Clear Sky Science · en
HCMV infection disrupts barrier functions and promotes epithelial–mesenchymal transition in a cholangiocyte organoid model
Why this virus-in-the-liver story matters
Human cytomegalovirus is a very common virus that most of us carry without knowing it. Yet in newborns and people with fragile immune systems, it can be linked to serious liver and bile duct problems, including a rare but devastating childhood disease called biliary atresia. This study uses miniature, lab-grown human bile ducts to show how the virus can directly damage these delicate tubes and trigger scarring changes that may help explain long-debated links between infection and lifelong liver disease.
A closer look at a common but quiet virus
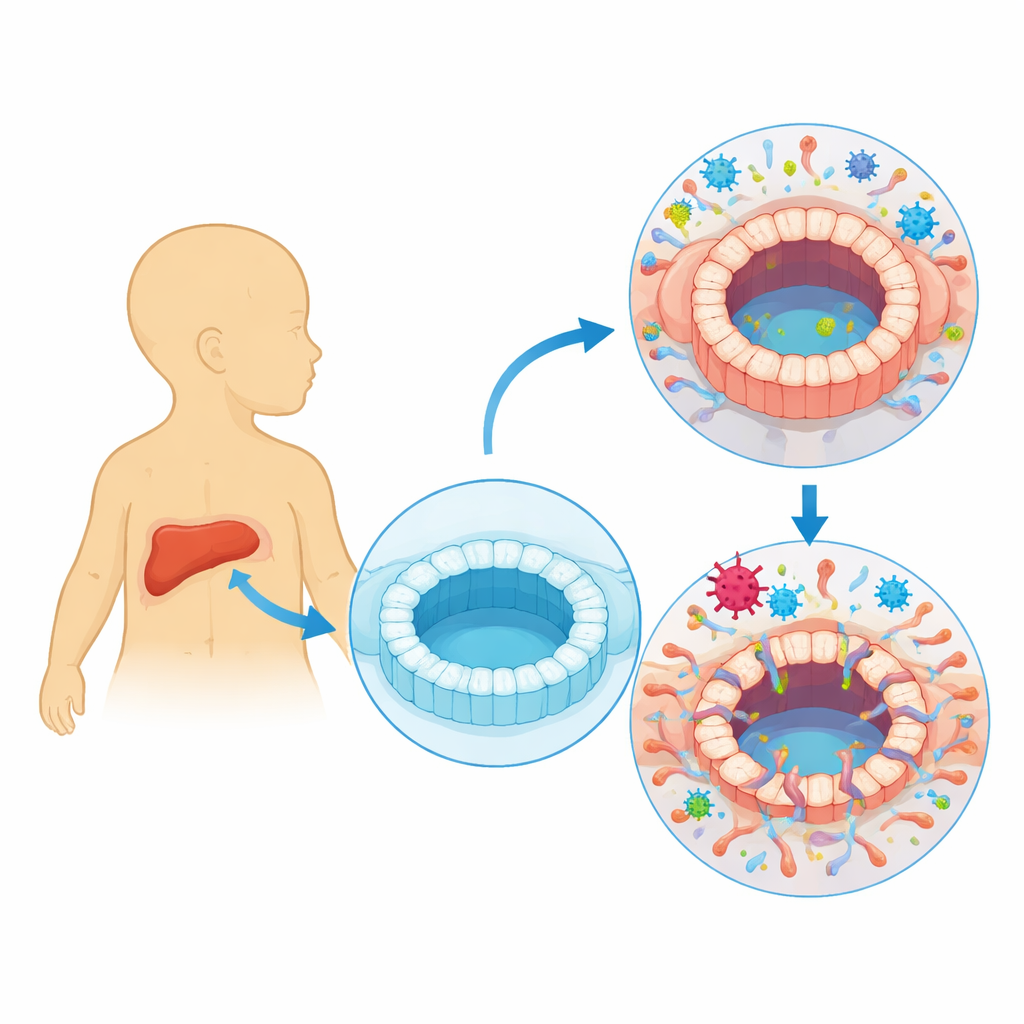
Human cytomegalovirus (HCMV) usually lives quietly in our bodies, switching between an active phase, where it makes new virus particles, and a silent phase, where it hides inside certain blood cells. Doctors have long noticed that people with clear HCMV infection can develop bile duct injury and cholestasis, in which bile cannot flow properly. Many infants with biliary atresia, a condition where bile ducts become blocked and scarred soon after birth, also show signs of recent HCMV infection. However, it has been unclear whether the virus actually infects the cells lining bile ducts or just shows up as a bystander during illness.
