Clear Sky Science · en
An allosteric inhibitor of the Zika virus NS2B-NS3 protease with oral efficacy in mouse models
A new way to outsmart a dangerous virus
Zika virus burst into global headlines a decade ago when infections in pregnant women were linked to babies born with abnormally small heads and brain damage. Yet today there is still no approved drug to treat or prevent Zika. This study describes a promising new pill-like compound that shuts down a key viral enzyme in an unexpected way and protects mice from severe Zika infection, pointing toward a future medicine that could one day safeguard vulnerable people, especially during outbreaks.
Why Zika still matters
Zika virus is spread mainly by Aedes mosquitoes and belongs to the same family as dengue and West Nile viruses. Most infected people feel little or no illness, but about one in five develop problems ranging from fever and rash to serious complications such as eye damage, organ failure, and nervous system disorders. The greatest danger is to fetuses: infection during pregnancy can lead to microcephaly and lifelong disability. Since there is no approved vaccine or antiviral treatment, doctors can only offer supportive care. Scientists therefore view Zika as a priority for pandemic preparedness, looking for drugs that could be used both to protect at-risk populations and to contain new outbreaks.
Hunting for hidden weak spots
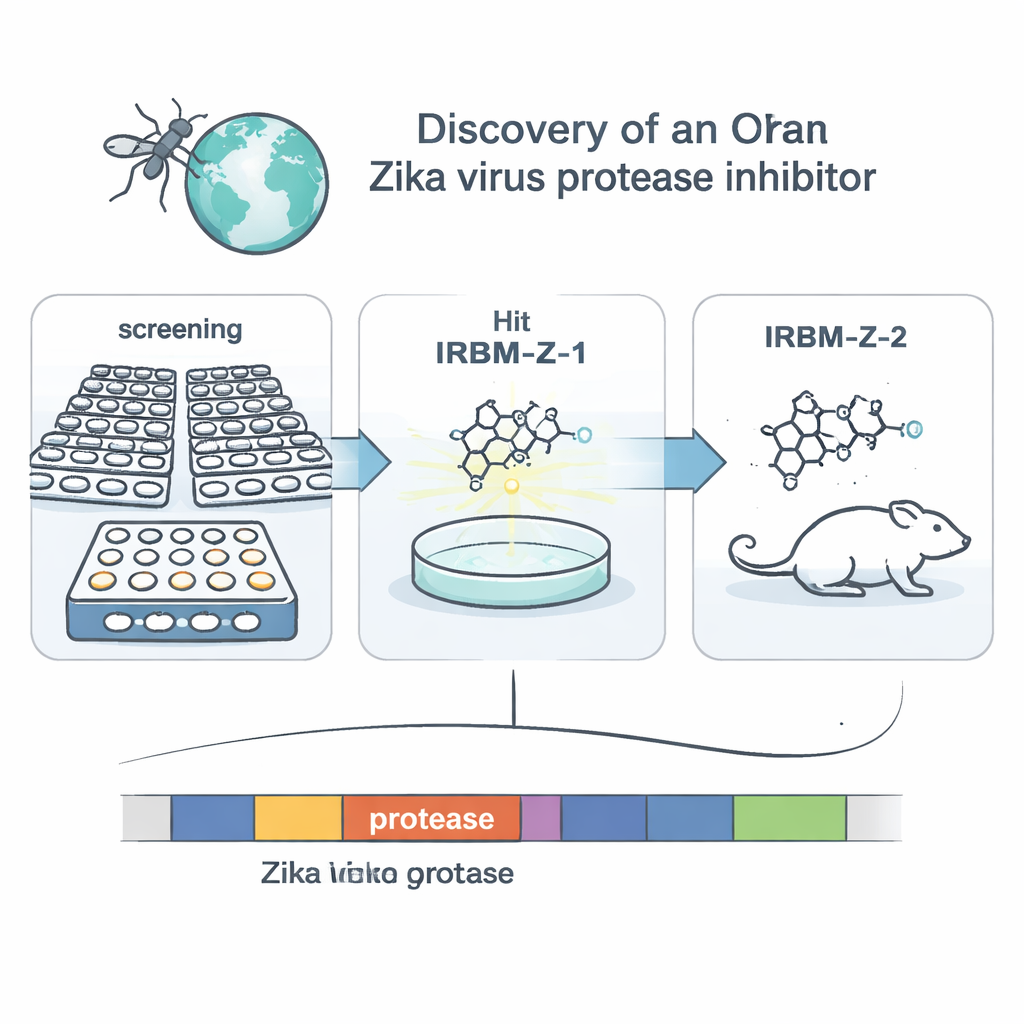
The researchers focused on a viral enzyme called NS2B–NS3 protease. This molecular “scissors” cuts the virus’s large starter protein into the pieces needed to build new viruses, making it an attractive drug target. Instead of designing a drug for the obvious cutting site, the team used a cell-based screening system that mimics Zika replication inside monkey kidney cells. They built a modified Zika genome, or replicon, that lacks structural genes but carries a luciferase reporter so that light output tracks viral copying. Screening more than 120,000 small molecules from a national chemical library, they selected those that strongly reduced the light signal without harming cells and then bred virus replicons that became resistant to each hit. This genetic sleuthing pointed to the protease as the key target for one family of compounds, led by an initial molecule called IRBM-Z-1.
