Clear Sky Science · en
Human brain prefrontal cortex proteomics identifies compromised energy metabolism and neuronal function in Schizophrenia
Why brain energy and wiring matter in schizophrenia
Schizophrenia is often described in terms of hallucinations and delusions, but underneath the symptoms lies a problem in how brain cells use energy and communicate. This study dives into the chemistry of the prefrontal cortex—the brain area crucial for planning, decision‑making, and working memory—by directly measuring thousands of proteins in donated human brains. By comparing people with schizophrenia to carefully matched individuals without the illness, the researchers uncover a pattern: the brain’s energy factories seem to be running low while its communication and signaling machinery is pushed into overdrive.
Looking closely at the thinking center of the brain
The team focused on the dorsal prefrontal cortex, a region repeatedly linked to cognitive problems in schizophrenia. Using laser‑guided dissection, they separated the upper and deeper layers of this cortex from 96 post‑mortem brains (47 with schizophrenia and 49 controls). They then applied advanced mass spectrometry to quantify more than 5000 different proteins. A key strength of the work is that they repeated the analysis independently in both sets of cortical layers and corrected for many possible distortions, such as age, time between death and tissue preservation, and technical processing differences. The pattern of changes in schizophrenia turned out to be highly consistent across the upper and deeper layers, suggesting a broad, layer‑wide disturbance rather than a localized defect.
Energy factories running below capacity
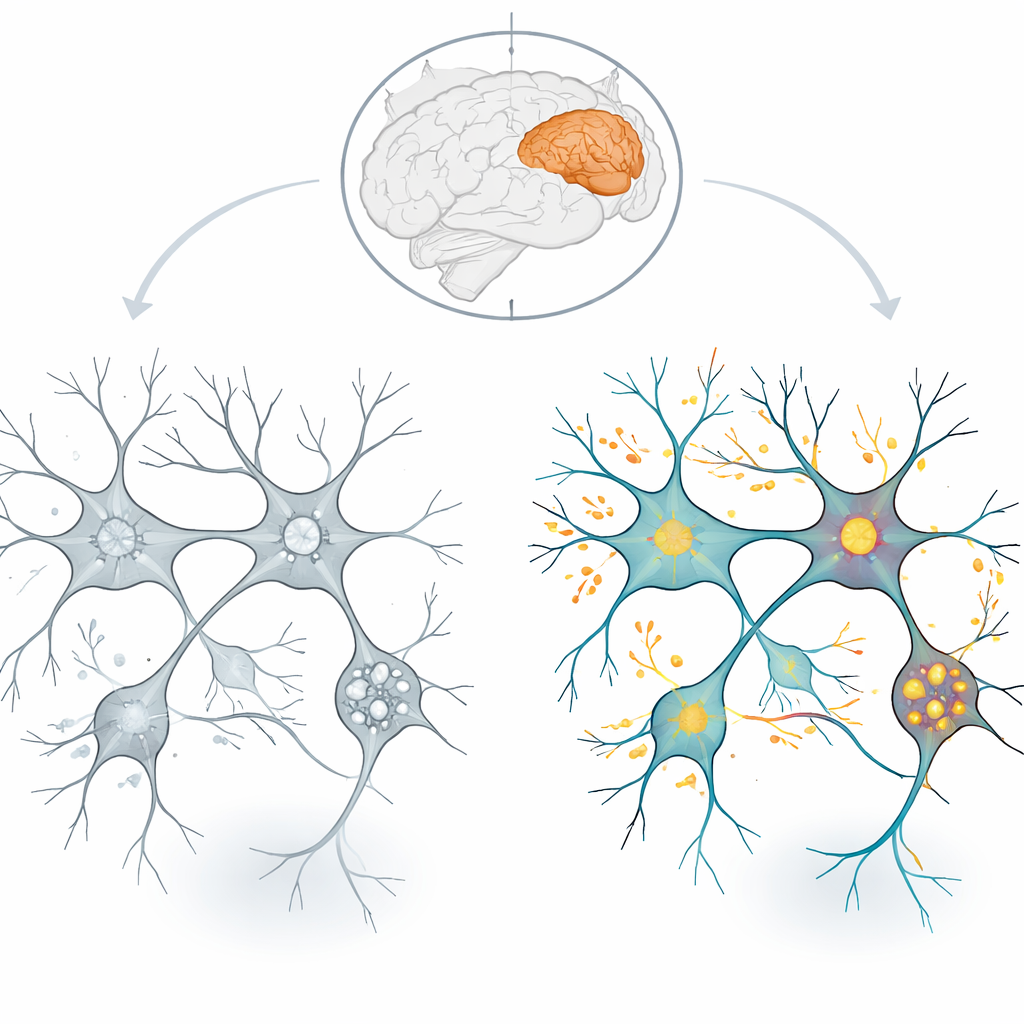
One of the clearest signals was a widespread drop in proteins that sit in the inner membrane of mitochondria, the tiny structures that generate most of a neuron’s fuel in the form of ATP. Components of several major steps in the energy‑producing chain—known as complexes I, II, IV, and V—were reduced. These complexes help move electrons and pump protons to drive ATP synthase, the final turbine that makes ATP. When the proteins that make up this machinery are scarcer, the system’s capacity to produce energy likely falls. The study also found fewer building blocks for ribosomes, the cell’s protein‑making machines, and elements of the proteasome, which breaks down worn‑out proteins. Together, these changes point to a brain environment with less energy and a reduced ability to constantly renew and clear cellular components.
Signaling and traffic systems in overdrive
