Clear Sky Science · en
AI-based multiomics profiling reveals complementary omics contributions to personalized prediction of cardiovascular disease
Why predicting heart trouble earlier matters
Heart disease and stroke remain the world’s top killers, often striking seemingly healthy people without warning. Doctors already use checklists of age, blood pressure, cholesterol, and smoking to gauge risk, but these tools can miss many future patients and overestimate danger in others. This study asks a timely question: if we look deeper into the molecules circulating in our blood and analyze them with artificial intelligence, can we spot cardiovascular disease years before it appears—and tailor prevention to each person?
Looking inside the blood for hidden warning signs
The researchers tapped the UK Biobank, a massive health project that tracks hundreds of thousands of volunteers over many years. For a subset of these participants, scientists had carefully measured thousands of molecules in blood samples: small metabolites related to fats, sugars, and amino acids, and proteins involved in inflammation, clotting, and other body processes. The team focused on six major cardiovascular conditions—coronary artery disease, stroke, heart failure, atrial fibrillation, peripheral artery disease, and venous blood clots—to see whether these molecular fingerprints could forecast who would develop which illness.
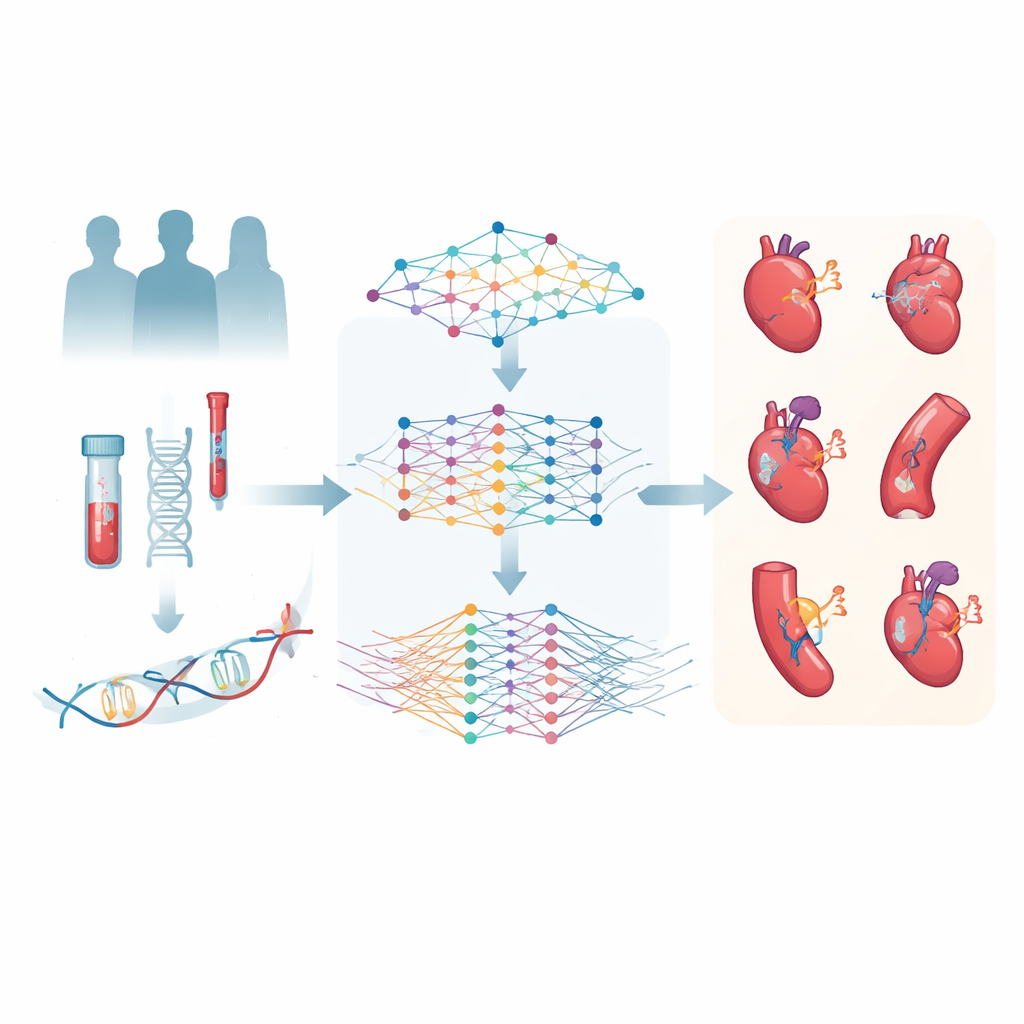
Teaching artificial intelligence to read molecular patterns
To make sense of nearly 3,000 proteins and 168 metabolites, the authors built two deep learning models, MetNet and ProNet. Rather than predicting one disease at a time, these models learned patterns related to all six cardiovascular outcomes at once. From the metabolite data, MetNet produced a combined risk score called MetScore; from the protein data, ProNet produced ProScore. Each person thus received six scores per system—one for each type of cardiovascular disease—summarizing millions of possible molecular interactions into a handful of numbers that a standard statistical model could use alongside age, blood pressure, medications, and genetic risk.
How much better are these molecular risk scores?
