Clear Sky Science · en
Understanding alkali metal promotion in hydrogenation catalysis through Strong Metal–Base Interaction
Why tweaking catalysts matters for everyday life
Turning simple molecules like carbon dioxide and hydrogen into useful fuels and chemicals is central to cleaner energy and greener industry. Many of the metal catalysts that drive these reactions already contain tiny doses of alkali metals such as sodium or potassium as “promoters,” but their true role has been surprisingly murky. This study uncovers a unifying principle for how these additives actually work, offering a way to design smarter catalysts that can dial reactions toward desired products and away from waste.
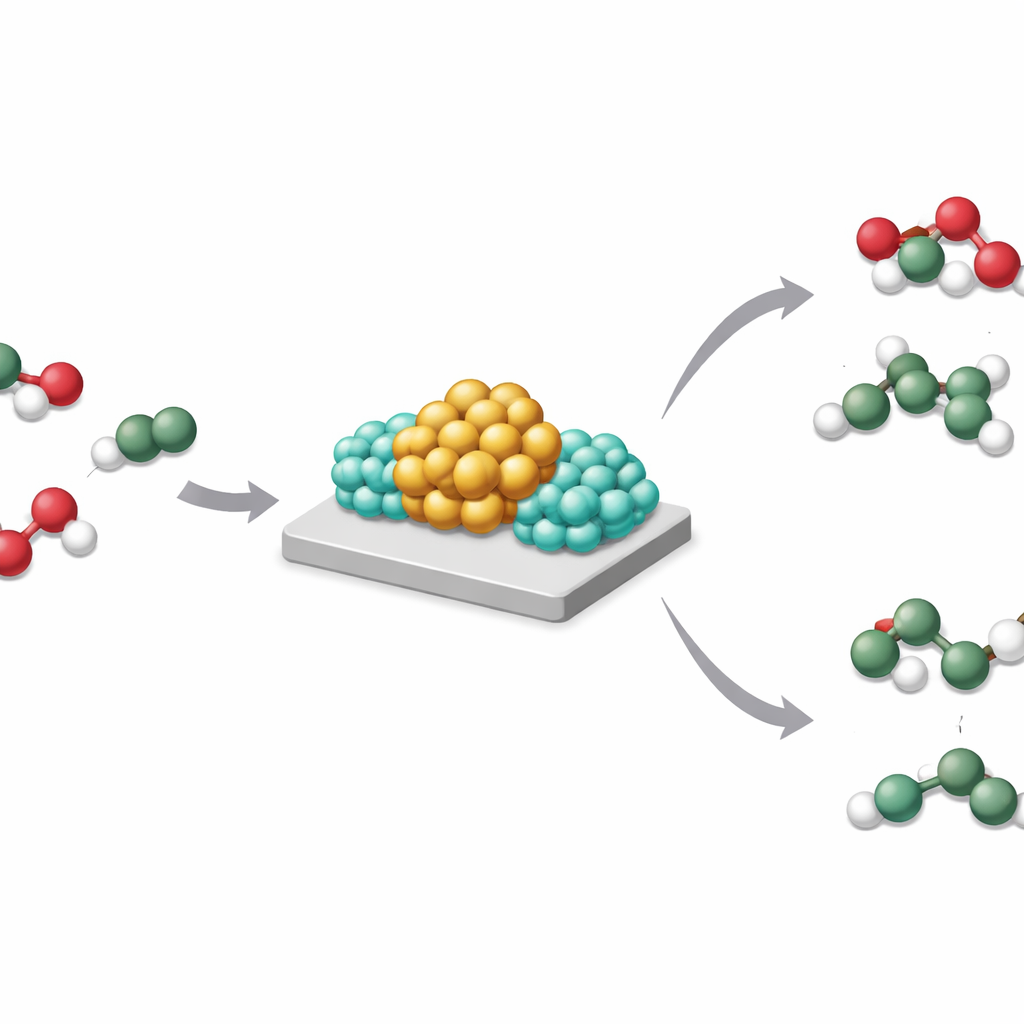
A closer look at boosted metal catalysts
Catalysts made from transition metals like rhodium and nickel sit on solid supports and help break and remake chemical bonds. For decades, industry has sprinkled in alkali metals to make these catalysts more active, more selective, or longer‑lived. The usual explanations focused on simple geometric effects (changing particle shapes and sizes) or direct electron donation from the alkali metal to the active metal. However, in real operating conditions these promoters exist mainly as oxides, not as bare metal atoms, which makes straightforward electron transfer an incomplete picture.
Discovering a strong partnership at the surface
The authors studied a model system: rhodium nanoparticles on titanium dioxide, with and without added sodium. Using advanced electron microscopy, X‑ray spectroscopy, and computer simulations, they found that sodium sits as an oxide, Na₂O, right at the edge where the rhodium particles touch the support. Under hydrogen, this arrangement creates what they call a Strong Metal–Base Interaction, or SMBI. At these special contact points, incoming hydrogen molecules split unevenly: the more basic Na₂O grabs the proton‑like part, while the rhodium holds the electron‑rich partner. This “heterolytic” split leaves the rhodium surface unusually rich in electrons and keeps hydrogen from spreading, or “spilling over,” onto the titanium dioxide support.
