Clear Sky Science · en
Divergent photochemical ring-replacement of isoxazoles
Shining Light on Drug Building Blocks
Many modern medicines are stitched together from small ring-shaped fragments that control how a drug behaves in the body. Chemists often want to swap one such ring for a closely related one to fine‑tune activity, but today that usually means rebuilding the whole molecule from scratch. This study shows that carefully chosen ultraviolet light can remodel a common ring, called an isoxazole, directly into several other useful rings, potentially saving time, cost, and effort in drug discovery.
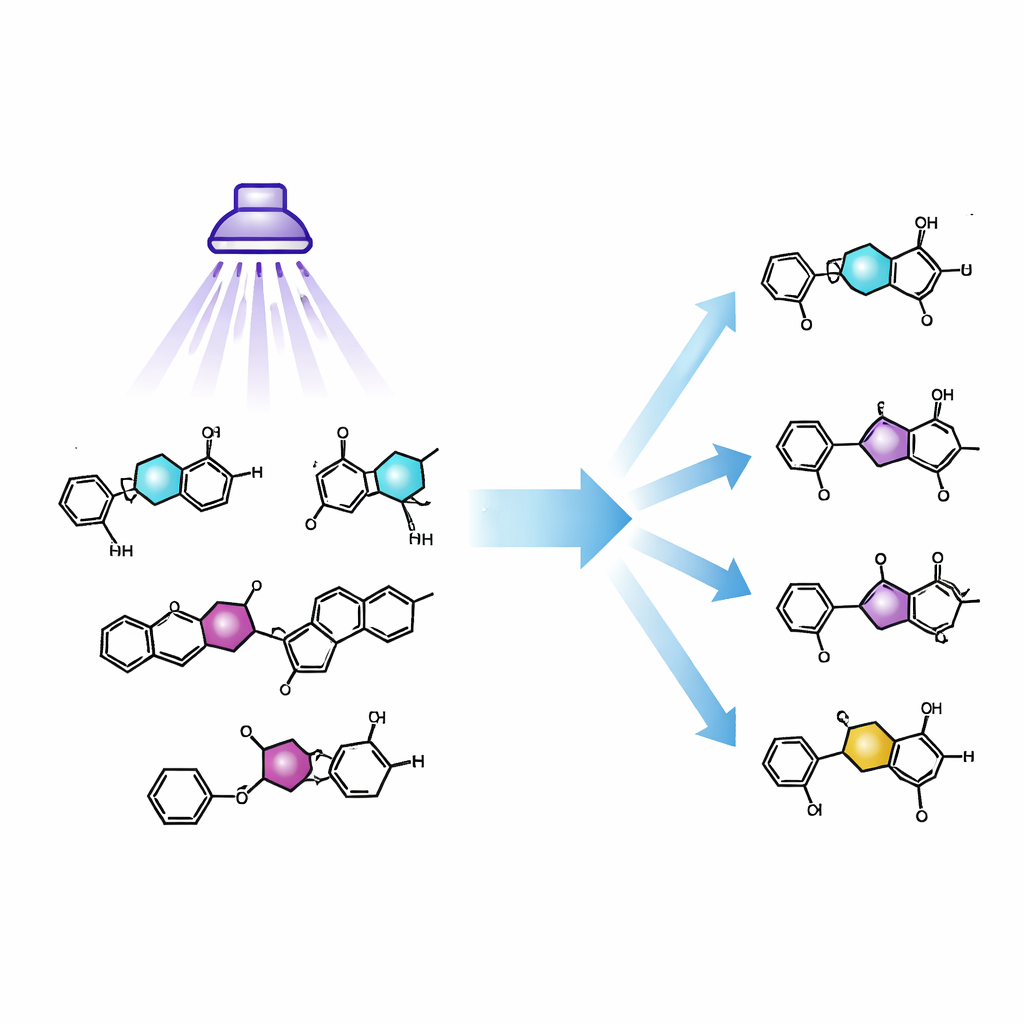
Why These Tiny Rings Matter
Five‑membered rings containing oxygen, nitrogen, or sulfur atoms are everywhere in pharmaceuticals and crop‑protection agents. Isoxazoles and oxazoles, in particular, can stand in for common features like ketones or esters while often improving a drug’s stability and potency. When researchers explore how changing a ring affects biological activity, they typically prepare a separate version of the molecule for each ring type, a laborious process known as de novo synthesis. A method that could start from a single isoxazole‑based lead and directly convert its ring into several close relatives would greatly accelerate this exploration of chemical space.
