Clear Sky Science · en
Unlocking enzymatic deracemization of structurally diverse allenes by designed photoenzymes
Why mixing and matching molecules matters
Many medicines work only when their atoms are arranged in a very specific three‑dimensional way, much like how a left hand fits a left glove but not a right one. Chemists can often make these “one‑handed” molecules, but it is hard to do so cleanly when the building blocks are complex. This paper tackles that challenge for a tricky class of twisted structures called allenes by using custom‑built light‑driven enzymes. The work shows how biology and photochemistry can be combined to sift out the desired molecular “hand” from a scrambled mixture, promising faster and cleaner routes to future drugs and materials.
Twisted building blocks with a big payoff
Allenes are linear chains of three carbon atoms with two adjacent double bonds, but this simple arrangement twists space in a way that gives the overall molecule a handedness. These axial twists show up in natural products, crop‑protection agents, and advanced catalysts. Unfortunately, traditional methods for making chiral allenes tend to be highly specialized: a catalyst that works beautifully for one allene often fails for another with small structural changes. Even recently developed light‑driven chemical catalysts struggle with this problem. As a result, chemists must repeatedly redesign catalysts from scratch when they want to explore new allenes, slowing discovery and increasing waste.
Borrowing nature’s selectivity and adding light
Enzymes in living systems excel at recognizing and transforming specific shapes with remarkable precision, but they evolved for natural targets, not man‑made allenes. The authors set out to create a “photoenzyme” that combines enzyme‑like selectivity with the ability to harvest light as an energy source. They started from a protein scaffold called CTB10 and genetically rewired it to carry a non‑natural amino acid that behaves like a tiny built‑in light antenna. When this special building block absorbs ultraviolet light, it can pass energy to a nearby allene, temporarily exciting it and allowing its twist to be scrambled. By repeating this process many times, the enzyme can selectively remove one hand from a 50:50 mixture and leave behind mostly the other. 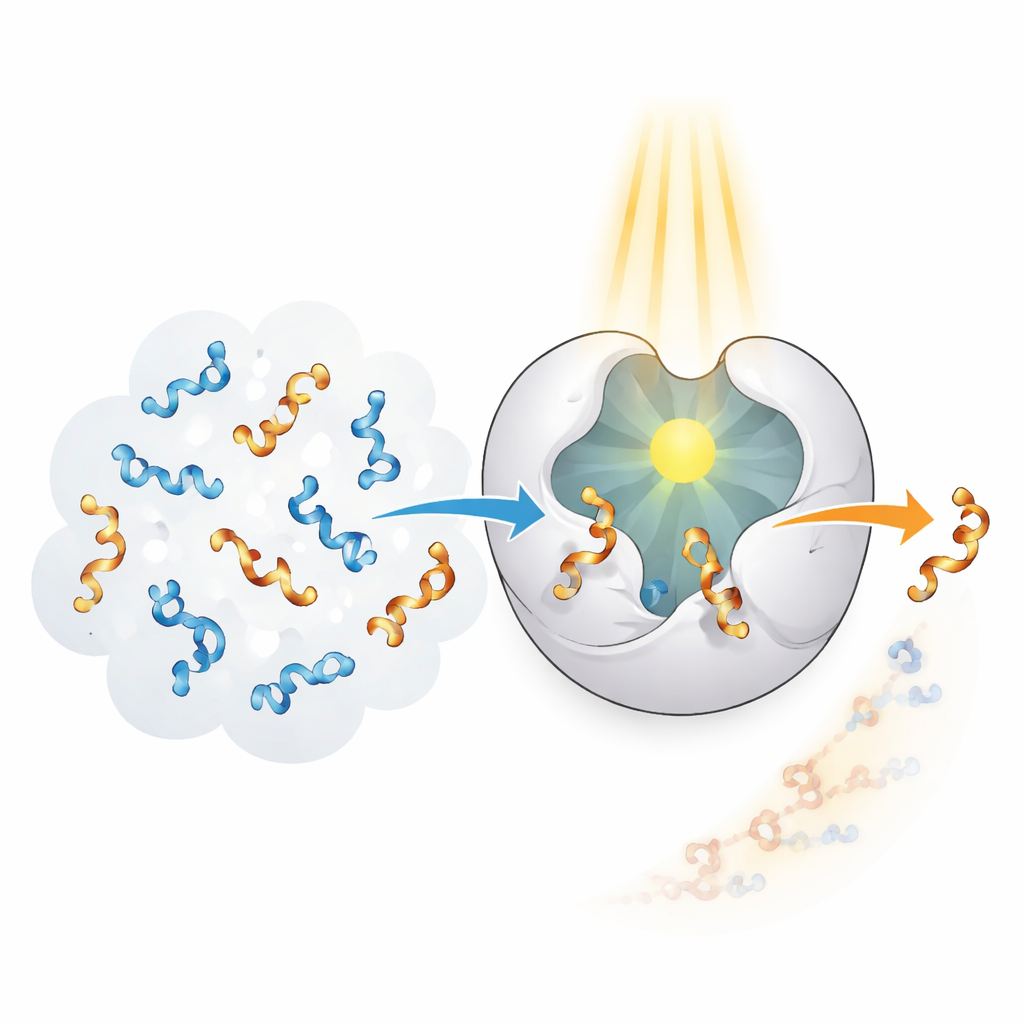
Carving snug pockets for many different guests
Designing such a system is not as simple as shining light on a protein. The team used computer modeling, strategic mutations, and X‑ray snapshots to sculpt several versions of the photoenzyme, each with a slightly different internal pocket. These pockets hold the allene in a defined pose next to the light antenna and stabilize key parts of the molecule through hydrogen bonds and subtle packing interactions. Step by step, the researchers improved both the binding strength and the alignment between enzyme and allene, which are crucial for efficient energy transfer. The final variants showed impressive performance, converting allene carboxylic acids, esters, and amides into single‑handed products with very high purity, often above 99% of one handedness, and doing so under air without the need for delicate conditions.