When the team tested the scores in an independent group of 24,287 people who had all types of data available, both MetScore and ProScore were strong predictors on their own, clearly separating participants into low-, medium-, and high-risk groups over 15 years of follow-up. Protein-based scores performed best, often outperforming traditional polygenic (DNA-based) risk measures by a wide margin. Adding ProScore and MetScore to conventional clinical factors improved the accuracy of risk prediction for every cardiovascular outcome studied, even when baseline models were already detailed. In some cases, particularly for peripheral artery disease and atrial fibrillation, the boost in performance was substantial, and decision-curve analyses suggested that doctors could make more beneficial choices about who should receive preventive treatment.
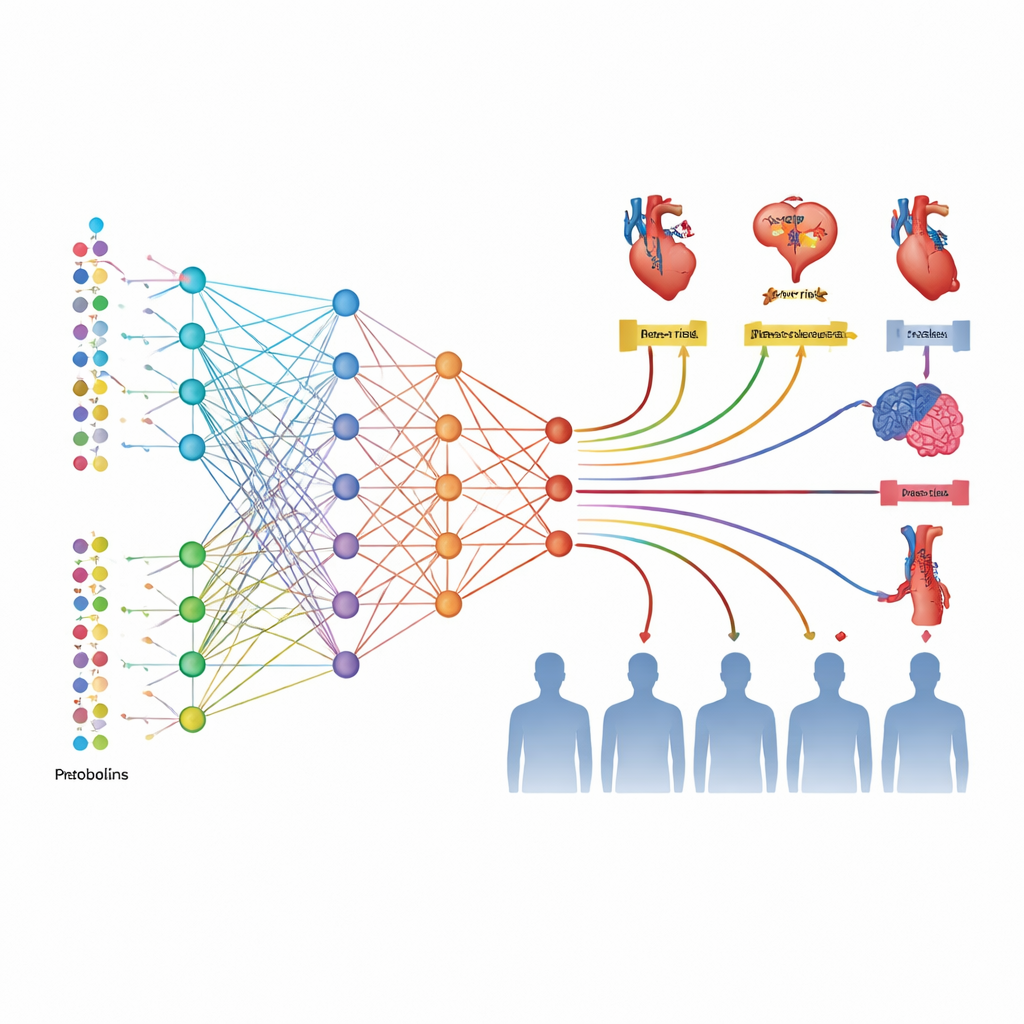
What the molecules reveal about disease biology
Beyond prediction, the researchers asked which specific molecules were most influential in the AI models using an explanation method called SHAP. They confirmed the importance of familiar markers such as creatinine and albumin (reflecting kidney and overall health), as well as inflammatory signals like GlycA and heart-stress proteins such as NT-proBNP. At the same time, the models highlighted lesser-known proteins and metabolites tied to inflammation, clotting, vessel remodeling, and even nerve injury, with partly shared and partly disease-specific patterns. Interestingly, no single molecule matched the predictive power of the combined MetScore or ProScore, underscoring that cardiovascular risk arises from many subtle changes acting together rather than a lone culprit.
From big data to more personal heart care
The study concludes that weaving together genetics, detailed molecular blood profiles, and routine clinical information can meaningfully sharpen our view of who is likely to develop major cardiovascular diseases, often a decade or more in advance. Protein measurements, in particular, appear to carry rich, actionable information about ongoing biological stress long before symptoms arise. While the required tests are not yet common or inexpensive, costs are falling, and the authors have made their CardiOmicScore approach available as a prototype tool. With further validation in more diverse populations, such AI-driven multiomics profiling could help clinicians move from one-size-fits-all checklists toward truly personalized prevention—identifying high-risk individuals earlier, matching treatments to underlying biology, and potentially reducing the global burden of heart and vascular disease.
Citation: Luo, Y., Zhang, N., Yang, J. et al. AI-based multiomics profiling reveals complementary omics contributions to personalized prediction of cardiovascular disease. Nat Commun 17, 2269 (2026). https://doi.org/10.1038/s41467-026-68956-6
Keywords: cardiovascular risk prediction, proteomics, metabolomics, deep learning, biomarkers