Clear Sky Science · en
Photoinduced polarity-mismatched transformations of isoquinolines into naphthalenes
Lighting Up New Paths for Medicines
Many modern medicines are built from flat, ring-shaped carbon frameworks. Swapping one kind of ring for another can dramatically change how a drug behaves in the body, but doing so usually demands long, expensive syntheses. This study introduces a light-driven shortcut: a way to directly reshape a common nitrogen-containing ring, called an isoquinoline, into a closely related carbon-only ring, a naphthalene. The method works under mild conditions, tolerates many functional groups, and can be applied late in the life of a drug molecule, opening faster routes to improved treatments.
Why Swapping Ring Cores Matters
In drug discovery, chemists build and test vast libraries of related molecules to tune properties such as potency, selectivity, and stability. Even changing a single atom in the central ring can sharply alter how a compound fits into its biological target or how long it survives in the body. Isoquinolines and naphthalenes are classic examples of such interchangeable “look‑alike” cores: they have similar size and shape, but one carries a nitrogen atom while the other is made only of carbon. Traditionally, moving from one core to the other requires rebuilding the molecule from scratch, step by laborious step. A direct one‑step conversion between the two would let chemists recycle existing molecules into new variants instead of starting over.
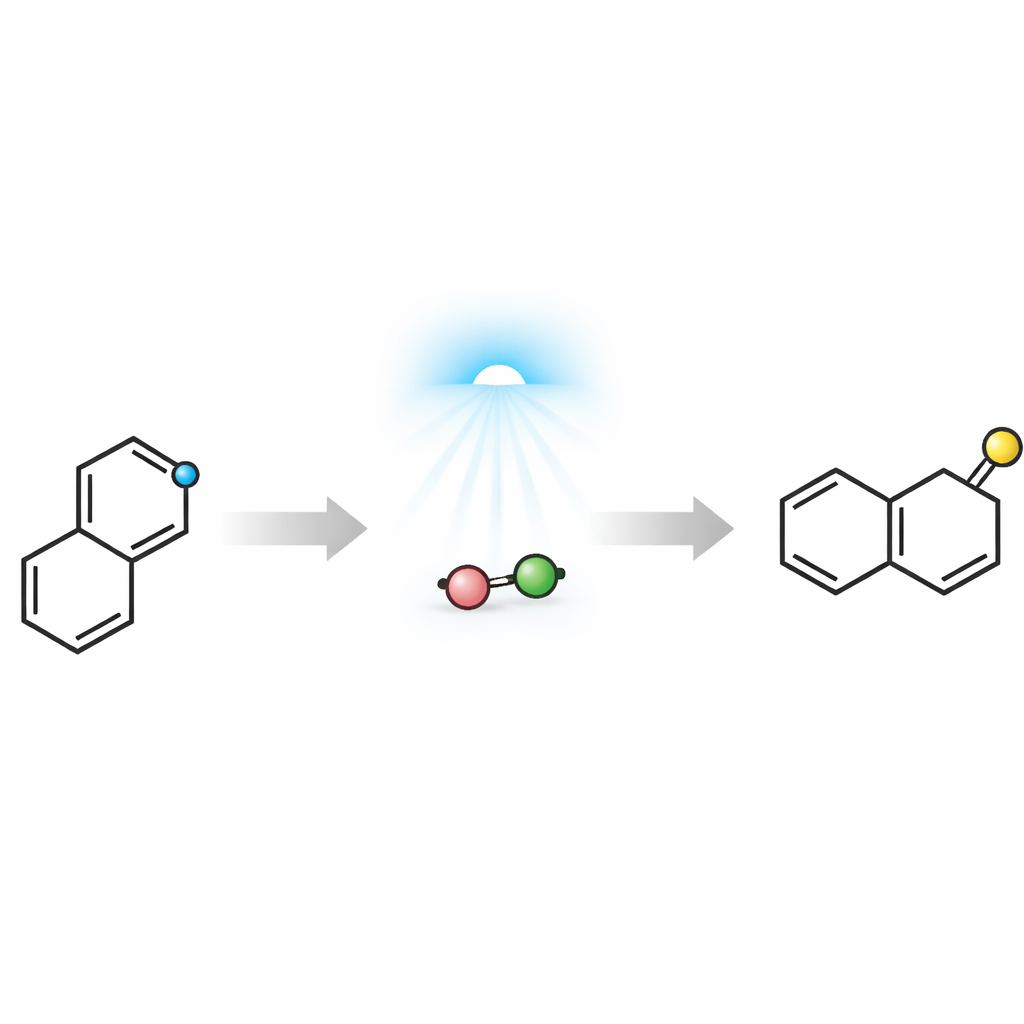
Turning Electronic Mismatch into an Advantage
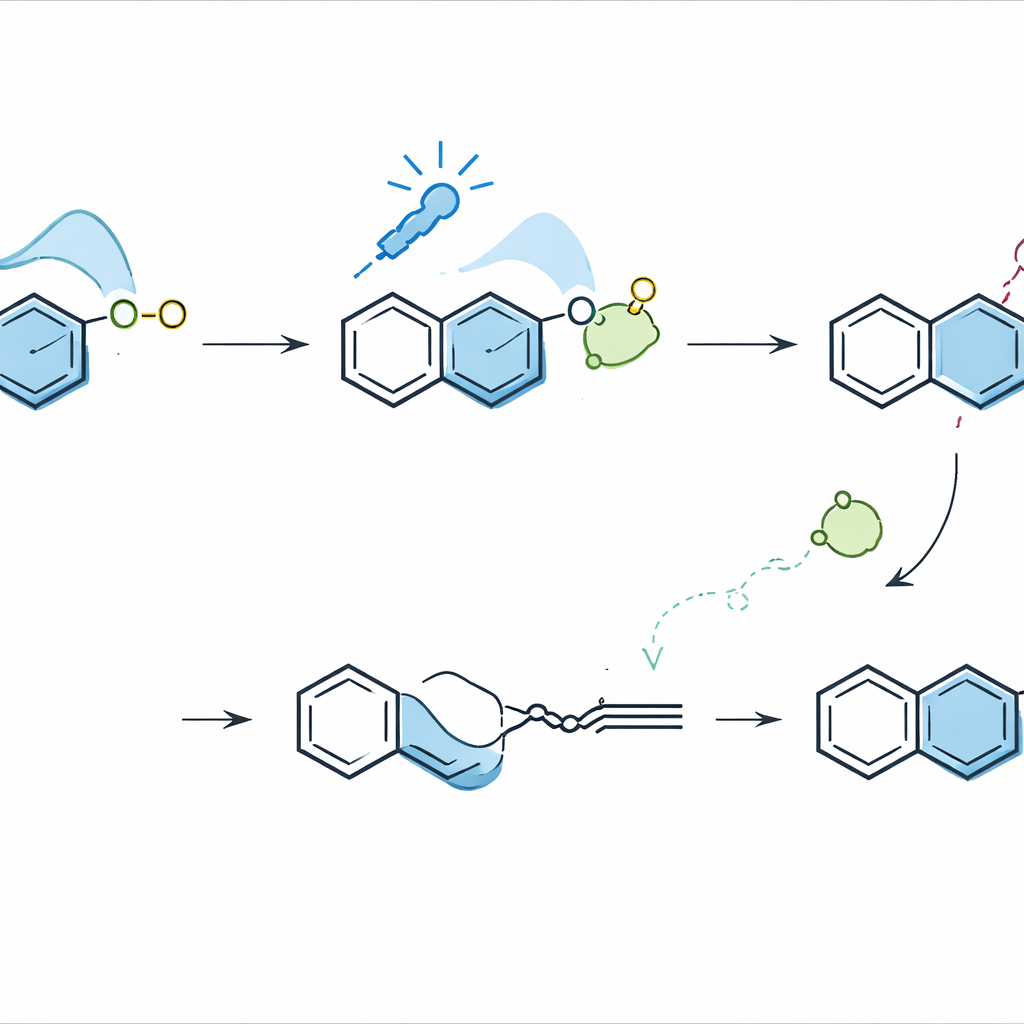
The challenge is that isoquinolines and the alkynes used to build naphthalenes are both relatively electron‑poor, so they normally repel each other in the key bond‑forming step, a type of six‑atom ring‑building reaction. The authors overcame this “polarity mismatch” by enlisting visible light and a simple inorganic base. When an isoquinoline is turned into a salt and paired with carbonate, the two form a loose association that can absorb blue light. Computational studies guided the design and showed that light excitation shuttles an electron from the carbonate to the isoquinolinium ring, temporarily converting it into an electron‑rich radical. In this activated state, the isoquinoline can now add stepwise to an electron‑poor alkyne, forming a new ring system that ultimately rearranges into a naphthalene while ejecting a nitrogen‑containing fragment.
Probing the Reaction in the Lab
