Clear Sky Science · en
Clonal expansion of cytotoxic CD8⁺ T cells in lecanemab-associated ARIA
Why this matters for people with Alzheimer’s
Lecanemab is one of the first drugs that can clear amyloid from the brain and modestly slow memory loss in Alzheimer’s disease. But some patients on this therapy develop brain swelling or tiny brain bleeds, changes seen on MRI scans and grouped under the term amyloid‑related imaging abnormalities (ARIA). This study asks a crucial question for patients and families: what is happening in the immune system when ARIA appears, and could a simple blood test one day help predict who is most at risk?
The promise and risk of a new Alzheimer’s treatment
Lecanemab is an antibody given by infusion that targets clumps of a protein called amyloid‑beta, a hallmark of Alzheimer’s disease. By helping the body clear amyloid, it can modestly slow cognitive decline. Yet this benefit comes with a safety concern: some people develop ARIA, which can include areas of brain swelling or pinpoint hemorrhages. These side effects require frequent MRI monitoring and can limit who receives the drug. Not everyone is equally vulnerable—carriers of a gene variant called APOE4 are at higher risk—but genes alone do not explain why ARIA happens, especially early after treatment begins. Because lecanemab is delivered through the bloodstream, the authors reasoned that looking closely at immune cells in blood might reveal early warning signs or even mechanisms that link treatment to vascular injury in the brain. 
Tracking immune cells in patients who did and did not develop ARIA
The researchers studied six people with Alzheimer’s receiving lecanemab at a regional memory clinic. Three developed different forms of ARIA, and three matched patients (similar in age, sex, APOE genotype, and number of infusions) did not. Blood was drawn just before infusions, and the team used an intensive "multi‑omic" approach: single‑cell RNA sequencing to read which genes were switched on in thousands of individual immune cells, antibody‑based tags to define cell types, sequencing of T‑cell receptors to track clones, and targeted metabolomics to measure hundreds of small molecules involved in cell metabolism. This allowed them to ask not only which cell types were present, but how active they were, what fuel they were using, and whether specific T‑cell families were expanding in response to some trigger.
Expansion of highly armed killer T cells
A clear pattern emerged in the patients who developed ARIA. Compared with controls, they had a higher proportion of CD8 "killer" T cells and fewer CD4 helper T cells. Within the CD8 pool, two subgroups stood out: effector memory cells and a terminally differentiated form known as TEMRA cells. These TEMRA cells are like veteran soldiers—highly specialized for attack, with strong machinery for killing target cells. In ARIA‑positive patients, TEMRA cells were more numerous, more clonally expanded (meaning particular T‑cell families had multiplied), and carried gene programs associated with cytotoxicity, migration through blood vessels, and signs of chronic stimulation. Similar T‑cell states have been observed in aging, chronic infections, multiple sclerosis, and in the spinal fluid of people with Alzheimer’s, where they can accumulate near blood vessels and the brain’s surface.
Metabolic rewiring and cross‑talk with other immune cells
The team also found that these CD8 effector cells had rewired their metabolism. Instead of relying mainly on efficient energy production in mitochondria, they shifted toward glycolysis, a faster but less efficient way to generate energy that supports short‑lived, high‑intensity activity. Metabolite measurements in blood cells backed this up: levels of lactate and pyruvate were increased, while key components of the citric acid cycle were reduced, a pattern typical of inflammatory immune cells. Monocytes, another type of white blood cell, showed complementary changes. In ARIA‑positive patients, monocytes turned on genes for presenting antigens, sticking to other cells, and producing chemokines—chemical signals that attract T cells. Computational modeling of ligand‑receptor pairs suggested that monocytes were sending stronger activating and homing signals to CD8 effector cells, creating an environment that encourages these killer cells to engage blood vessel walls.