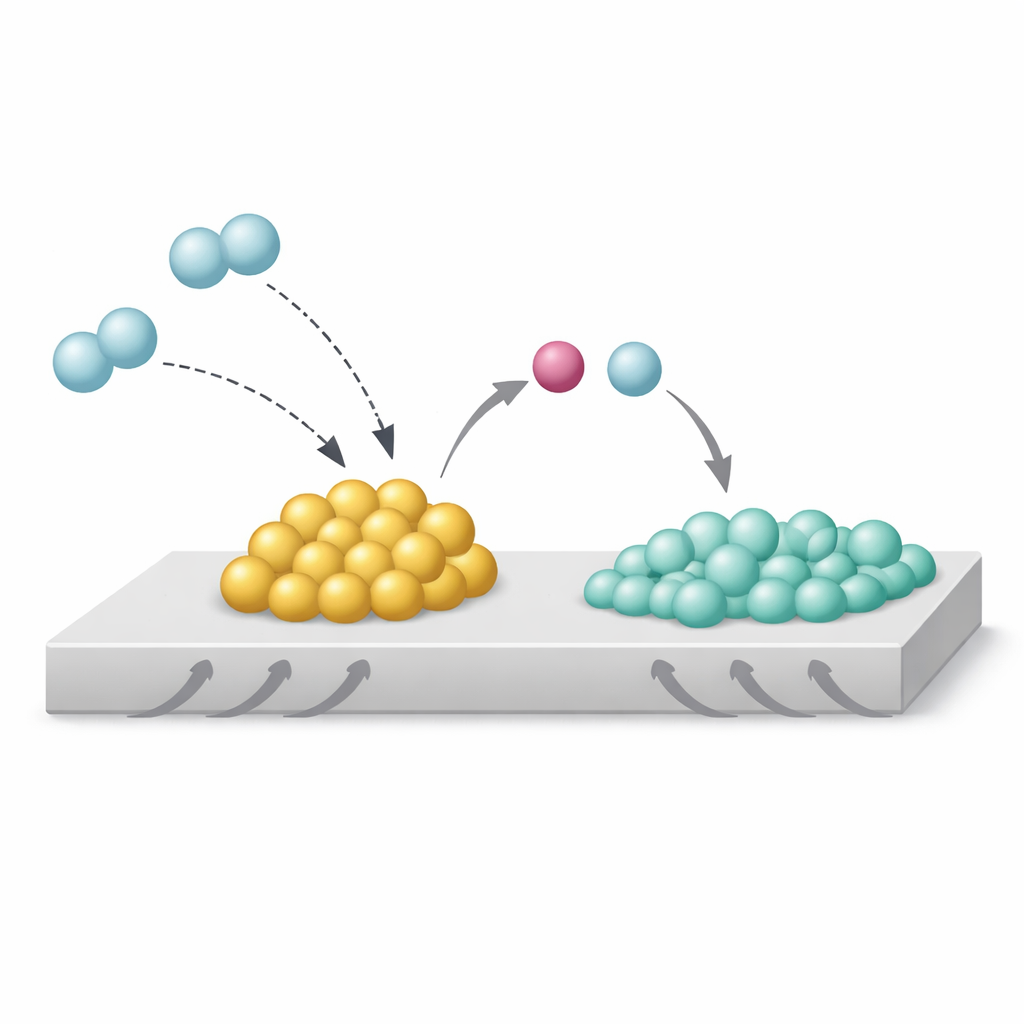
How this hidden effect steers reactions
This subtle reshaping of where hydrogen and electrons reside has big consequences. In carbon dioxide hydrogenation, plain rhodium on titanium dioxide favors strong hydrogenation all the way to methane. When sodium is added, the same catalyst instead produces mainly carbon monoxide, a valuable building block, while making far less methane. Similar behavior appears when lithium or potassium are used, or when rhodium is swapped for nickel. The authors show that when hydrogen can freely spill over the support, deep hydrogenation dominates; when SMBI keeps hydrogen concentrated at the metal–base boundary and off the support, milder products are favored.
Different double bonds, different outcomes
The team then probed how SMBI affects common organic hydrogenation reactions involving carbon–carbon, carbon–oxygen, and nitrogen–oxygen double bonds. For a carbon–carbon double bond in vinyl acetate, sodium promotion makes the catalyst faster, likely because electron‑rich rhodium helps weaken that bond while more weakly bound hydrogen adds to it efficiently. In contrast, for molecules like acetophenone and nitrobenzene, whose reactive groups prefer to sit on the oxide support, the same sodium addition slows the reaction. Here, the suppressed hydrogen spillover means the support no longer receives enough hydrogen to drive those transformations, revealing that not all double bonds benefit equally from the same promotional effect.
Designing catalysts with intentional control
By combining experiments and theory, the authors argue that SMBI provides a unified way to understand how alkali metal promoters reshape catalytic surfaces. Rather than acting as simple electron donors, alkali oxides act as strong proton traps, forcing hydrogen to split in a polarized way and confining reactive hydrogen near the metal–base interface. This insight explains puzzling trends across many hydrogenation reactions and metals, and it points to a practical design rule: by tuning how much and where alkali oxides are placed around metal particles, chemists can deliberately steer reactions toward specific products while optimizing activity and stability for cleaner chemical processes.
Citation: Jung, M., Dickieson, M.P., Chen, P. et al. Understanding alkali metal promotion in hydrogenation catalysis through Strong Metal–Base Interaction. Nat Commun 17, 2465 (2026). https://doi.org/10.1038/s41467-026-68952-w
Keywords: hydrogenation catalysis, alkali metal promoters, CO2 conversion, heterogeneous catalysts, metal–oxide interfaces