Clear Sky Science · en
TMEM63 proteins act as mechanically activated cholesterol modulated lipid scramblases contributing to membrane mechano-resilience
How Cells Stay Intact Under Physical Stress
Our bodies’ cells are constantly squeezed, stretched and jostled, whether in beating hearts, working muscles or tumors pushing through tissue. This study uncovers how a family of membrane proteins, called OSCA/TMEM63, help cells survive such harsh mechanical forces by rearranging the fats in their outer shell. Understanding this built-in shock absorber system could reveal new angles on disorders of hearing, nerve insulation and even cancer spread.
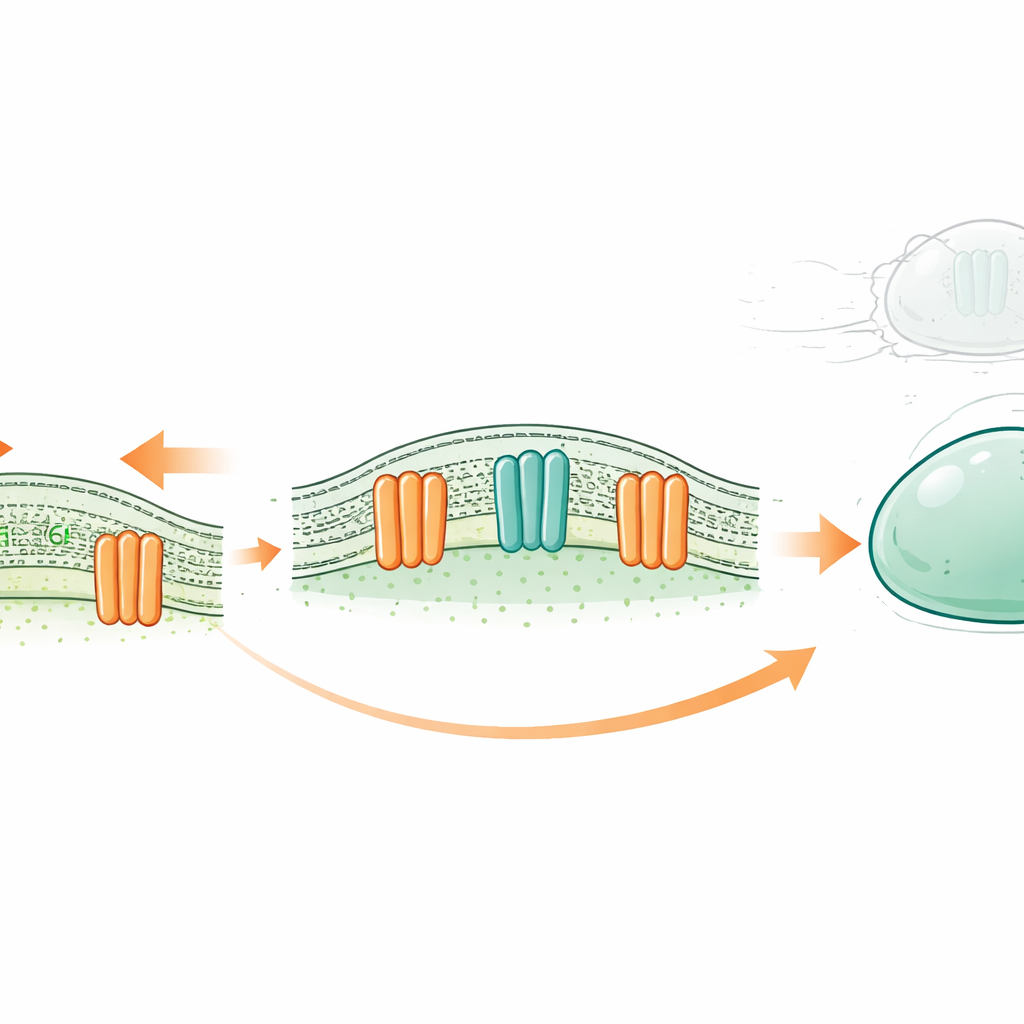
A New Kind of Cellular Shock Absorber
Every cell is wrapped in a thin oily membrane made of two layers of fat-like molecules called lipids. Traditionally, certain membrane proteins were thought to act only as ion channels—tiny valves that let charged particles flow in and out in response to stimuli such as stretch. The OSCA/TMEM63 family, found from plants to humans, is known to open in response to membrane tension. Earlier structural work hinted that, unlike classic ion channels, their pores are lined partly by lipids themselves. This raised a provocative possibility: could these channels also act as “scramblases,” flipping lipids between the inner and outer layers of the membrane when the cell is under force?
