Clear Sky Science · en
Rochelle salt-based biodegradable piezoelectric devices for nerve regeneration and intestinal motility monitoring
Melting Implants That Talk to the Body
Doctors increasingly rely on tiny electronic implants to help damaged nerves regrow and to watch how our organs move. But most of today’s devices are made from hard, permanent materials that can irritate soft tissue and must be removed in a second surgery. This study introduces a new class of “vanishing” implants built from a common food additive and a medical plastic. These soft devices turn natural body motion or gentle ultrasound into electricity that can coach injured nerves to heal and quietly track how the gut squeezes food along, then safely dissolve when their job is done.
Building Electricity into Soft, Disappearing Films
The heart of the work is a flexible material that generates voltage when it is pressed or bent, a behavior known as the piezoelectric effect. The researchers start with Rochelle salt, a century-old crystal once used in microphones and now approved as a food ingredient. Rochelle salt is strongly responsive to mechanical forces but is brittle and dissolves in water. To tame it, the team grinds the crystals into microscopic particles and spins them together with strands of poly(L-lactic acid), a biodegradable plastic already used in medical sutures. By carefully electrospinning the mix into highly aligned nanofibers and then compressing the mat, they create centimeter-scale films in which the crystals are locked inside a soft, skin-like scaffold. These films bend easily yet produce electrical signals far stronger than previous biodegradable options.
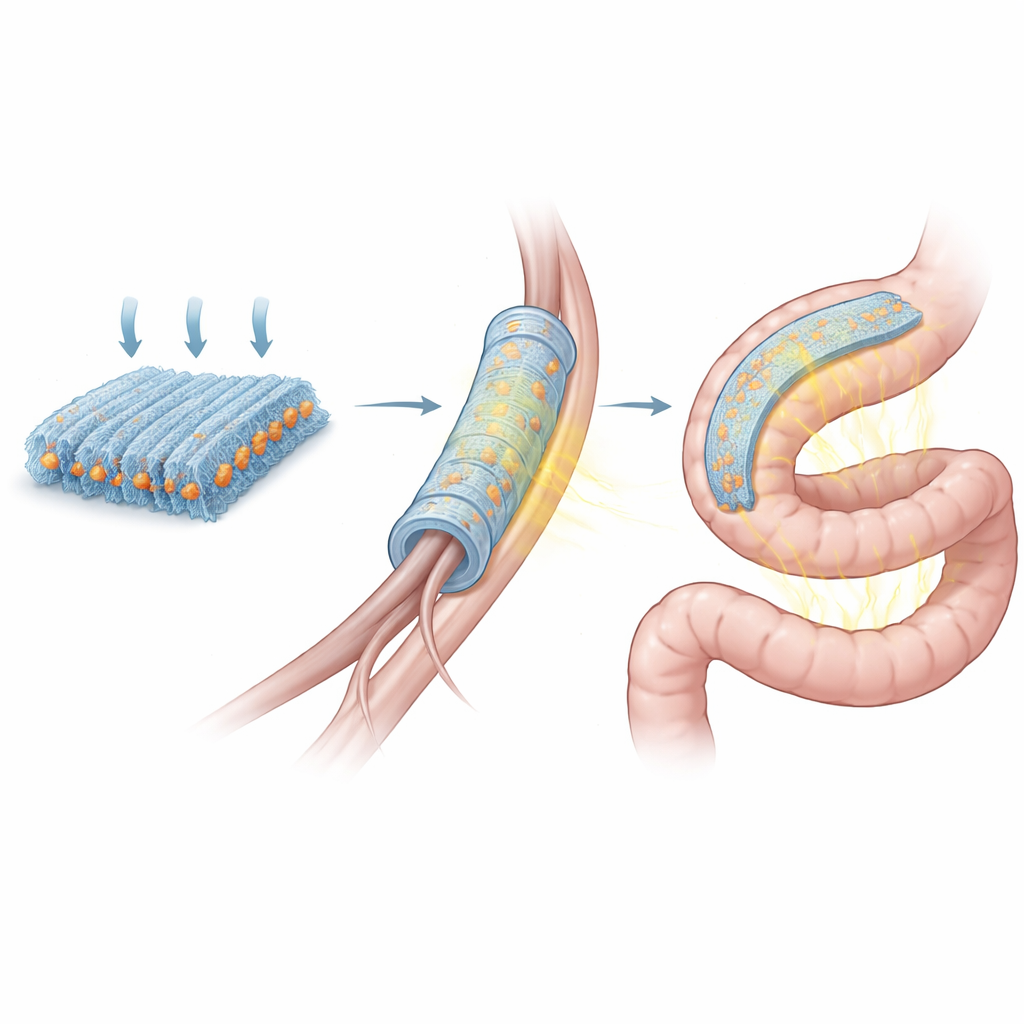
Why Strong Signals Matter for Living Tissues
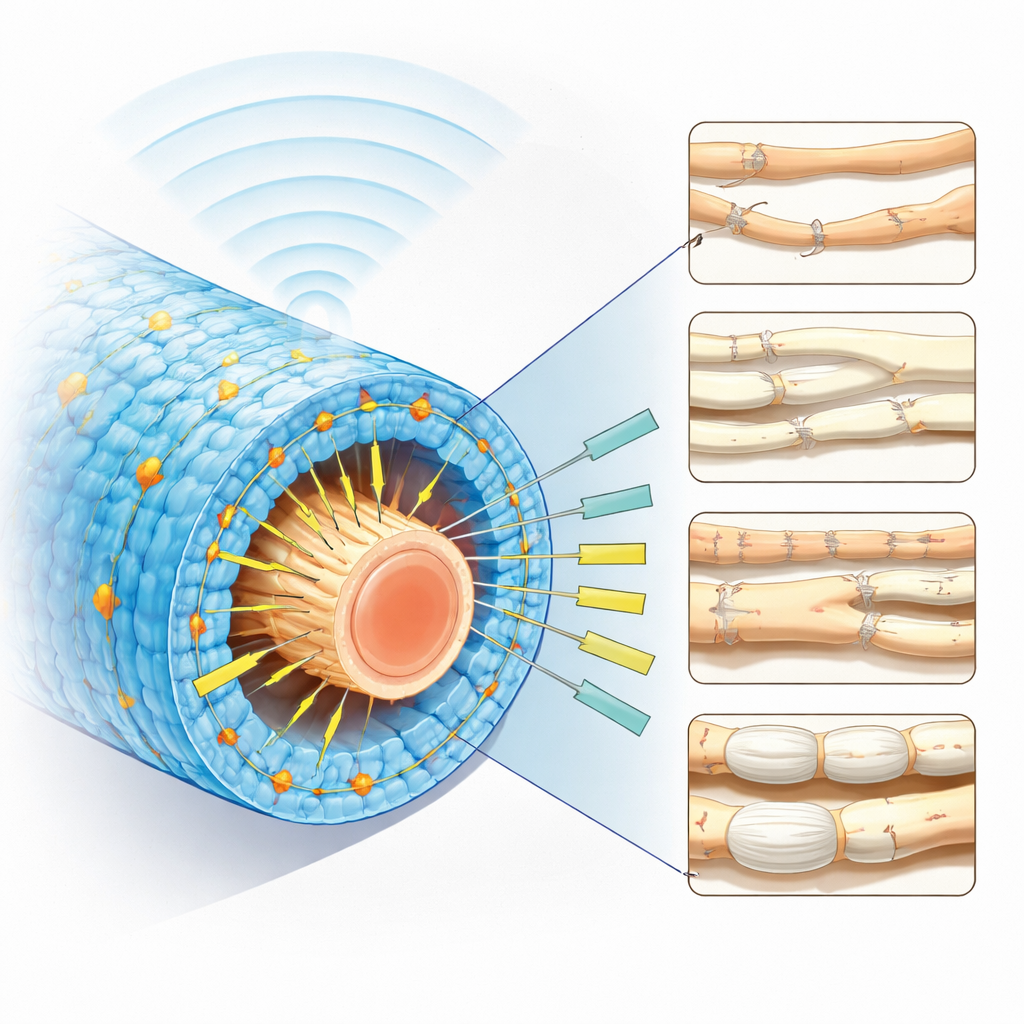
For an implant to influence cells without wires or batteries, it must turn faint mechanical cues into useful bursts of electricity. Tests show that the new films generate more than ten times the charge of the plastic alone and even outperform many non-degradable piezoelectric materials in voltage output. The films keep working for days to weeks in warm salty water, mimicking the body, and their lifetime can be tuned with protective coatings. When driven by ultrasound—sound waves at frequencies used in medical imaging—the material converts deep-tissue vibrations into small but repeatable voltage spikes. Because the films are soft, with stiffness closer to that of nerves and muscle than of ceramics, they can conform to moving organs without scraping or cutting.
Coaching Damaged Nerves with Gentle Ultrasound
