Clear Sky Science · en
Synthesis of poly(ester disulfide)s from S8-involved step-growth addition polymerization at ambient temperature
Turning Waste Sulfur into Smart Plastics
Modern life depends on plastics, but most are made from expensive ingredients and are hard to recycle or safely break down. This study shows how an industrial waste—yellow elemental sulfur left over from oil refining—can be transformed at room temperature into a new family of smart plastics. These materials are not only strong and stretchy but can also be broken apart on demand, offering possibilities for greener packaging, medical devices, and environmental cleanup tools.
From Oil Refinery Leftovers to Useful Materials
Each year, refineries produce some 80 million tons of elemental sulfur that often sits unused in massive stockpiles. Chemists have long known sulfur can form polymers, the long chains that make up plastics, but getting it to behave has required high heat, harsh conditions, or complicated starting materials. Earlier methods produced brittle, glassy solids or relied on tricky ring-shaped sulfur compounds that are hard to make in bulk. The challenge has been to find a simple, gentle way to turn this abundant, cheap element into tunable materials with real-world performance.
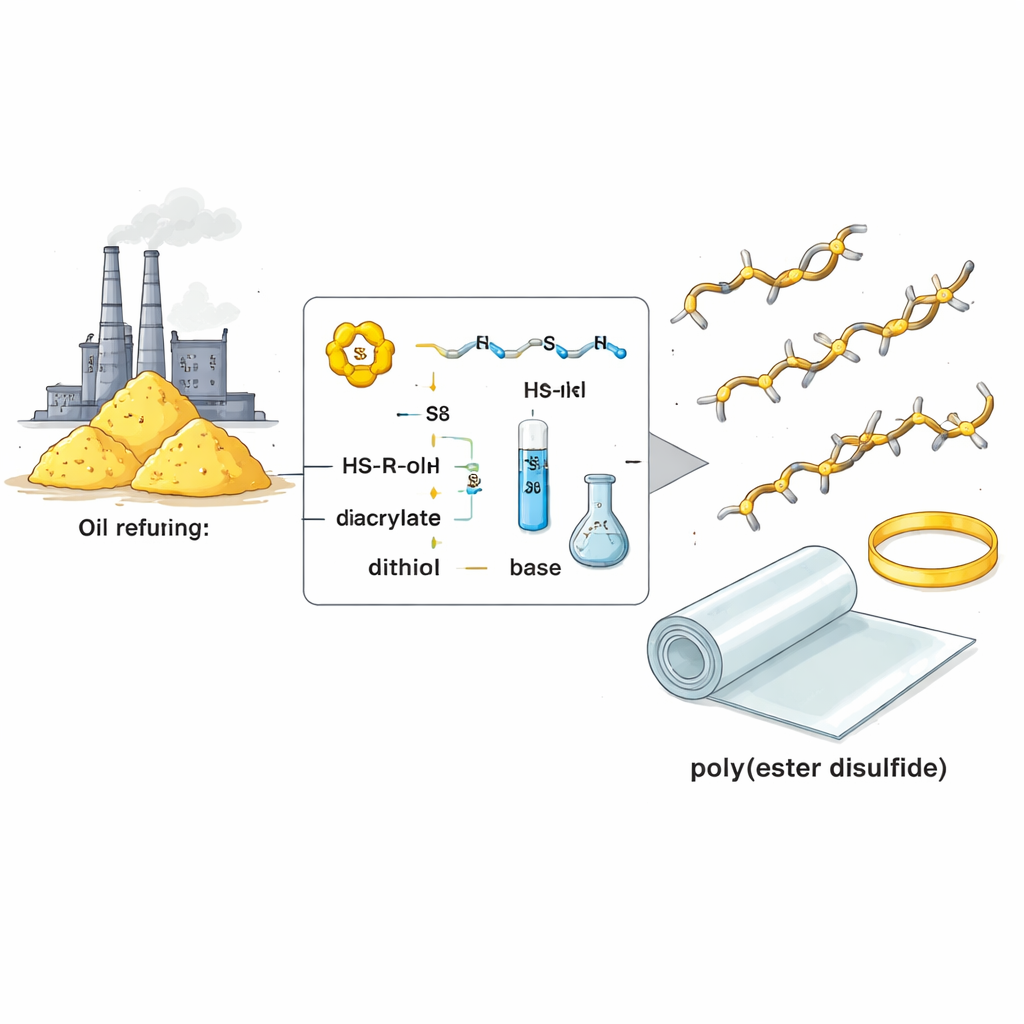
A Gentle Three-Part Chemical Assembly
The researchers devised a room-temperature recipe that combines three widely available ingredients: elemental sulfur, small molecules with two sulfur–hydrogen groups (dithiols), and small molecules with two carbon–carbon double bonds next to ester groups (diacrylates). With a tiny amount of an organic base as a catalyst, sulfur rings open and link up with the dithiols, then add cleanly to the diacrylates. The result is a polymer chain made of alternating ester groups and sulfur–sulfur links. Careful analysis showed that this reaction is highly selective: it avoids common side products and converts the starting materials into the desired building blocks with greater than 95 percent yield.