Experimentally, the transformation is simple to run: isoquinolinium salts, alkynes, and sodium carbonate are stirred in ethanol under blue LEDs with no additional photocatalyst. The team optimized the conditions, showing that carbonate is crucial not only as a base but also as an electron donor, and that bromide ions help promote the process. Radical traps shut the reaction down and captured intermediate adducts, supporting the radical pathway proposed by the calculations. Spectroscopic experiments confirmed that the isoquinolinium–carbonate pair is the true light‑absorbing complex, and switching the counter‑ions or bases altered yields in ways consistent with their ability to participate in electron transfer.
From Model Systems to Drug‑Like Molecules
Once the reaction conditions were tuned, the authors demonstrated a broad scope. Many differently substituted alkynes participated, including those bearing sensitive groups such as iodine, extra double bonds, and fragments from natural products or existing drugs. A wide range of isoquinolinium salts also worked, even crowded or heterocycle‑containing ones, giving access to multi‑substituted naphthalenes that are hard to prepare by traditional routes. The method could even reshape complex bioactive molecules like the PRMT3 inhibitor SGC707 and the vasospasm drug fasudil into their naphthalene analogues in a single late‑stage operation. The new products carry ester groups that act as both potential protein‑binding handles and versatile synthetic entry points.
Building Larger Architectures from the New Rings
The freshly made naphthalene esters are not just end points; they serve as building blocks for more elaborate structures. The team showcased a series of follow‑up reactions that convert these esters into high‑value polycyclic aromatic hydrocarbons and chiral ligands, such as benzofluorenones, carbazoles, BINOLs, and QUINOLs—scaffolds widely used in materials science and asymmetric catalysis. In another demonstration, they used the method as a key step in preparing an analogue of adapalene, a dermatology drug, starting from a readily assembled isoquinoline precursor.
What This Means Going Forward
By using visible light to flip the electronic character of a nitrogen‑containing ring, this work turns a previously disfavored reaction into a powerful skeletal editing tool. Chemists can now convert abundant isoquinolines directly into richly functionalized naphthalenes, sidestepping long synthetic routes and preserving delicate side chains. For non‑specialists, the takeaway is that “recycling” existing molecules into new cores is becoming more practical, which could speed up the search for better drugs and advanced materials while reducing cost and waste.
Citation: Zhang, C., Zhang, J., Lan, Y. et al. Photoinduced polarity-mismatched transformations of isoquinolines into naphthalenes. Nat Commun 17, 2547 (2026). https://doi.org/10.1038/s41467-026-68969-1
Keywords: skeletal editing, photochemistry, isoquinoline, naphthalene, drug discovery