Clear Sky Science · en
Bio-based oxalic acid production in Issatchenkia orientalis enables sustainable rare earth recovery
Turning Microbes into Clean-Energy Helpers
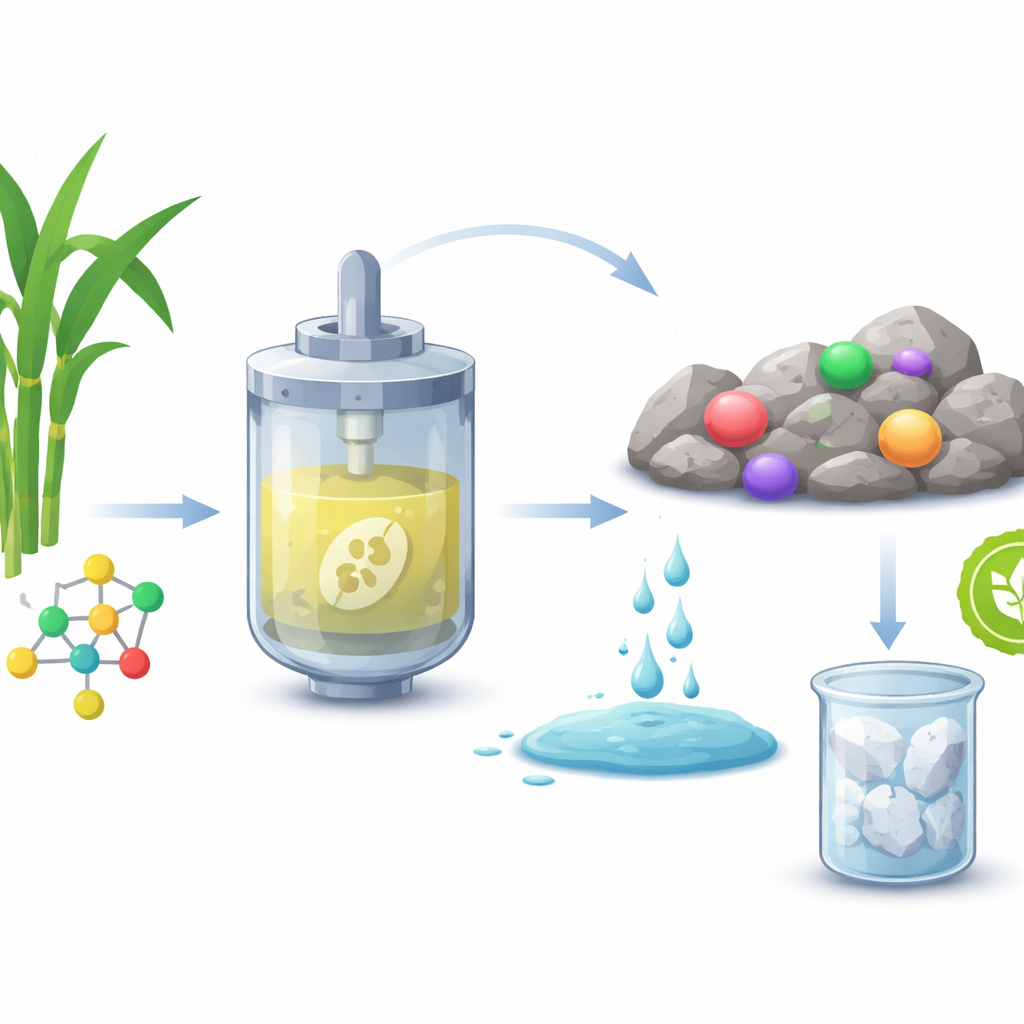
From smartphones to wind turbines, many modern devices rely on rare earth elements that are hard to extract without polluting the environment. This study shows how engineers turned a hardy yeast into a tiny chemical factory that makes oxalic acid, a simple organic acid, from plant sugars. That biological oxalic acid can then pull rare earth metals out of watery mixtures efficiently, offering a cleaner and potentially cheaper way to secure the materials needed for the energy transition.
Why Rare Metals and Simple Acids Matter
Rare earth elements sit at the heart of powerful magnets used in electric vehicles, wind turbines, and advanced electronics. Getting them out of rock and recycling streams, however, typically involves long chemical processes and fossil-based reagents. Today, most oxalic acid—an important tool for grabbing rare earths from solution and turning them into solid crystals—is made using petroleum-derived ingredients under harsh conditions. That means high energy use, hazardous chemicals, and extra waste. As demand for rare earths climbs, the need for cleaner and more reliable supplies of both the metals and the processing chemicals becomes urgent.
