Clear Sky Science · en
Association-induced folding governs surrogate light chain and pre-B cell receptor core assembly
How Our Bodies Test-Drive New Antibodies
Before a young B cell in your bone marrow is allowed to join the immune system, it has to pass a tough test: can it build a working antibody core? This paper explores how a special “practice” version of part of the antibody, called the surrogate light chain, helps carry out that test. By watching how these proteins fold and fit together, the authors reveal a hidden quality-control system that decides which future antibodies are good enough to keep—and which are discarded.
A Safety Checkpoint for Young Immune Cells
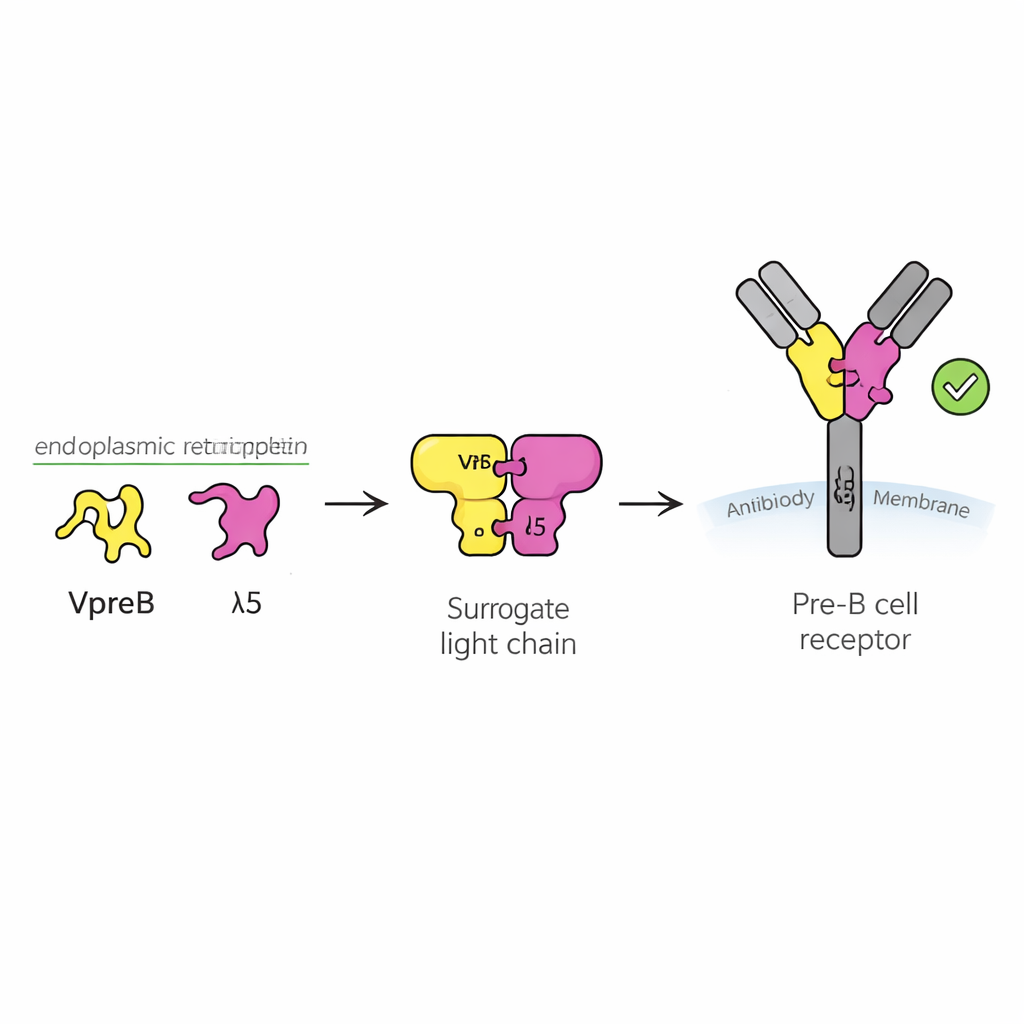
Antibodies are Y‑shaped proteins that recognize invading germs. They are built from two main parts: heavy chains and light chains. During B cell development, the heavy chain is made first, and the cell must figure out if this new heavy chain is useful before investing energy in making a matching light chain. To do this, the cell uses a temporary stand‑in called the surrogate light chain, made of two proteins named VpreB and λ5. Together with the heavy chain, they form the pre‑B cell receptor, a sensor on the cell surface that sends a “go” or “stop” signal. Around half of newly rearranged heavy chains fail this test, so understanding how the surrogate light chain works is key to understanding how a healthy antibody repertoire is built.