Building tiny bile ducts in a dish
To answer this, the researchers created three-dimensional “organoids” that mimic human bile ducts. They started with induced pluripotent stem cells, reprogrammed from human blood, and guided them to form cholangiocyte-like cells—the type that line bile ducts. These cells self-organized into hollow spheres with a central cavity and tight cell–cell connections, resembling a miniaturized bile duct. The team then exposed these organoids to a strain of HCMV tagged with a fluorescent marker that lights up when the virus is actively replicating. This allowed them to track infected cells and measure how the organoids changed over time.
How infection warps structure and weakens the barrier
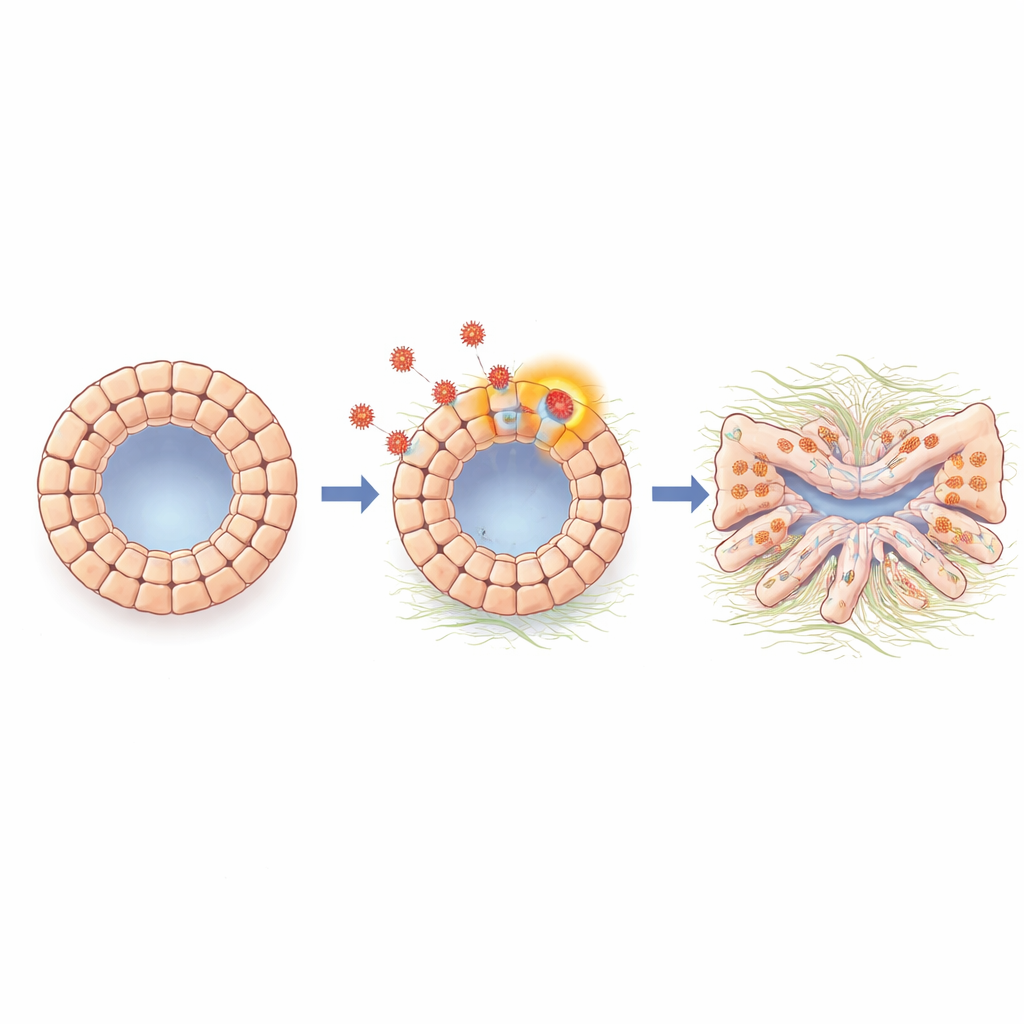
Once infected, the organoids stopped growing normally and became misshapen, with thickened and irregular walls. Some cells glowed brightly, showing full viral activity and production of new virus particles; others carried viral genetic material at lower levels, suggesting a more dormant, latency-like state. Functional tests revealed that infected organoids lost key bile duct abilities. They no longer swelled properly in response to a stimulus that depends on ion transport channels, and they leaked more readily in an electrical resistance assay that measures barrier tightness. Proteins that normally seal the spaces between cells, such as tight junction components, were reduced, and transport of a test dye into and out of the inner cavity became abnormal. Together, these changes show that HCMV can directly compromise the bile duct wall, making it leaky and dysfunctional.
Cells shift identity toward a scarring state
Digging deeper, the scientists analyzed gene activity across the organoids in bulk and at single-cell resolution. They found a strong activation of an epithelial–mesenchymal transition–like program, a process in which tightly connected surface cells loosen their bonds, adopt a more mobile, fibrous character, and start producing molecules associated with tissue scarring. Infected organoids showed increased levels of N-cadherin and other markers linked to this shift, as well as genes involved in fibrosis and matrix production. A signaling molecule called