Clear Sky Science · en
Exploiting human fucosyltransferase 8 allostery with a covalent inhibitor for core fucosylation suppression
Why blocking a tiny sugar can matter for cancer
Our cells constantly decorate proteins with small sugar chains that fine‑tune how those proteins work. One particular sugar mark, called core fucosylation, helps tumors grow, spread, dodge the immune system, and even resist modern cancer drugs. This paper describes how scientists found a hidden control point on the human enzyme that adds this sugar, and built a new, low‑toxicity molecule that latches onto that control point to turn the enzyme off. Their work opens a new path for medicines that subtly rewire cell‑surface sugars instead of attacking DNA or proteins directly.
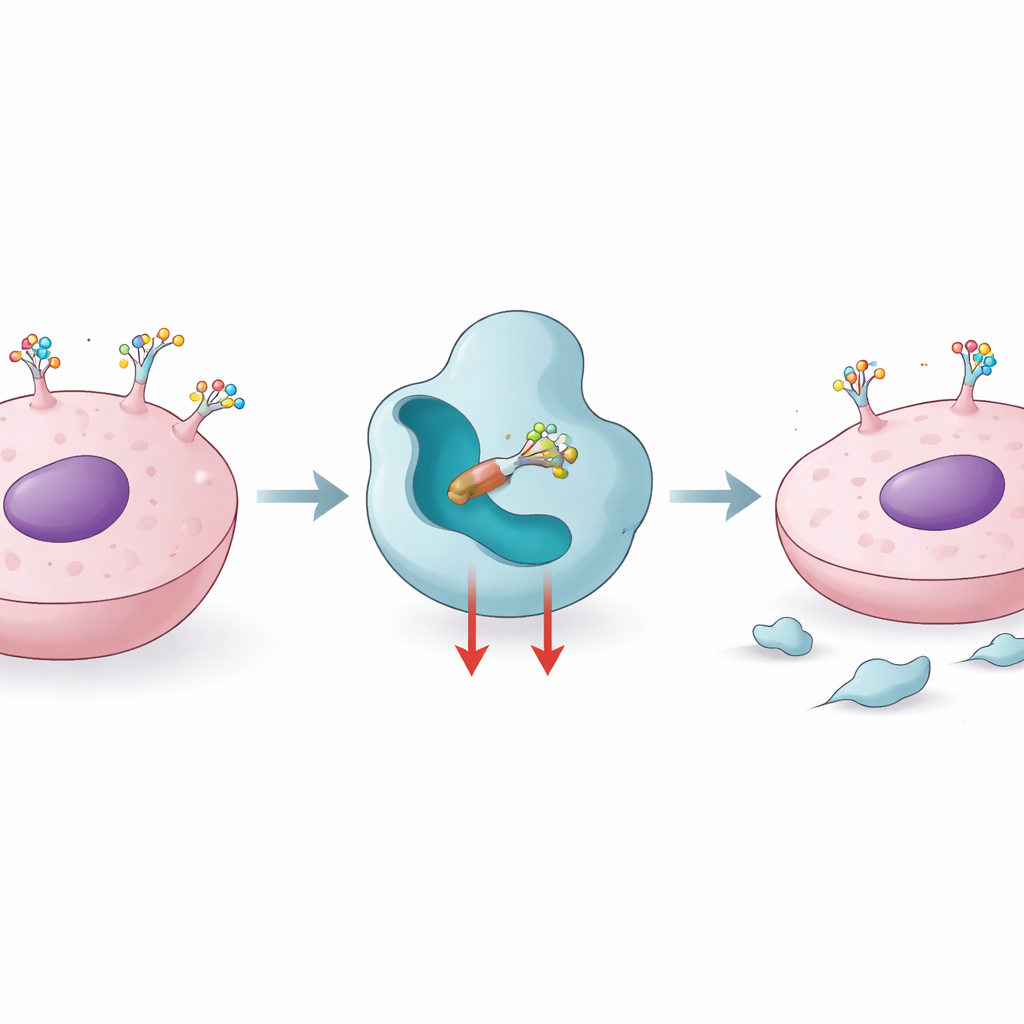
How cells use sugar tags to shape disease
Proteins leaving a cell’s inner factory pass through a molecular “finishing line” where sugar chains are attached and edited. Core fucosylation is one such finishing step, carried out by an enzyme called FUT8, which adds a single fucose sugar to a key position on many protein‑linked glycans. When FUT8 levels rise, this sugar mark becomes more common on molecules that control cell growth, movement, and immune recognition. Studies have linked high FUT8 activity to aggressive melanoma, triple‑negative breast cancer, lung cancer, and resistance to targeted drugs and some antibody therapies. Blocking FUT8 can strip certain immune brake proteins, like PD‑1, from the cell surface and make tumors more visible to the immune system, making FUT8 an attractive target for cancer immunotherapy and for improving biologic drugs.