How the Reaction Chooses Its Path
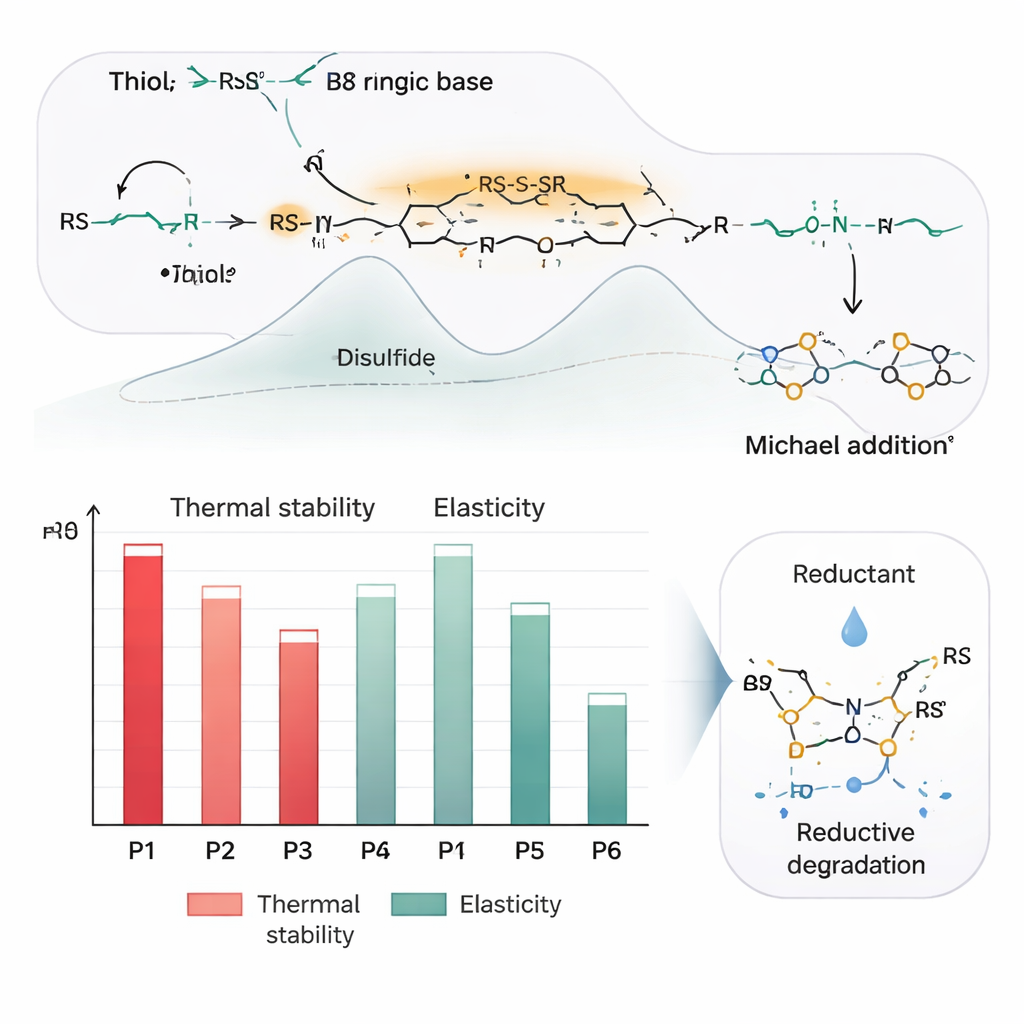
To understand why the chemistry works so reliably at room temperature, the team used quantum-chemical calculations to map out the reaction steps. The base first strips a proton from the dithiol, creating a reactive sulfur species that attacks the sulfur ring (S8) and opens it. This short sulfur chain then snaps down onto the double bond of the acrylate in a so-called Michael addition. A rapid exchange between sulfur–sulfur bonds shuffles the atoms into three closely related products that all have almost the same energy, explaining why they appear in nearly equal amounts. Experiments using nuclear magnetic resonance, mass spectrometry, Raman spectroscopy, and X-ray photoelectron spectroscopy confirm that the final polymers contain the designed mix of ester and disulfide links without leftover elemental sulfur or longer polysulfide chains.
Plastics with Built-In Flexibility and a Self-Destruct Button
By choosing different dithiols and diacrylates, the scientists could dial in a range of physical behaviors. Some combinations produced soft, rubbery materials with very low glass transition temperatures—so flexible that one sample could stretch to more than twenty times its original length and snap back like a rubber band. Others, containing rigid ring-shaped units or extra hydrogen-bonding groups, became tougher and more crystalline, with melting points above the boiling point of water and higher breaking strength. All of these polymers withstand temperatures above roughly 250 °C before beginning to decompose, making them robust enough for demanding uses. Yet their sulfur–sulfur bonds remain sensitive to mild reducing agents, such as dithiothreitol, which can cut the chains back into small molecules within hours. This switchable degradability points to a circular life cycle, in which products can be disassembled for recycling or safe disposal.
What This Means for Future Plastics
In simple terms, this work turns a problem waste stream into a toolbox for next-generation plastics. Using only room temperature, common solvents, and small amounts of organic catalysts, the authors created sulfur-rich polymers with tunable strength, stretchiness, and the ability to fall apart when triggered. Because the chemistry is modular, future designers can mix and match building blocks to program how these materials behave in the body, in the environment, or in industrial settings. For non-specialists, the main message is that yesterday’s refinery byproduct can become tomorrow’s smart, degradable plastic—helping bridge the gap between performance and sustainability.
Citation: Sun, Y., Cao, Y., Liu, X. et al. Synthesis of poly(ester disulfide)s from S8-involved step-growth addition polymerization at ambient temperature. Nat Commun 17, 2066 (2026). https://doi.org/10.1038/s41467-026-68963-7
Keywords: sulfur polymers, dynamic covalent plastics, biodegradable materials, recyclable elastomers, green polymer chemistry