Clear Sky Science · en
Nitroreductase-triggered indazole formation
Turning Simple Ingredients into Valuable Medicines
Many modern medicines are built on a handful of popular molecular “frames” that fit well into the body’s targets. One of these frames, called an indazole, appears in drugs used against cancer and severe nausea. Until now, making indazoles has usually required harsh chemicals, high temperatures, and metals that are not environmentally friendly. This study shows how scientists can coax natural enzymes to build indazoles gently in water, opening the door to greener ways of producing important pharmaceuticals.
Why These Ring-Shaped Molecules Matter
Drug designers often reuse certain ring-shaped structures because they so reliably interact with proteins in the body. Indazoles are one such favored structure and are found in approved medicines for cancer and chemotherapy-induced nausea. Chemists already know several ways to make indazoles, but the standard routes rely on reactive building blocks, strong bases or acids, and metal catalysts such as copper and palladium. These methods work, yet they create waste, demand energy, and pose safety risks, which clashes with the growing push for sustainable manufacturing in the pharmaceutical industry.
Enzymes as Gentle Construction Workers
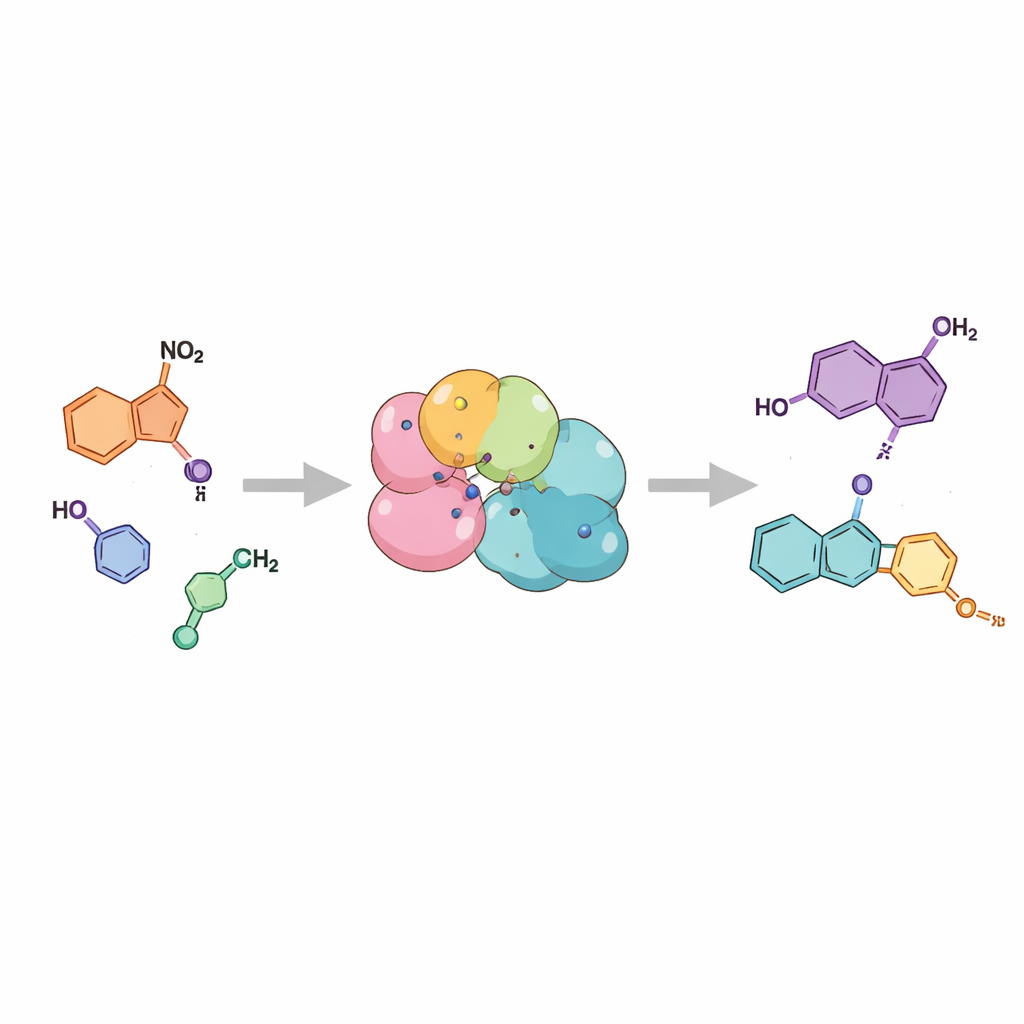
Instead of forcing atoms together with heat and strong reagents, the authors turn to biocatalysis—the use of enzymes, nature’s own catalysts, to run reactions in water at room temperature. They focus on enzymes called nitroreductases, which normally help cells detoxify nitro-containing compounds by stepwise reduction. The team reasoned that if one particular intermediate in this reduction pathway could be released at just the right moment, it would fold onto itself and form the characteristic indazole ring system on its own. Using two different nitroreductases, they demonstrate that simple 2-nitrobenzylamine starting materials are smoothly converted into indazoles with excellent efficiency, often above 99% conversion, under mild, aqueous conditions.
