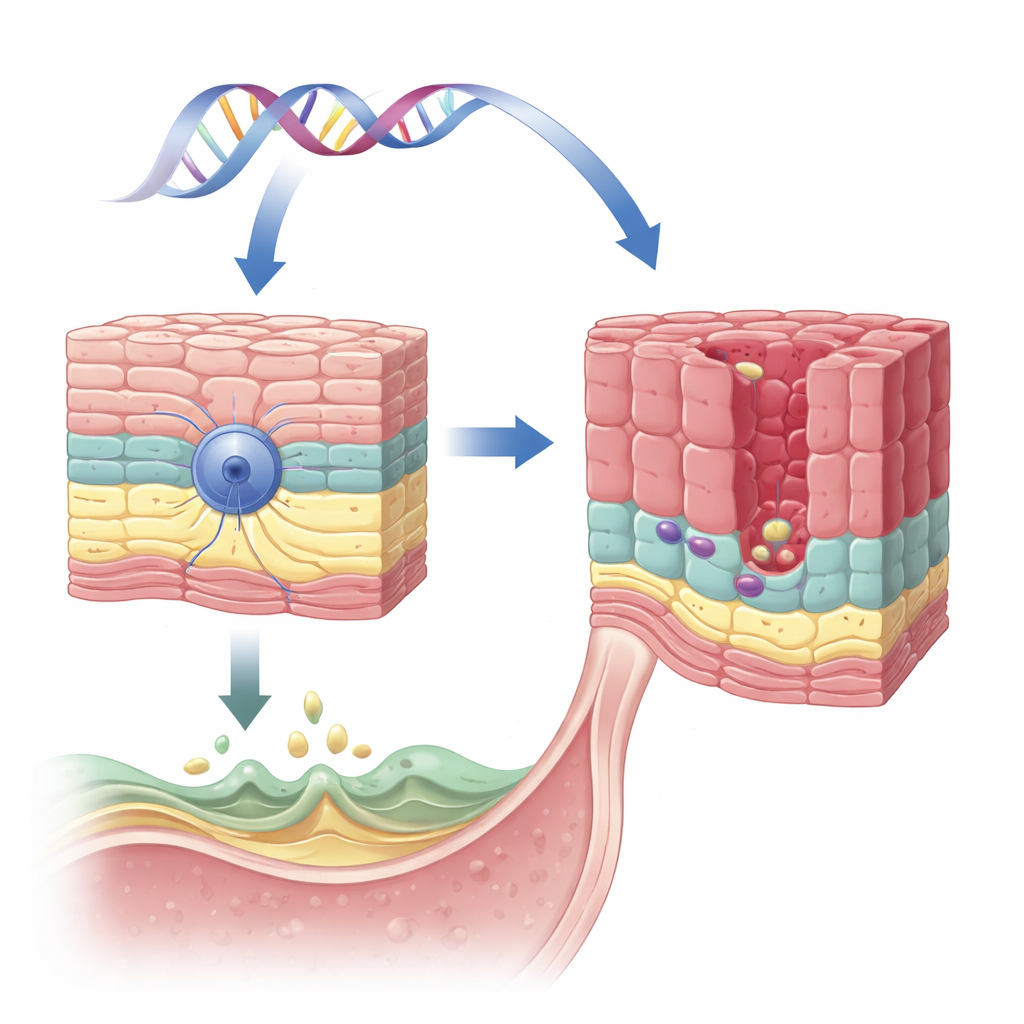
A protective gene in the esophageal lining
The team focused on a gene called VSIG10L, previously flagged in one large family with many members affected by Barrett’s esophagus and related cancers. VSIG10L is active in the upper layers of the flat (squamous) cells that line the esophagus, but not in the deeper, stem-like basal cells. Using a sensitive RNA detection method in human, pig, and mouse tissues as well as three-dimensional cultures of human esophageal cells, the researchers showed that VSIG10L is consistently switched on only in “maturing” cells just above the basal layer. This pattern suggests that VSIG10L helps squamous cells complete their final steps of maturation and form a stable barrier.
Inherited changes that weaken the barrier
By sequencing the VSIG10L gene in 684 people from 302 Barrett’s and esophageal cancer families, the investigators uncovered several rare, damaging variants. To test what these variants do, they engineered human stem-cell–derived esophageal organoids from a patient carrying one such mutation. Compared with organoids from a healthy donor, the mutant organoids failed to build the normal, layered squamous structure. Instead, they often formed abnormal, gland-like balls of cells that remained stuck in a basal, immature state, marked by a protein called p63. This distortion of normal layering and maturation mirrors early steps thought to precede Barrett’s esophagus in patients.
Mouse models that replay human disease
To move beyond cell cultures, the team created mice carrying either a precise copy of the human familial VSIG10L mutation or a complete loss of the gene. In healthy mice, the corresponding Vsig10l gene is again active only in suprabasal squamous cells. Under the electron microscope, mutant mice showed a striking loss of desmosomes—the tiny “rivets” that fasten neighboring cells together—in these layers. Gene activity maps from the squamous forestomach, a mouse region that resembles the lower esophagus, revealed broad disruption of structural and differentiation programs. When the mice were fed a diet containing the bile acid deoxycholate, mimicking chronic reflux, 70–100% of Vsig10l-mutant animals developed extensive Barrett’s-like mucin-rich patches at the squamous–columnar junction, whereas wild-type mice developed only small, limited lesions.