The need for smarter, more selective blockers
Designing drugs against FUT8 has been hard because many related enzymes all use the same basic sugar fuel. Earlier inhibitors often mimicked that fuel, leading to broad, non‑selective shutdown of many fucosylation pathways and unwanted side effects, including clotting problems that halted a clinical trial. The authors reasoned that a better strategy would be to avoid the crowded main active site and instead hunt for a unique “remote” site on FUT8 that could act as a control switch. Using a high‑throughput screening test that monitored the enzyme’s activity, they sifted through over six thousand compounds and flagged a few chain‑shaped molecules that slowed FUT8 while also reducing cancer cell invasion in lab dishes.
Discovering a hidden control pocket
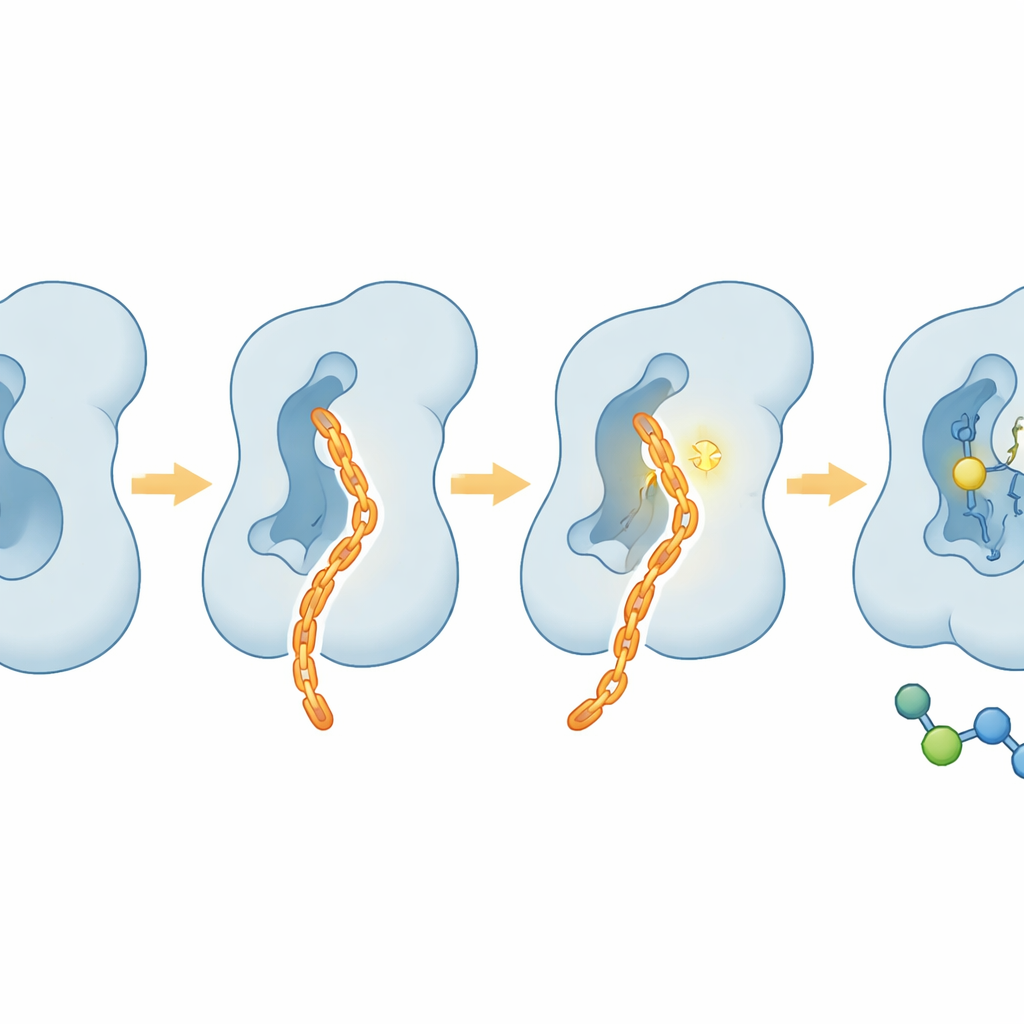
Using X‑ray crystallography, the team solved the three‑dimensional structures of FUT8 bound to two of these inhibitors. To their surprise, neither compound sat in the usual spot where the sugar donor and acceptor bind. Instead, both nestled into a long, channel‑like pocket between two major lobes of the enzyme, close to but distinct from the active center. Binding at this site nudged certain loops and key amino acids out of position, particularly elements that normally help clasp the sugar donor. Computer simulations showed that when the pocket was filled, FUT8’s overall motion and energy landscape shifted, favoring shapes that are poor at catalyzing the reaction. In other words, the inhibitors worked allosterically: they pressed a distant button that caused the working parts of the enzyme to misalign.
Turning a hit molecule into a precise covalent tool
Armed with this structural map, the researchers searched for molecules that could not only occupy the pocket but also form a permanent bond to a nearby lysine residue (K216) that naturally swings toward the channel. They found that a reagent called SSO, which carries a reactive N‑hydroxysuccinimide ester, bound in the same groove and formed a covalent link to K216, locking the enzyme in an inactive state. Building on the shared features of SSO and their earlier hit NH125, they merged fragments