Folding by Association: Helping an Unstable Partner
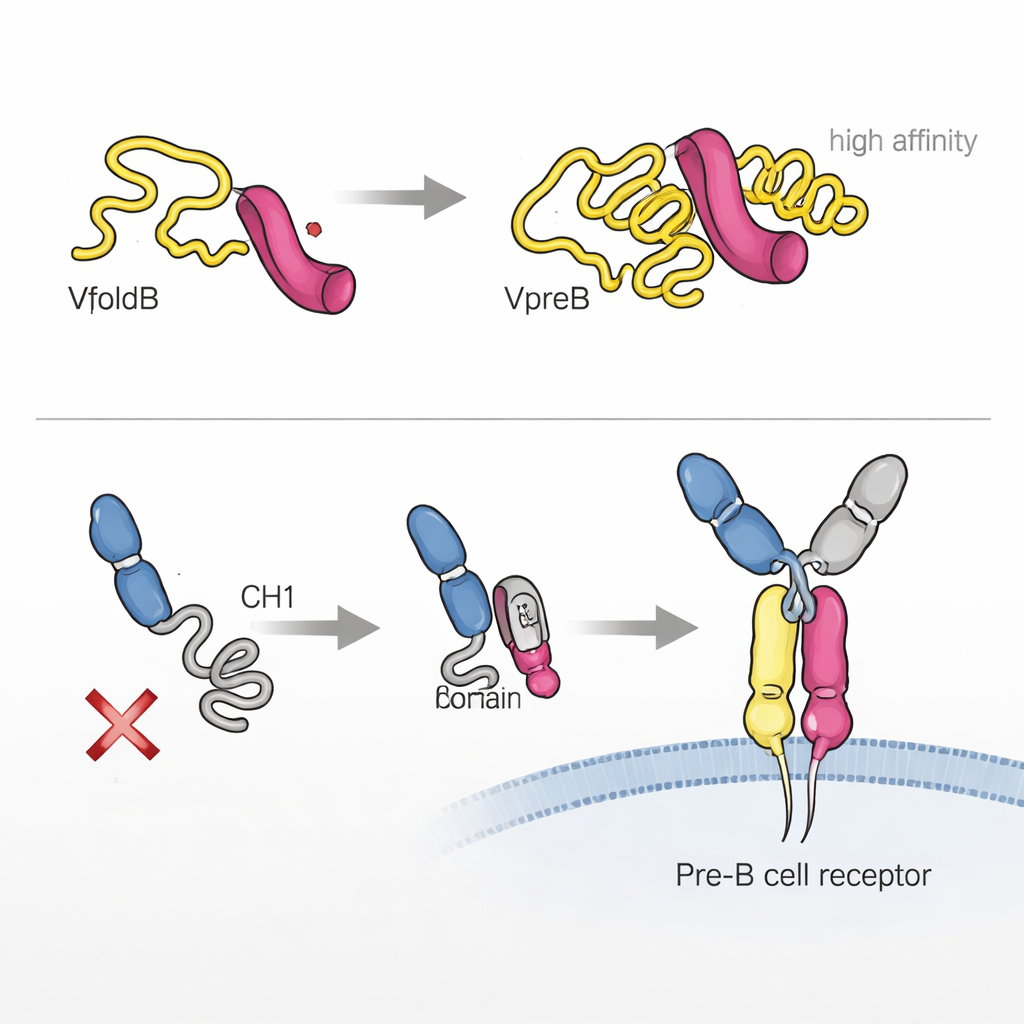
The authors found that one component, VpreB, is largely unfolded and unstable when it is on its own. Using biophysical techniques, they showed that it only snaps into its proper three‑dimensional shape when it binds to λ5. A short stretch of λ5, shaped like a beta‑strand, slots into VpreB and completes its structure, like inserting a missing tooth into a gear. This “association‑induced folding” not only stabilizes VpreB, it also creates a very tight partnership between the two proteins, with an affinity in the low nanomolar range. In cells, only when VpreB and λ5 assemble into this surrogate light chain do they escape retention inside the endoplasmic reticulum and become secreted or moved onward, highlighting how folding and quality control are tightly coupled.
Finishing the Heavy Chain and Passing Inspection
The story does not end with VpreB and λ5. A crucial region of the heavy chain, called CH1, is initially unstructured and kept inside the cell. The researchers showed that λ5, acting somewhat like a chaperone, can trigger CH1 to fold when they bind. This second association‑induced folding step is essential for building the full pre‑B cell receptor and allowing it to leave the endoplasmic reticulum and reach the cell surface. Interestingly, both parts of the surrogate light chain—VpreB and λ5—can touch the heavy chain, but only λ5 is able to drive CH1 into its finished, fold‑competent state. When this does not happen, the partial receptor is held back and the cell effectively rejects that heavy chain.
Flexible Tails that Fine‑Tune Binding and Signaling
Both VpreB and λ5 carry floppy, unstructured segments known as unique regions, which do not resemble any known protein sequences. Although these tails are not strictly required for the two proteins to assemble in a test tube, the team found that they strongly influence how fast the surrogate light chain forms, how stable it is, and how well it binds to heavy‑chain segments. In cells, removing these regions reduced how efficiently the assembled complexes were secreted. The λ5 tail in particular emerged as a multi‑tasker: it helps speed up assembly, is essential for certain contacts with the heavy‑chain variable region, and makes major contributions to how the pre‑B cell receptor engages antigens and other ligands. In some cases, the antigen‑binding strength of a surrogate light chain–heavy chain combination approached that of a normal antibody fragment.
Why This Matters for Antibody Diversity
By mapping these stepwise folding and assembly events, the authors propose that the surrogate light chain does more than simply hold the heavy chain in place. It acts as a dynamic inspector, using its structured core and flexible tails to sense whether a heavy chain can fold correctly and interact productively with potential targets. Successful combinations are folded, stabilized, and allowed to reach the cell surface, where they send signals that let the B cell continue maturing. Faulty combinations are trapped and eventually eliminated. For a layperson, the takeaway is that your immune system runs a sophisticated “test bench” for future antibodies, built from proteins that literally fold each other into shape, ensuring that only well‑behaved antibody cores move forward to defend you.
Citation: König, J., Sarmiento Alam, N.C., He, R. et al. Association-induced folding governs surrogate light chain and pre-B cell receptor core assembly. Nat Commun 17, 1202 (2026). https://doi.org/10.1038/s41467-026-68965-5
Keywords: B cell development, antibody folding, surrogate light chain, protein quality control, pre-B cell receptor