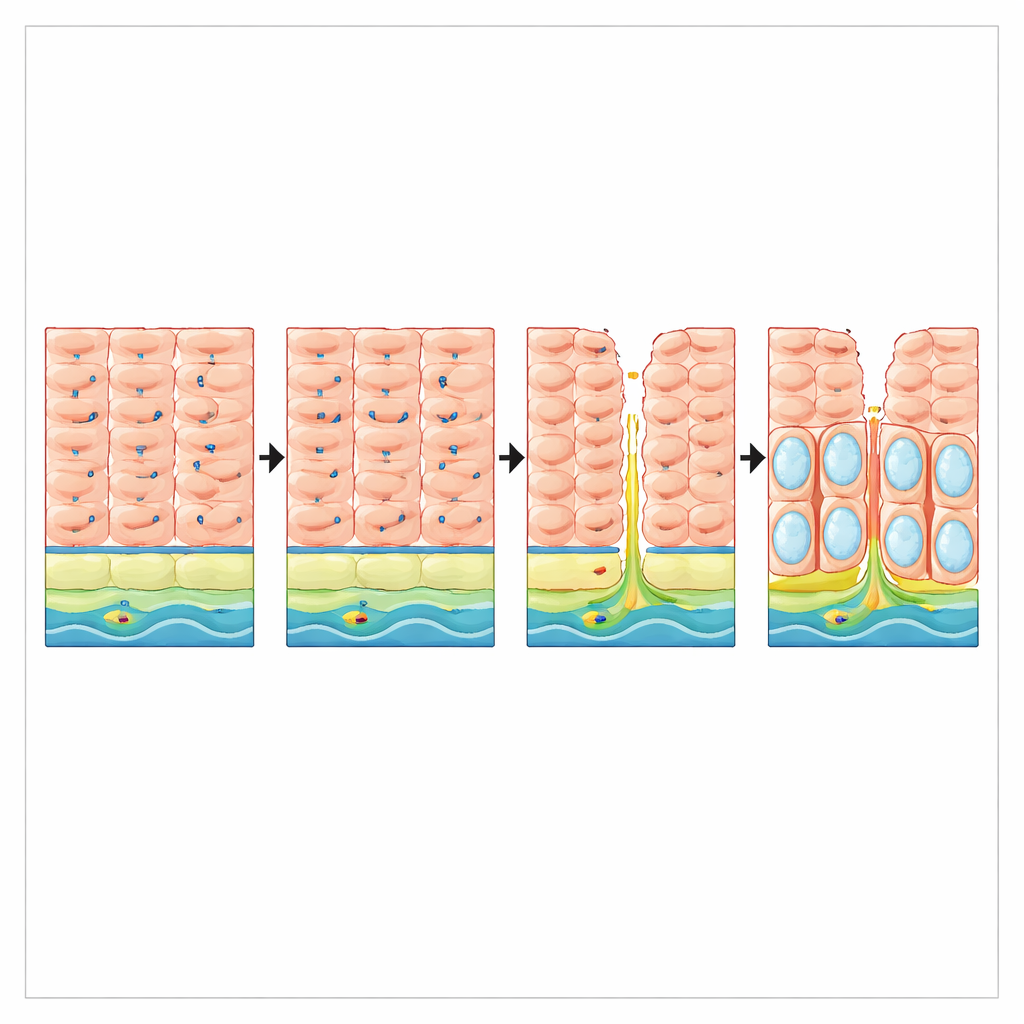
Reflux injury and loss of protection in patients
The researchers then asked whether reflux itself might disturb VSIG10L in humans. In biopsies from patients with long-standing gastroesophageal reflux disease but without Barrett’s esophagus, VSIG10L RNA levels were about three times lower than in people without reflux, even though the basic squamous cell marker TP63 was unchanged. In tissue sections, healthy esophageal lining showed a clear band of VSIG10L-positive cells above a basal layer. In contrast, reflux-damaged mucosa displayed an expansion of basal-like, p63-rich cells and a near-complete loss of the VSIG10L-positive differentiation gradient. These findings suggest that chronic reflux erodes not only the physical integrity of the lining but also the gene program that maintains its orderly renewal.
Putting the pieces together
Taken together, the work supports a simple model: VSIG10L is a key guardian of the esophageal squamous barrier. Inherited defects in this gene, or acquired suppression through chronic reflux, lead to poor maturation of the lining, fewer cell–cell anchors, and a leakier surface. In this vulnerable state, repeated exposure to acid and bile promotes abnormal, gland-like healing instead of restoration of the original squamous lining, setting the stage for Barrett’s esophagus. For patients and families, this research points toward future genetic tests that may flag those at highest risk and toward new therapies aimed at preserving or restoring VSIG10L function, with the long-term goal of preventing Barrett’s esophagus and its progression to cancer.
Citation: Ravillah, D., Singh, S., Katabathula, R.M. et al. VSIG10L is a major determinant of esophageal homeostasis and inherited predisposition to Barrett’s esophagus. Nat Commun 17, 2167 (2026). https://doi.org/10.1038/s41467-026-68975-3
Keywords: Barrett’s esophagus, gastroesophageal reflux, genetic predisposition, epithelial homeostasis, esophageal cancer risk