Clear Sky Science · en
FCHo2, not talin, enables inside-out activation of integrin ɑvβ5 in curved adhesions
How Cells Feel the Shape of Their Surroundings
Our bodies are built from cells that constantly grab onto their surroundings, pulling and pushing to move, divide, or stay put. This paper reveals that cells do not just sense how stiff their environment is—they also read its shape. By discovering a special adhesion system that switches on only where the cell membrane is tightly curved, the authors show that cells use different internal "grips" for flat versus fibrous surroundings, with important implications for how tissues form and how cancers spread.
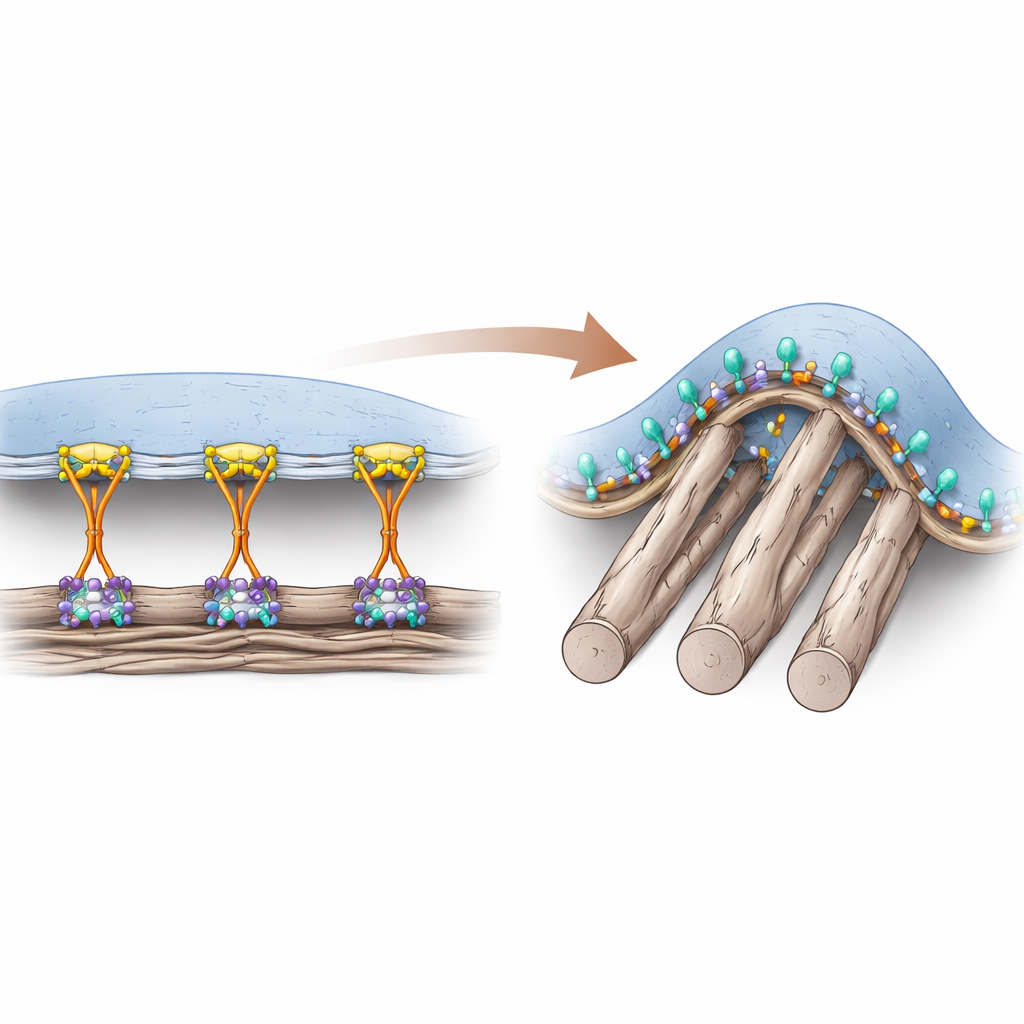
Two Ways Cells Hold On
Cells attach to the protein-rich material around them using surface receptors called integrins. For years, researchers thought a helper protein named talin was absolutely essential for turning integrins "on" from the inside of the cell, enabling strong attachment in classic anchoring sites known as focal adhesions. These adhesions form mainly on flat, rigid surfaces and transmit large pulling forces through the cell skeleton. However, in living tissues, many of the surrounding fibers are soft and cylindrical, creating curved contact points that do not easily support these force-heavy structures. The authors previously found a different type of adhesive structure, called a curved adhesion, that appears right where the membrane bends around thin fibers and that relies on a specific integrin, αvβ5.
Curved Grips Use a Different Helper
In the new study, the team shows that talin, while still present, is not the key activator of integrin αvβ5 in curved adhesions. Instead, a curvature-sensing protein called FCHo2 takes over this job. Using precisely engineered nano-sized bars and pillars to bend cell membranes in controlled ways, they observed that αvβ5 clusters strongly at curved regions, but only when FCHo2 can bind to a very short sequence—an HDRRE motif—just inside the cell on the β5 tail. When this motif is disrupted, integrin still works in focal adhesions but can no longer form curved adhesions. Knocking down FCHo2 leaves the classic focal adhesions largely intact yet wipes out curved adhesions, whereas removing talin severely weakens focal adhesions but leaves curved adhesions mostly unaffected.