Locking the enzyme in the wrong shape
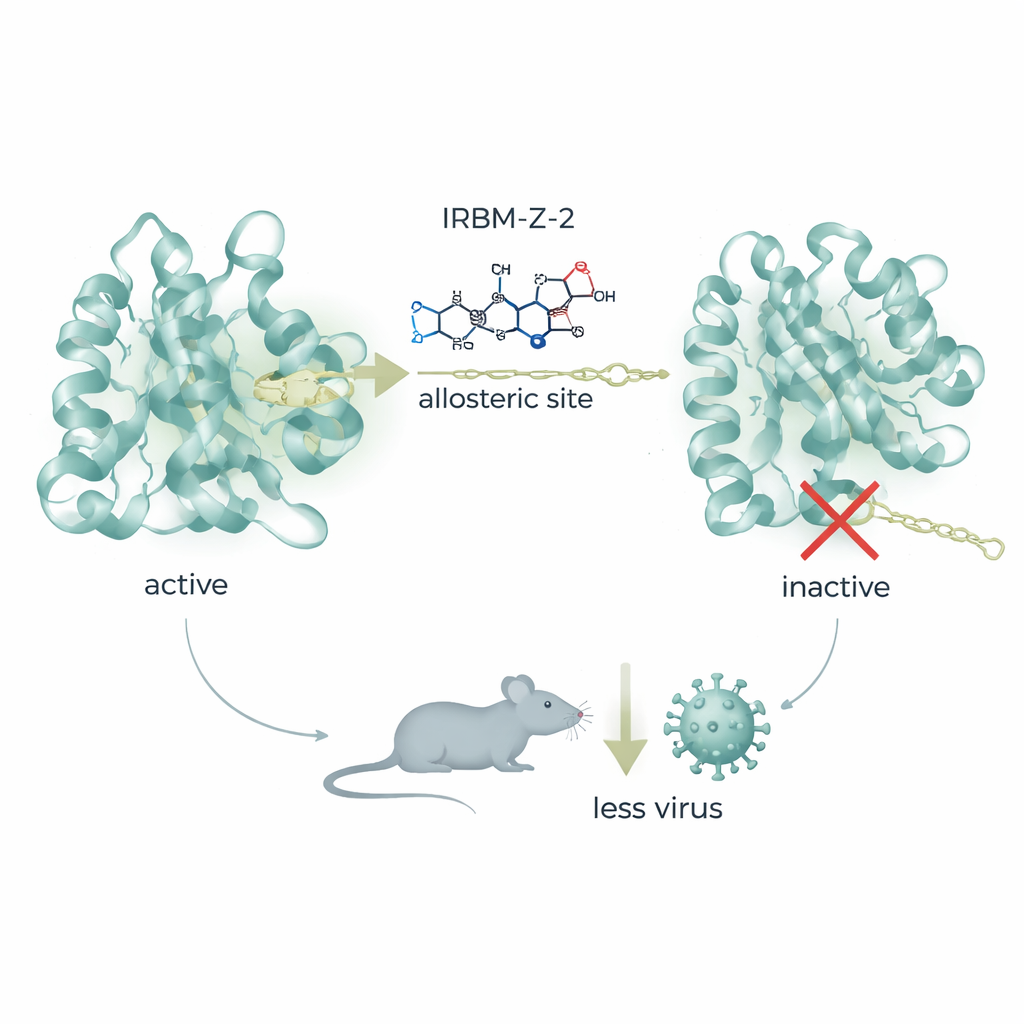
Biochemical tests showed that IRBM-Z-1 did not compete with the usual substrate at the cutting site. Instead, it behaved as a non-competitive, or allosteric, inhibitor—binding at a separate pocket and preventing the enzyme from adopting its active shape. High-resolution X‑ray crystallography revealed that the compound nestles into a previously unknown pocket far from the catalytic center. There, its unusual “N‑acylsydnone imine” core forms a web of hydrogen bonds and stacking interactions that push a key region of the protease into an open, inactive conformation. A single amino-acid change in this region, at position 156, makes the virus resistant, underscoring how precisely the compound exploits this hidden site. This pocket also differs from allosteric sites described in related flaviviruses, offering a genuinely new foothold for drug design.
From hit compound to protective drug in mice
Guided by structural insights, the team refined IRBM-Z-1 into a more potent molecule named IRBM-Z-2. This improved compound blocks the Zika protease at nanomolar concentrations, powerfully inhibits viral replication in several cell types, and shows activity against related dengue and West Nile proteases while sparing human enzymes. In animal studies, IRBM-Z-2 displayed favorable “drug-like” behavior: it was stable in the body, had good oral absorption, and maintained blood levels well above those needed to shut down the virus, without clear signs of toxicity. In highly susceptible AG129 mice infected with Zika, daily treatment—either by injection or by mouth—dramatically lowered the amount of viral RNA in the blood, prevented weight loss and disease symptoms, and allowed all treated animals to survive, while untreated controls succumbed to infection.
What this could mean for future outbreaks
Together, these findings introduce a novel class of Zika antivirals that work by freezing a critical viral enzyme in an inactive pose rather than blocking its active site directly. Because IRBM-Z-2 can be given orally, shows strong protection in stringent mouse models, and has a clean safety profile so far, it stands out as a promising candidate for further development as a preventive drug—potentially to shield pregnant women, healthcare workers, and travelers if Zika resurges. More broadly, the newly discovered allosteric pocket may inspire similar drugs against related viruses, opening a fresh front in the fight against mosquito-borne diseases.
Citation: Ontoria, J.M., Torrente, E., Missineo, A. et al. An allosteric inhibitor of the Zika virus NS2B-NS3 protease with oral efficacy in mouse models. Nat Commun 17, 1439 (2026). https://doi.org/10.1038/s41467-026-68943-x
Keywords: Zika virus, antiviral drug, protease inhibitor, allosteric modulation, mosquito-borne disease