Clear Sky Science · en
Inverse palladocenes
A New Twist on Metal Sandwich Molecules
Metals arranged in tiny, well-ordered clusters already underpin catalysts, electronics, and medicines. This study introduces a surprising new family of such molecules, called inverse palladocenes, where the usual roles of metal and nonmetal are flipped. These miniature structures not only challenge how chemists think about bonding, but also turn invisible near-infrared light into heat with remarkable efficiency, hinting at future uses in laser shielding, controlled heating, and high-temperature materials.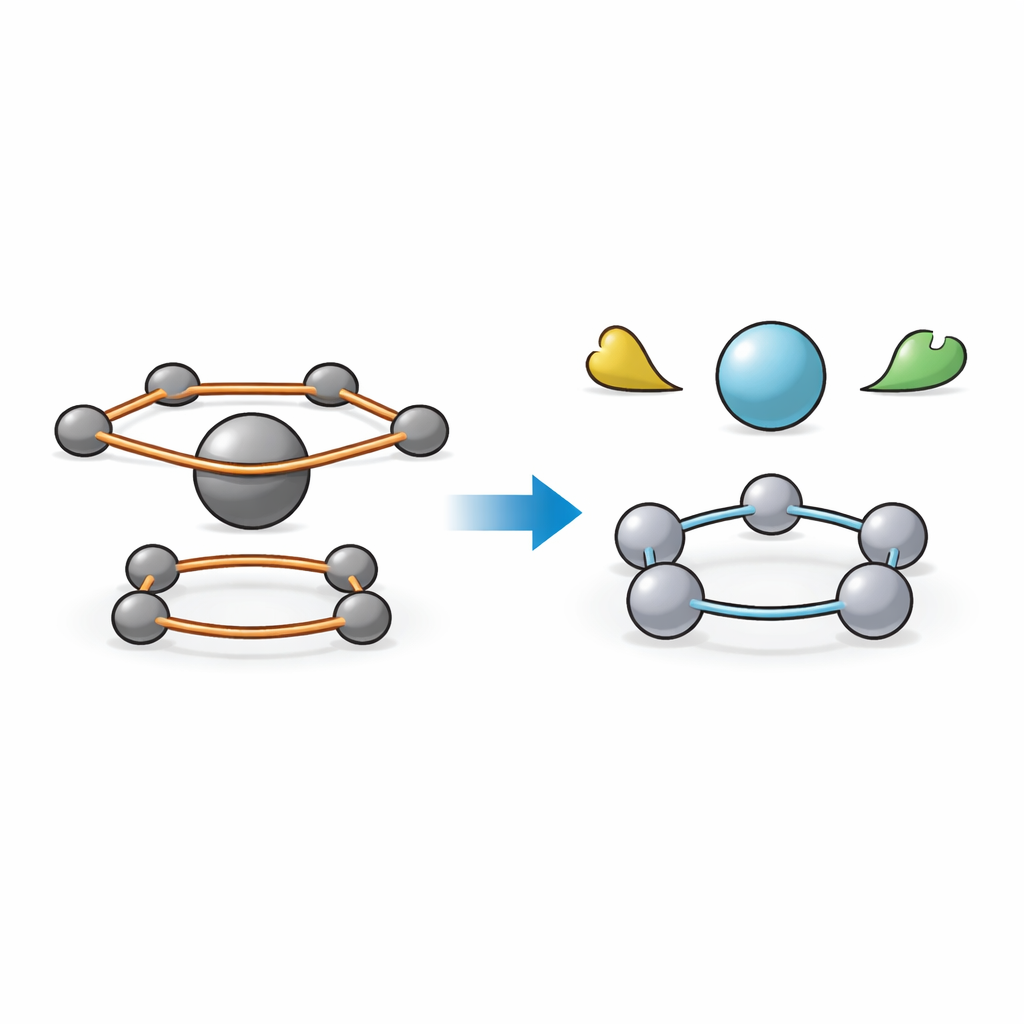
From Classic Sandwiches to Inside-Out Designs
Traditional "metallocenes" look a bit like a burger: a metal atom sits between two flat carbon rings, which provide stability and special electronic properties. The new work asks what happens if you invert that idea. Instead of a metal center held by carbon rings, the researchers built a flat ring made of five palladium metal atoms, coordinated to a central phosphorus atom and surrounded by protective organic groups. This is the key "inverse" unit. The team synthesized several related clusters, each containing this five-atom palladium ring, showing that the structure is not a one-off curiosity but a repeatable building block for a broader class of materials.
A Metal Ring that Behaves Like Aromatic Carbon
Chemists prize so-called aromatic rings, such as those in benzene, because their electrons are shared evenly around the ring, making them unusually stable. Using X-ray crystallography and advanced quantum calculations, the authors showed that their five-palladium ring behaves in an analogous way: electrons circulate and delocalize over all five metal atoms. They introduced a simple way to gauge how well this shared-electron network works by looking at how equal the metal–metal bond lengths are and how flat the ring is. The more uniform and planar the ring, the stronger the conjugation, or electron sharing. Across the series of clusters, one called Pd5–C had the most evenly shared bonds and nearly perfect planarity, marking it as the most strongly conjugated and most aromatic member.
Blurring the Boundary Between Metals and Molecules
In the crystal, the metal rings in Pd5–C stack face-to-face with nearby carbon rings from surrounding ligands at distances similar to the familiar "π–π" stacking seen between organic aromatic molecules. Computations showed that the interaction between the metal ring and the carbon ring is dominated by gentle electrostatic attraction, much like the forces that hold stacked aromatic molecules together. This finding reveals that the metal ring behaves much like a classic organic aromatic ring, but built from palladium atoms instead of carbon. It also demonstrates that tiny changes in the attached ligands—a single oxygen atom swapped for a carbon, for example—can reorganize how the clusters assemble in the solid state, giving rise to different one-dimensional and layered superstructures.