Mapping What the Enzymes Can Do
To test how generally this strategy works, the researchers fed the enzymes a family of related starting materials bearing different side groups and positions on the aromatic ring. They found that both enzymes tolerated a broad range of chemical decorations, from small groups like methyl to bulkier ring systems and various chlorine, bromine, and methoxy substitutions. In many cases the reactions proceeded cleanly to the desired indazole, with isolated yields high enough to be practical. The way the starting material was decorated also controlled which of two closely related indazole forms appeared, a feature that is important because both forms can matter in drug design. The same concept could even be stretched to make a larger cousin of indazoles, called cinnolines, highlighting the versatility of the approach.
Building a Two-Step Enzymatic Assembly Line
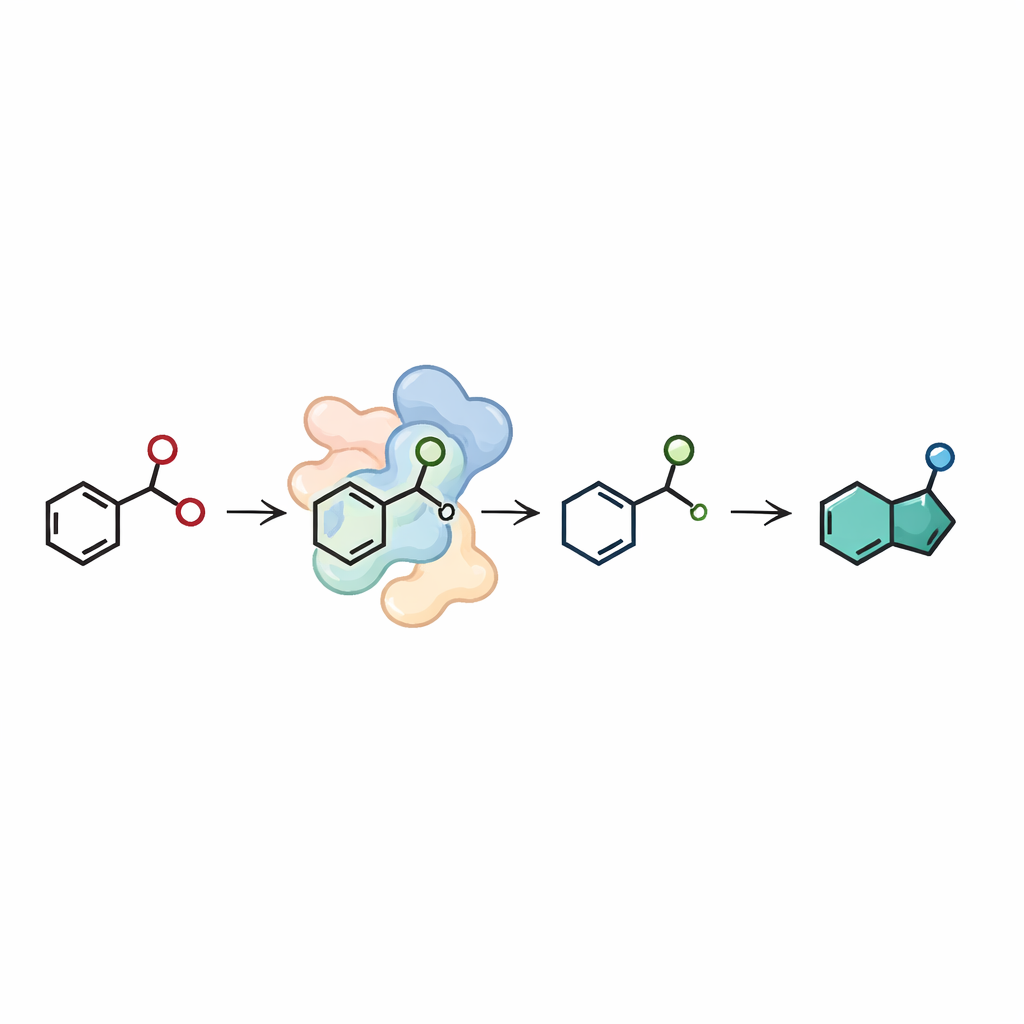
Indazoles in this work do not come directly from specialty starting materials; instead, the team constructed a short enzymatic assembly line beginning from inexpensive building blocks. In the first step, a different class of enzyme, an imine reductase, joins a simple aldehyde and an amine to make the needed 2-nitrobenzylamine. In the second step, the nitroreductase converts this intermediate into the indazole ring. By carefully tuning the amounts of enzymes and the system that recycles their cofactors, the authors ran this two-enzyme cascade on a preparative scale, starting from 50 milligrams of aldehyde in water. They obtained several indazole products in good overall yields, including examples with different side chains that could later be modified further for drug discovery.
A Cleaner Route to Complex Drug Building Blocks
For a lay reader, the key message is that the researchers have taught natural catalysts to assemble a medically important ring system in a way that is gentler on both workers and the environment. Their enzyme-based route avoids hazardous nitrogen–nitrogen reagents and precious metal catalysts, operates in water at moderate temperatures, and can be combined with other enzymes into an efficient mini-factory. This work adds a powerful new tool to the chemist’s toolbox, bringing sustainable manufacturing a step closer while supplying the pharmaceutical world with versatile indazole building blocks for future medicines.
Citation: Terholsen, H., Medema, L., Chernyshova, E. et al. Nitroreductase-triggered indazole formation. Nat Commun 17, 2261 (2026). https://doi.org/10.1038/s41467-026-68926-y
Keywords: biocatalysis, indazole synthesis, nitroreductase, green chemistry, enzyme cascades