Watching the mechanism at atomic resolution
To understand why the new photoenzymes prefer one hand over the other, the authors crystallized enzyme–substrate pairs and examined them at atomic resolution. They found that the favored allene hand sits closer to the light‑harvesting group and can adopt several slightly different shapes, all compatible with efficient energy transfer. The disfavored hand either binds weakly or sits farther away, making it much less likely to be excited. Additional experiments with pure right‑ and left‑handed starting materials confirmed this bias: the enzyme rapidly converts the preferred hand into a reactive intermediate, which then relaxes in solution back into a mixture where the opposite hand accumulates. In essence, the protein behaves like a molecular turnstile, repeatedly exciting and consuming one hand while allowing the other to pile up. 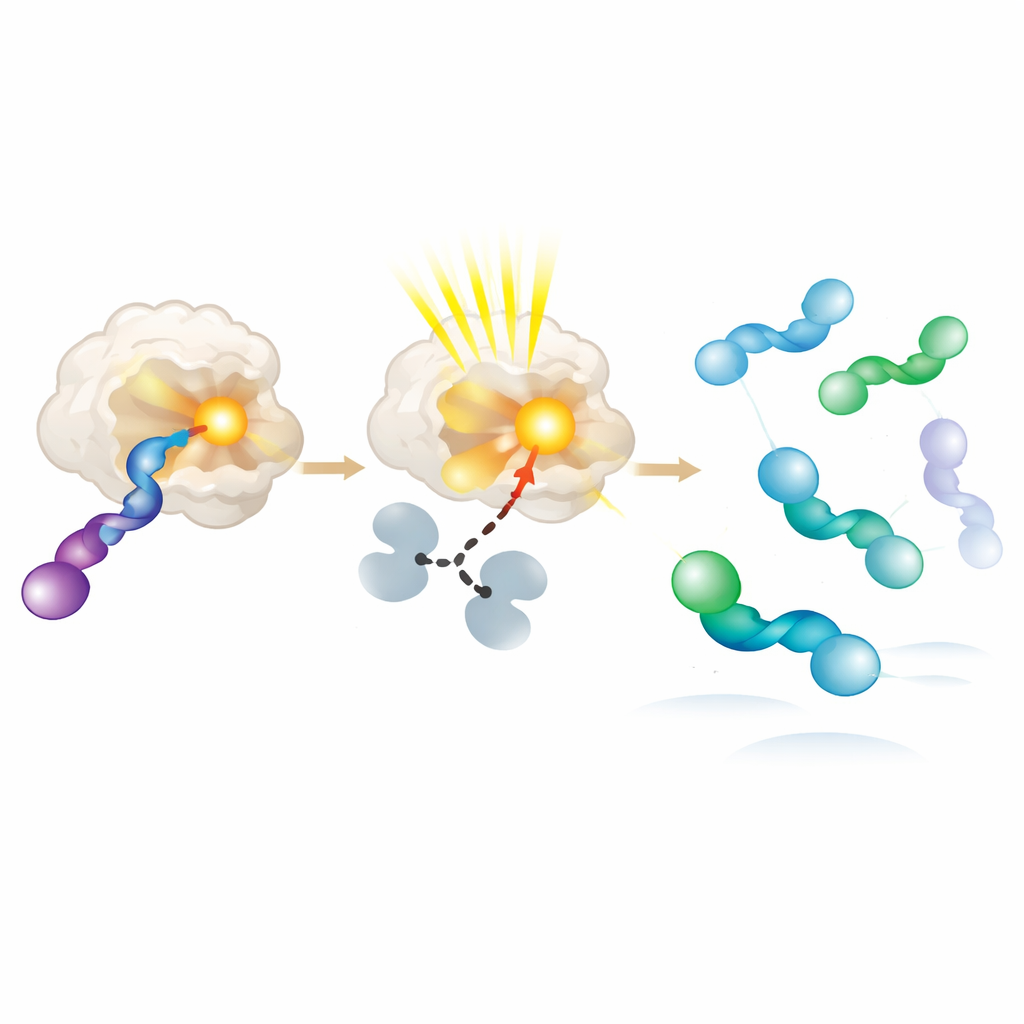
What this means for future chemistry
For non‑specialists, the main message is that the team has created a programmable protein machine that uses light to sort and upgrade twisted molecules with unprecedented versatility. Instead of designing a brand‑new chemical catalyst for each target, researchers can now start from this photoenzyme platform and tune its internal pocket to accept many different allenes. This approach narrows the gap between nature’s exquisite control and the chemical industry’s need for broad, robust methods. In the long term, such light‑powered enzymes could help chemists prepare complex, single‑handed building blocks for medicines and materials more efficiently, with fewer steps and less waste.
Citation: Fu, K., Li, M., Deng, Z. et al. Unlocking enzymatic deracemization of structurally diverse allenes by designed photoenzymes. Nat Commun 17, 2082 (2026). https://doi.org/10.1038/s41467-026-68964-6
Keywords: photoenzyme, deracemization, chiral allene, biocatalysis, triplet energy transfer