Address codes that point T cells toward brain vessels
Because this study only sampled blood, the authors turned to an independent single‑cell dataset from brains of lecanemab‑treated patients to ask whether the same T‑cell programs are linked to vascular niches in the brain. When they projected their CD8 subsets onto this brain map, the TEMRA and effector memory cells from ARIA‑positive patients showed transcriptional "address codes"—combinations of adhesion molecules and chemokine receptors—that matched endothelial (blood vessel) cells in the diseased brain. These signatures suggest that the expanded killer T cells are not only highly armed and metabolically revved up, but also primed to home to and interact with stressed brain blood vessels at sites of amyloid clearance. 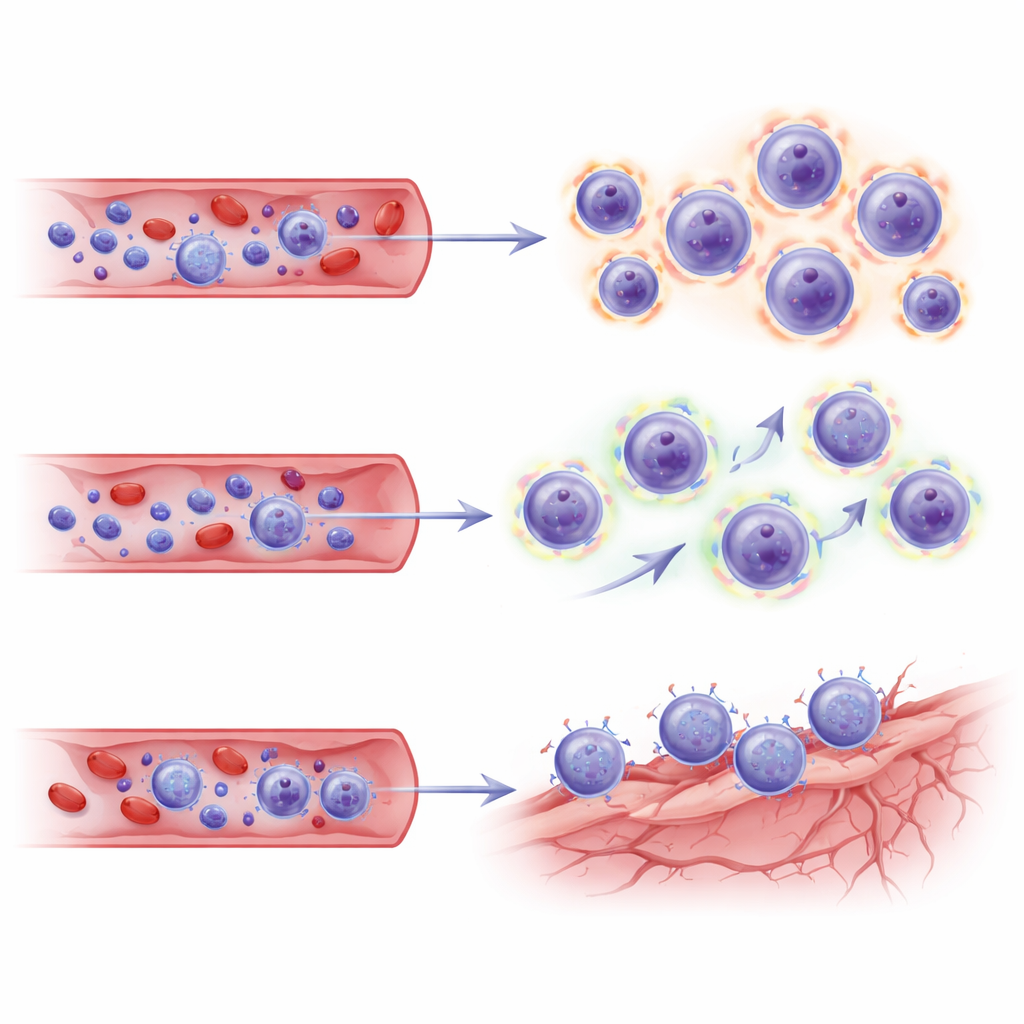
What this could mean for patients and future care
Taken together, this small but detailed study paints ARIA as more than a passive side effect of amyloid removal. Instead, it appears linked to a coordinated immune response in the blood: expansion and metabolic activation of specific killer T‑cell clones, strengthened signaling from monocytes, and molecular features that favor engagement with brain blood vessels. The work does not prove that these cells cause ARIA—immune changes could be a reaction to vascular injury rather than its source—but it provides testable hypotheses and a blueprint for blood‑based biomarkers. In the future, measuring the abundance and metabolic state of CD8 TEMRA cells, along with key signaling molecules, might help identify patients at highest risk for ARIA, guide monitoring schedules, or even inspire targeted strategies to temporarily dial down this immune response while preserving the benefits of amyloid‑clearing therapies.
Citation: Johnson, L.A., Saito, K., Pallerla, A.V. et al. Clonal expansion of cytotoxic CD8⁺ T cells in lecanemab-associated ARIA. Nat Commun 17, 2180 (2026). https://doi.org/10.1038/s41467-026-68921-3
Keywords: Alzheimer’s disease, lecanemab, immune cells, brain blood vessels, treatment side effects