To turn the material into a therapy tool, the team rolls the film into a tube and adds an outer support layer, forming a hollow scaffold that can bridge a gap in a severed sciatic nerve in rats. From outside the body, a focused ultrasound probe periodically bathes the implanted scaffold in pulses of energy. Inside, the piezoelectric wall flexes and produces electric fields that bathe regenerating nerve fibers. Cell studies show that this stimulation boosts the length of growing nerve branches and increases activity in genes tied to repair. In animals with a 10-millimeter nerve injury, the ultrasound-activated scaffold leads to longer regrown fibers, thicker insulating myelin, stronger muscle contractions, and better walking patterns than control scaffolds, approaching the performance of today’s gold-standard nerve graft taken from the animal’s own body.
Listening to the Gut without Wires
In a second application, the films act as highly sensitive strain sensors that track how the colon contracts. The researchers sandwich a strip of the piezoelectric composite between dissolvable metal electrodes and soft plastic layers, then attach the device to the outside of a rabbit’s colon using a biodegradable glue. Each time the gut wall tightens or relaxes, the sensor bends and produces a distinct voltage waveform, which is relayed wirelessly to an external receiver. By processing these signals, the team can extract the strength, rhythm, and travel speed of the muscular waves that move contents along. After giving a drug that speeds up motility, the sensor records more vigorous and complex contractions; after cutting off blood flow to mimic a dangerous intestinal emergency, it captures an initial surge followed by a sharp collapse in activity—early warning signs that are hard to catch with current tools.
A Glimpse of Future Vanishing Bioelectronics
Taken together, the work shows that a simple blend of food-grade crystals and a medical plastic can serve as a powerful, temporary bridge between mechanical motion and electrical cues inside the body. These soft, biodegradable devices can both stimulate healing—by nudging injured nerves with precisely timed electrical pulses—and provide rich, real-time readouts of organ function, such as how smoothly the colon moves. After their useful life, the components gradually break down into benign products, eliminating the need for surgical removal. The study points toward a future in which implantable electronics become more like dissolvable stitches: smart helpers that guide recovery, report back on hidden problems, and quietly disappear when they are no longer needed.
Citation: Dai, F., Cheng, H., Qi, H. et al. Rochelle salt-based biodegradable piezoelectric devices for nerve regeneration and intestinal motility monitoring. Nat Commun 17, 2169 (2026). https://doi.org/10.1038/s41467-026-68930-2
Keywords: biodegradable electronics, nerve regeneration, ultrasound stimulation, intestinal motility, piezoelectric materials