Recruiting a Tough Yeast as a Mini Factory
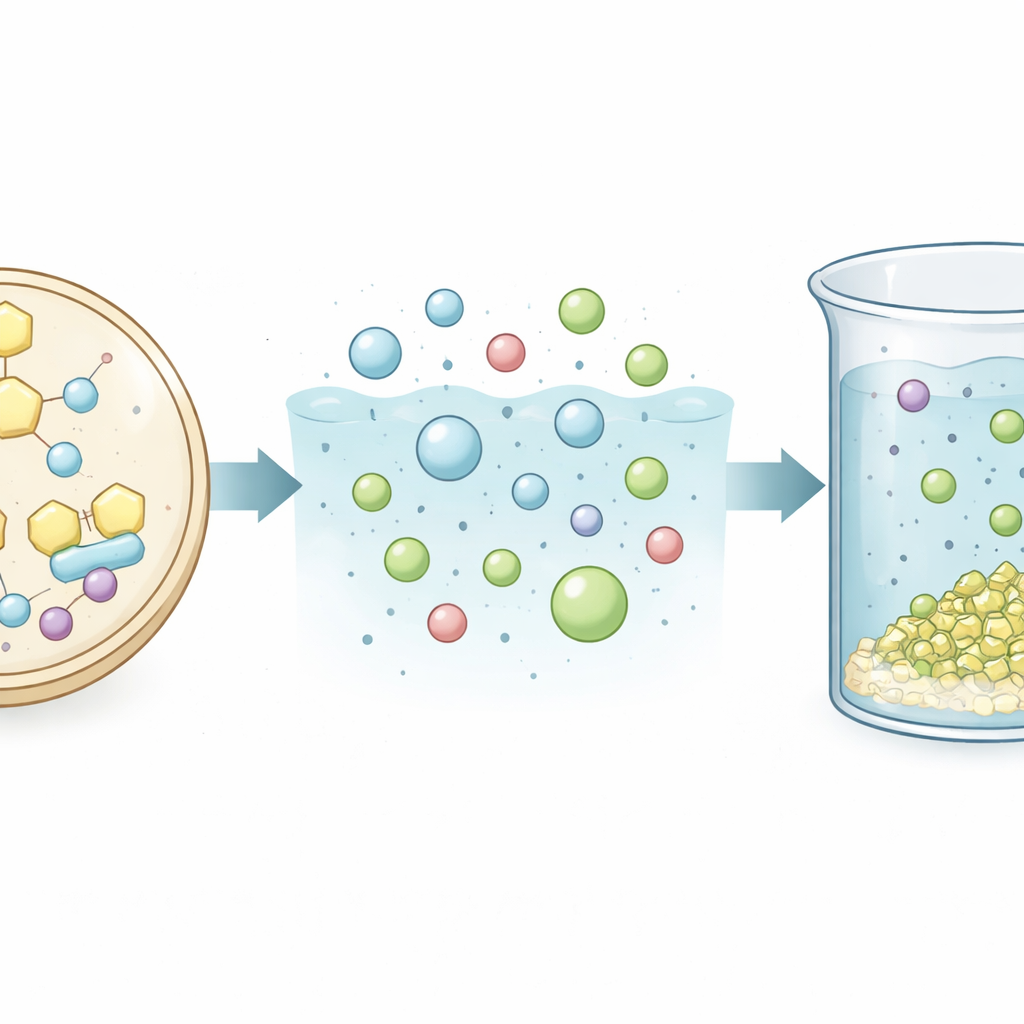
The researchers chose an unusual yeast species, Issatchenkia orientalis, as their production workhorse. Unlike many microbes that struggle in acidic surroundings, this yeast thrives across a very low pH range, which fits well with the acidic conditions already used in rare earth processing. The team rewired its metabolism by inserting genes from fungi and plants so that the yeast could convert sugar first into an intermediate called oxaloacetate and then into oxalic acid. They added extra copies of key enzymes to push more carbon through this route, removed one gene to stop wasteful glycerol byproduct formation, and tweaked how the cell balances energy use. Step by step, they built a final strain that produced nearly 40 grams of oxalic acid per liter in fed-batch fermentation at pH 4, all while maintaining a simple, easy-to-handle cell shape.
Using Fermentation Broth Straight from the Tank
Instead of purifying the oxalic acid—a step that normally adds cost, energy use, and waste—the team tested whether the raw fermentation liquid could be used as is. They mixed this broth with solutions containing individual rare earth salts such as neodymium, dysprosium, and lanthanum. The bio-based oxalic acid caused more than 98–99% of these metals to form solid crystals and fall out of the liquid, closely matching the performance of high-purity, commercially made oxalic acid. When they moved to a tougher challenge—an acidic leachate made by dissolving a low-grade rare earth ore rich in impurities—the crude broth still pulled out more than 99% of the total rare earth content while leaving most unwanted metals behind. Structural tests using X-ray diffraction and infrared spectroscopy showed that the crystals formed with bio-oxalic acid were nearly indistinguishable from those made with the conventional product.
Counting Costs and Carbon Footprints
To understand whether this bio-based route could compete at industrial scales, the authors modeled a full facility that turns sugarcane into oxalic acid and then ships it to rare earth processors. Their techno-economic analysis suggests a minimum selling price of about $1.79 per kilogram—squarely within today’s market range for oxalic acid. A life cycle assessment went further, indicating that the process could even become carbon-negative when surplus electricity from burning sugarcane residues is used to offset fossil power. Compared with standard fossil-derived oxalic acid, the modeled system cuts greenhouse gas emissions by more than half, and potentially by more than 100% when electricity displacement is counted. The analysis also highlights that improving fermentation yield and production rate would drive costs down further, while very high peak concentrations matter less because the product can be used without purification.
What This Means for Future Green Metals
By marrying metabolic engineering with mineral processing, this work outlines a new way to connect biology and critical material supply chains. A specially designed yeast can churn out oxalic acid under acidic, industry-relevant conditions, and the resulting liquid can be poured directly into rare earth recovery steps to crystallize metals with high efficiency and purity. The approach promises a more sustainable and flexible supply of a key processing chemical, with lower carbon emissions and fewer hazardous reagents. With further improvements in strain robustness, fermentation performance, and integration into real-world mining and recycling operations, bio-based oxalic acid could become a cornerstone of cleaner rare earth production and, by extension, of the clean-energy technologies that rely on these metals.
Citation: Lu, J., Guo, W., Dong, Z. et al. Bio-based oxalic acid production in Issatchenkia orientalis enables sustainable rare earth recovery. Nat Commun 17, 2193 (2026). https://doi.org/10.1038/s41467-026-68957-5
Keywords: rare earth elements, bio-based oxalic acid, metabolic engineering, sustainable mining, yeast fermentation