TGF-beta, well known for driving scarring in many organs, was boosted during infection. When the team blocked TGF-beta signaling with a small-molecule inhibitor or an antibody, the organoids regained a more normal shape, and many of the scarring- and transition-related genes dropped back toward baseline, indicating that TGF-beta is a key middleman connecting viral presence to damaging cell changes.
From lab-grown tubes back to sick children
To see whether these lab observations relate to real disease, the researchers examined liver tissue from children with biliary atresia and from other pediatric liver conditions. Using sensitive probes, they detected HCMV DNA in cholangiocytes—the bile duct cells—in about two-thirds of the biliary atresia samples, but far less often in non–biliary atresia livers. In the same regions where viral DNA was found, the bile duct cells displayed higher levels of TGF-beta and N-cadherin, matching the transition and scarring pattern seen in the organoids. This suggests that in at least a subset of children, HCMV infects bile duct cells directly and tips them toward a fibrotic state by turning on TGF-beta–driven pathways.
What this means for understanding and treating bile duct disease
In simple terms, this work shows that a common virus can invade the cells that line bile ducts, loosen their tight connections, and push them toward a scarring, less specialized identity through a TGF-beta–based signal. These changes weaken the ducts’ barrier and transport functions and may contribute to the blocked, fibrotic bile ducts seen in diseases like biliary atresia. While not every case of bile duct disease will be caused by HCMV, the study strengthens the case that, in vulnerable infants, viral infection could be one important piece of the puzzle—and that targeting TGF-beta–related pathways might one day help protect or restore fragile bile ducts.
Citation: Ye, Z., Hu, X., Rahaman, S.M. et al. HCMV infection disrupts barrier functions and promotes epithelial–mesenchymal transition in a cholangiocyte organoid model. Nat Commun 17, 2273 (2026). https://doi.org/10.1038/s41467-026-68962-8
Keywords: cytomegalovirus, biliary atresia, bile duct organoids, epithelial mesenchymal transition, TGF-beta signaling