In striking contrast, many proteins that control signals and cellular traffic were increased in schizophrenia brains. The researchers observed higher levels of numerous kinases, enzymes that turn other proteins on or off by adding phosphate groups, as well as proteins that regulate small molecular switches called GTPases. These switches help govern membrane trafficking and the movement of cargo within cells. Proteins involved in moving materials along the cell’s internal skeleton and in shuttling vesicles to and from the cell surface were also more abundant. Analyses focused on synapses—the junctions where neurons exchange information—showed that both sides of the connection, presynaptic and postsynaptic, carried altered sets of proteins. This suggests that the machinery that releases chemical messengers and the structures that receive them are both disturbed.
Connecting genes, brain cells, and disease
To place these protein changes in a broader biological context, the authors compared their results with large genetic studies of schizophrenia and with single‑nucleus RNA sequencing from the same brain cohort. Many of the altered proteins belonged to gene sets already implicated by genome‑wide association studies, particularly for schizophrenia and bipolar disorder, indicating that the observed protein disturbances ride on top of inherited risk. A substantial portion of the affected pathways—especially those tied to mitochondrial function and oxidative phosphorylation—showed similar trends at the RNA level, strengthening the case that these are not random noise. However, some features, such as the broad rise in kinases and specific synaptic regulators, stood out more clearly at the protein level, hinting at additional layers of control after RNA is made.
What this means for understanding and treating schizophrenia
For a lay reader, the main message is that schizophrenia in the prefrontal cortex looks like a mismatch between supply and demand. On one side, the brain’s energy‑producing and housekeeping systems are dialed down; on the other, the signaling and communication networks are revved up and reshaped. Neurons are among the body’s most energy‑hungry cells, especially at their synapses, so even modest, long‑lasting dips in ATP production could make it harder for them to sustain precise, fast communication. The authors argue that this chronic energy shortfall, combined with altered synaptic and trafficking proteins, may underlie the cognitive difficulties and other symptoms seen in schizophrenia. Their work points toward strategies that bolster mitochondrial function and restore healthy synaptic balance as promising directions for future therapies.
Citation: Koopmans, F., Dijkstra, A.A., Li, WP. et al. Human brain prefrontal cortex proteomics identifies compromised energy metabolism and neuronal function in Schizophrenia. Nat Commun 17, 2131 (2026). https://doi.org/10.1038/s41467-026-68950-y
Keywords: schizophrenia, prefrontal cortex, mitochondria, synaptic proteins, brain proteomics