Turning Invisible Light into Intense Heat
When solutions containing these inverse palladocenes were illuminated with near-infrared light, especially in the so-called NIR-II window around 980 nanometers, they heated up dramatically. Measurements showed that all of the new clusters absorb strongly in this region, but again Pd5–C stood out: it converted about 74% of the incoming light into heat, far outperforming many reported photothermal materials. On a per-atom basis, each palladium atom in Pd5–C was responsible for an average conversion efficiency of roughly 15%, a remarkably high figure. Detailed calculations and experiments showed that this heating originates almost entirely from the five-palladium ring itself, not from the surrounding ligands. Even after stripping many of those ligands away, the heat-generation performance persisted over repeated heating–cooling cycles, underscoring the robustness of the metal ring.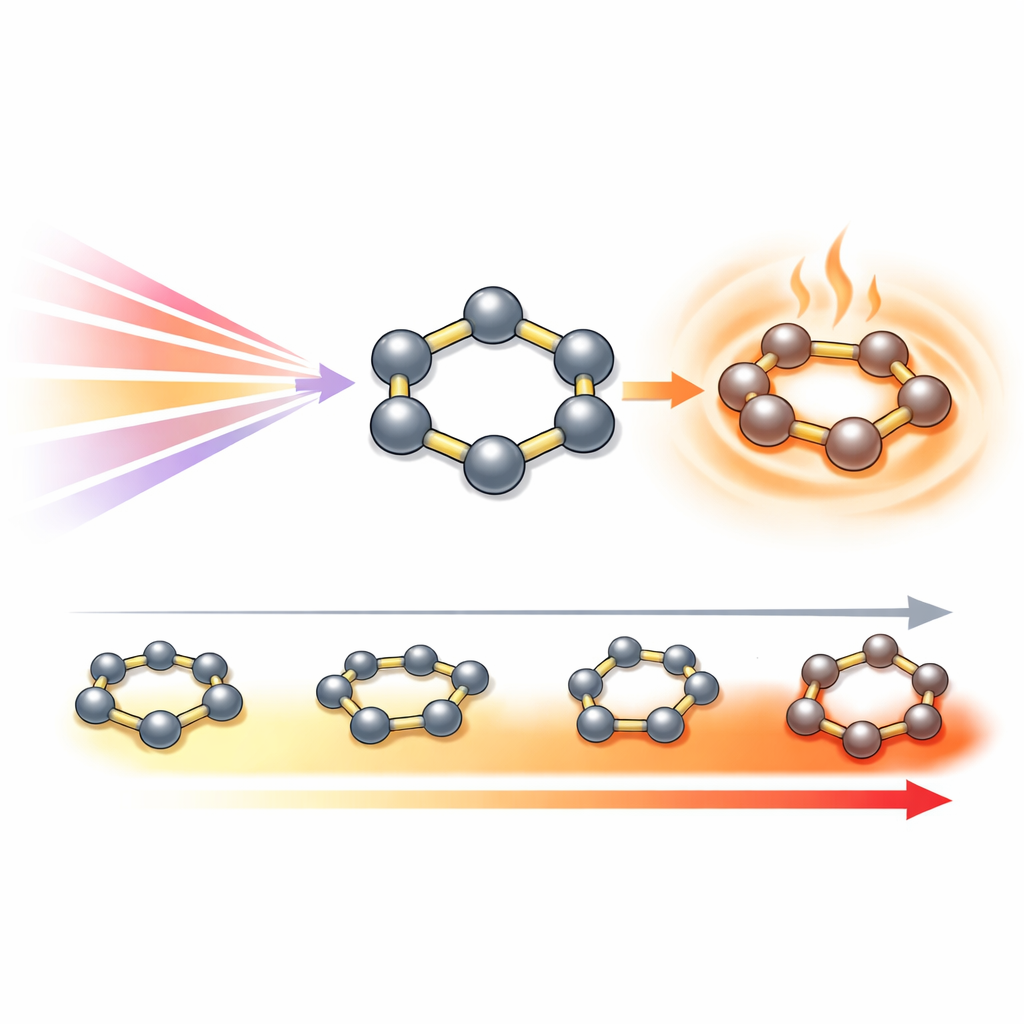
Real-World Uses: From Laser Shields to Precision Heating
The exceptional light-to-heat conversion translates directly into practical effects. Concentrated solutions of Pd5–C can absorb and dissipate more than 95% of a strong 980-nanometer laser beam, acting as an effective optical shield. When embedded in plastics such as polystyrene or polyurethane, the clusters enable rapid, localized heating: they can help degrade high-melting polymers, ignite cotton under laser exposure, or maintain a film at a very stable elevated temperature under continuous illumination. Because the same tiny metal ring controls both stability and photothermal behavior, these inverse palladocenes act like molecular "heat pixels" that can be placed into different hosts. Altogether, the work establishes a new class of inside-out metallocenes built around aromatic palladium rings, opening routes to materials that blur the line between metal clusters and organic molecules while offering powerful, controllable responses to near-infrared light.
Citation: You, Q., Jiang, XL., Zhao, Y. et al. Inverse palladocenes. Nat Commun 17, 2171 (2026). https://doi.org/10.1038/s41467-026-68955-7
Keywords: inverse palladocenes, metal aromaticity, palladium nanoclusters, near-infrared photothermal, laser shielding materials