Using Light as a Precision Tool
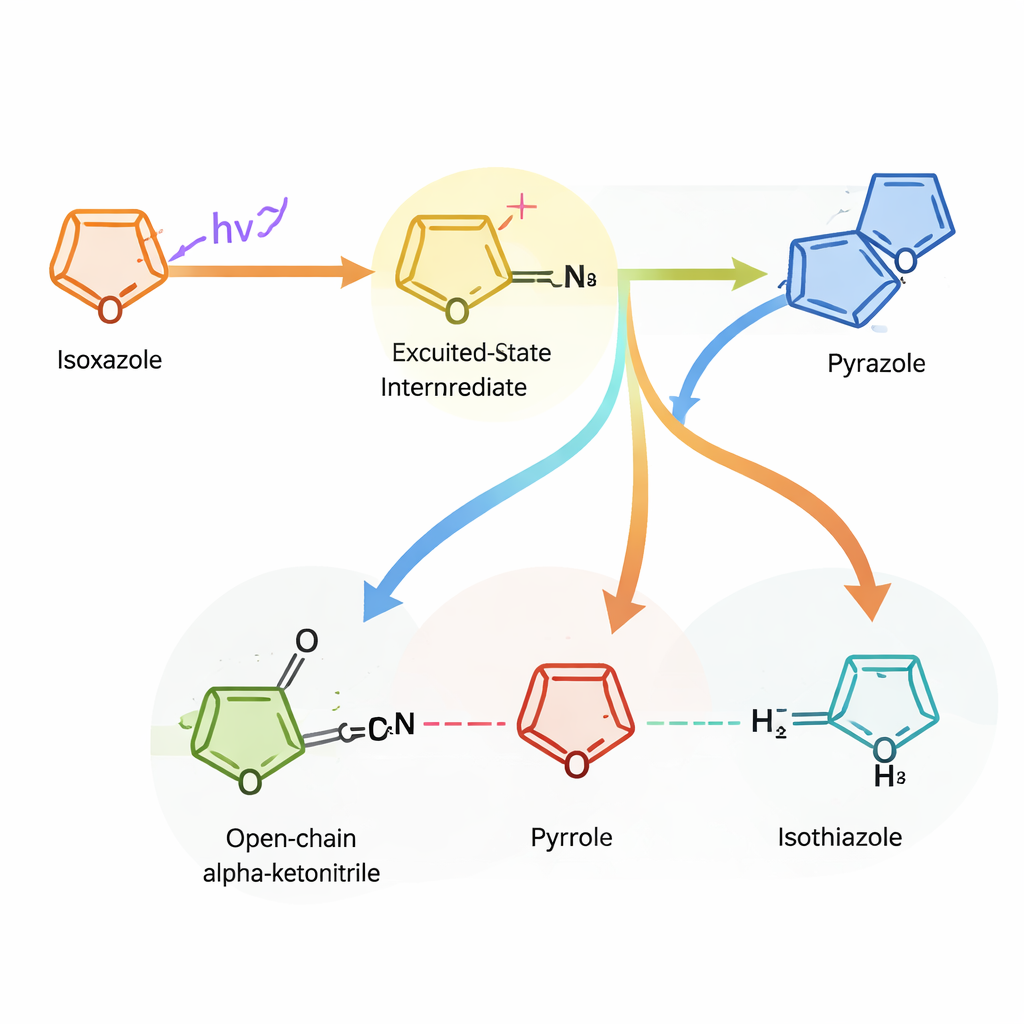
The authors discovered that shining ultraviolet light on isoxazole rings can trigger highly selective rearrangements, provided the molecule’s “decorations” and the surrounding liquid are chosen with care. Beginning with a simple isoxazole, they found conditions where light converts it into an oxazole ring or, alternatively, breaks the ring open to form an intermediate called an alpha‑ketonitrile. Earlier work had hinted at such behavior but suffered from poor yields and messy product mixtures. Here, the team systematically varied the ring’s substituents and the solvent, mapping when clean ring‑swapping occurs and when the molecule instead falls apart or remains unchanged.
Peeking Under the Hood with Theory
To understand why small structural tweaks lead to such different outcomes, the researchers turned to quantum‑chemical calculations. These computer studies show that, after absorbing light, the isoxazole briefly visits an excited state in which a key bond snaps, creating a highly energetic intermediate. From there, the system can either re‑close into the original ring, contract into a three‑membered “azirine,” or rearrange further. Whether the process continues cleanly toward a new ring or fizzles out into decomposition depends sensitively on where substituents sit on the original ring and how well each intermediate absorbs additional light. This analysis revealed that isoxazoles bearing certain groups at a specific ring position are especially well suited for controlled ring replacement.
From One Ring to Many
Armed with these insights, the team focused on a family of isoxazoles that respond reliably to light. In alcohol solvents, these substrates smoothly morph into oxazoles under mild conditions and tolerate a wide range of additional functional groups, including fragile ones often found in drug candidates. In a less polar solvent, the same light instead yields the alpha‑ketonitrile intermediate, which can be transformed in one pot into several other rings—pyrazoles, pyrroles, amino‑substituted isoxazoles, and isothiazoles—using straightforward follow‑up reactions. Starting from just seven commercially available isoxazoles, the authors assembled a library of 34 distinct heterocyclic products without rebuilding any molecule from the ground up.
Implications for Future Medicines
This work introduces a practical way to “edit” the core ring of complex molecules late in the synthesis, rather than rebuilding them from simpler pieces. By using light to trigger selective ring remodeling, chemists can rapidly generate families of closely related structures and test how each behaves in biological systems. The method’s gentle conditions and compatibility with real drug molecules suggest it could become a valuable tool for medicinal chemists seeking better treatments with fewer synthetic steps.
Citation: Xu, Y., Poletti, L., Arpa, E.M. et al. Divergent photochemical ring-replacement of isoxazoles. Nat Commun 17, 2141 (2026). https://doi.org/10.1038/s41467-026-68960-w
Keywords: photochemical ring editing, isoxazole remodeling, heterocycle diversification, medicinal chemistry methods, scaffold hopping