of both to create a new molecule named CAIF. Crystal structures confirmed that CAIF threads along the channel, fits snugly among hydrophobic residues, and makes a covalent bond to K216. This design boosted FUT8 inhibition several‑fold compared with its parent compounds while sparing closely related enzymes, highlighting the unique shape of FUT8’s allosteric pocket.
Soft on cells, tough on tumor behavior
Beyond test tubes, the team examined how CAIF behaves in living cells. Across several human cell lines, CAIF showed minimal toxicity even at high doses, indicating it does not broadly poison cell machinery. Yet it markedly reduced core fucosylation on the cell surface, as revealed by sugar‑binding dyes, and did so with far better selectivity for FUT8 than earlier pan‑inhibitors. In cancer cell invasion assays, CAIF outperformed a commonly used sugar‑based blocker in limiting the ability of tumor cells to migrate through a matrix, a hallmark of metastatic potential. These effects align with FUT8’s known role in enhancing invasion and immune evasion rather than directly driving cell division.
What this means for future cancer treatments
This study does not claim that CAIF itself is ready to be a drug. Instead, it demonstrates that FUT8 contains a previously unknown, druggable allosteric pocket that can be targeted with carefully shaped molecules that form a durable bond to a single lysine. By exploiting this site, researchers can selectively dial down a sugar‑modifying enzyme that lies at the crossroads of cancer progression, immune escape, and drug resistance, while leaving related enzymes largely untouched. The work offers a blueprint for designing next‑generation inhibitors that reshape disease‑relevant sugar patterns on proteins and may ultimately broaden the toolkit for cancer immunotherapy and improved antibody medicines.
Citation: Jiang, J., He, D., Ke, M. et al. Exploiting human fucosyltransferase 8 allostery with a covalent inhibitor for core fucosylation suppression. Nat Commun 17, 2607 (2026). https://doi.org/10.1038/s41467-026-68971-7
Keywords: FUT8 inhibition, core fucosylation, covalent allosteric inhibitors, cancer glycosylation, glycosyltransferase drug design