A Single Amino-Acid Switch as a Shape Selector
One puzzle was why a closely related integrin, αvβ3, cannot form curved adhesions even though its inner tail looks very similar to that of β5. By swapping domains between the two integrins and introducing point mutations, the authors identified a single pivotal position near the talin-binding site: most β integrins have a tryptophan (W), but β5 uniquely has a tyrosine (Y766). When β5’s tyrosine was replaced with tryptophan, the integrin lost its ability to participate in curved adhesions and behaved more like β3, favoring only focal adhesions. The reverse change—putting tyrosine into the β3-like chimera—restored curvature sensitivity. Further experiments with variants that mimic phosphorylated or non-phosphorylated states of this tyrosine suggest that its chemical modification can bias the integrin toward either flat, high-force focal adhesions or curved, low-force adhesions.
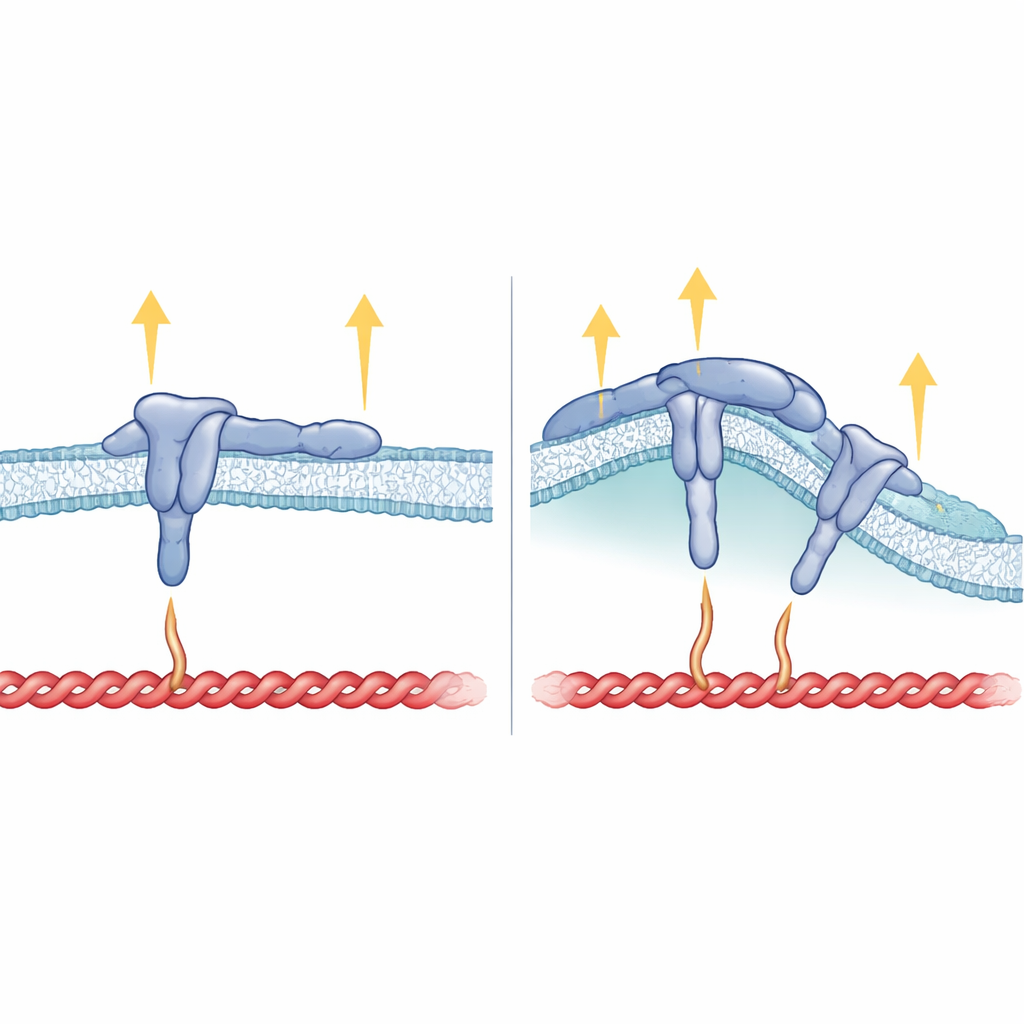
How Membrane Shape, Integrin Structure, and Force Fit Together
Beyond the inner tail, the stretch of β5 that spans the membrane also proved critical. When the researchers replaced β5’s transmembrane segment with that of β3, the resulting integrin could still form focal adhesions but no longer preferred curved sites or tracked with FCHo2. This suggests that the way the integrin sits in the membrane helps orient the HDRRE motif so that FCHo2 can latch on at curved regions and stabilize ligand-bound, active αvβ5. In curved adhesions, talin appears to touch only a more distant part of the integrin tail and does not bear high tension, which explains why other force-hungry components such as vinculin, tensin, and kindlin are largely absent there. In contrast, in focal adhesions, talin grips both near and far sites on the tail and the membrane, supporting strong pulling forces but blocking FCHo2 from accessing the same region.
What This Means for Cell Behavior
To a lay observer, the message of this work is that cells carry two separate "gripping modes" for sticking to their surroundings. On flat, rigid areas they use talin-driven focal adhesions that are built to pull hard. On soft, fibrous, and curved structures they switch to FCHo2-driven curved adhesions that stabilize integrin αvβ5 with much lower force. A tiny change in the integrin’s sequence—and whether that site is chemically modified—helps decide which mode is used. This dual system lets cells interpret both stiffness and shape in three-dimensional environments, influencing how they migrate, organize tissues, and potentially how they invade during disease.
Citation: Lu, CH., Lee, C.E., Zhang, W. et al. FCHo2, not talin, enables inside-out activation of integrin ɑvβ5 in curved adhesions. Nat Commun 17, 2419 (2026). https://doi.org/10.1038/s41467-026-68942-y
Keywords: integrins, cell adhesion, membrane curvature, FCHo2, cytoskeleton