Watching Lipids Flip Across the Membrane
To test this idea, the authors combined computer simulations with reductionist lab systems built from purified components. In simulations, open versions of several OSCA and TMEM63 proteins were embedded in model membranes. Lipid molecules spontaneously moved through a groove in these proteins from one side of the bilayer to the other, forming a belt that linked the two leaflets while leaving a water-filled path for ions. In parallel experiments, the team built giant artificial “bubbles” (vesicles) containing fluorescent lipids and specific proteins. When an external agent quenched fluorescence on the outer surface, any further drop in signal could only be explained by lipids flipping from the inner layer to the outside. Vesicles containing OSCA1.1, OSCA1.2, OSCA2.2 or TMEM63A/B showed this extra loss of fluorescence, indicating active scrambling, whereas control channels that do not scramble lipids did not.
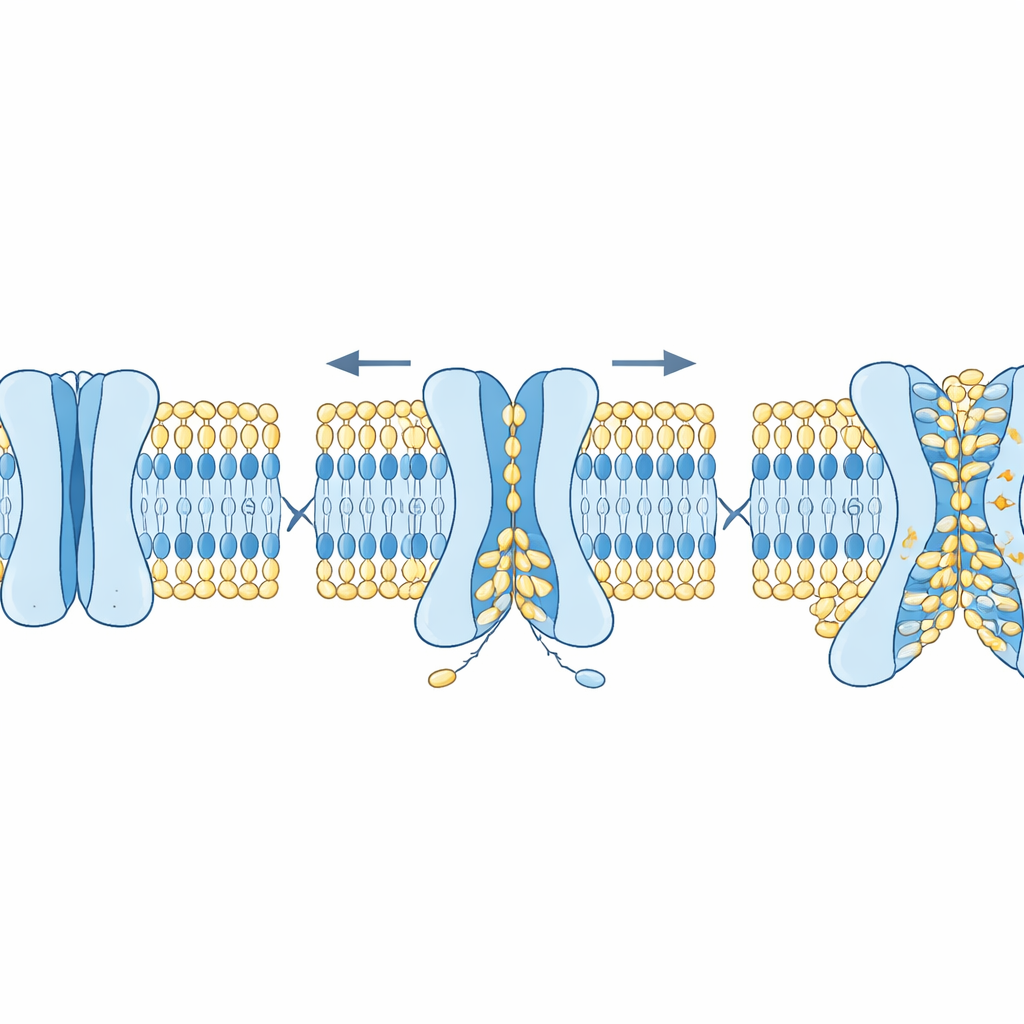
How Structure and Cholesterol Tune the Process
The researchers then asked what features of TMEM63 proteins control this dual behavior. Using AlphaFold2, they generated many plausible shapes of human TMEM63 proteins, ranging from closed to increasingly open. Simulations revealed that a handful of bulky amino acids act as “bottlenecks” in the groove. One residue mainly limits ion flow, while others block lipid movement. Mutating the ion bottleneck increased electrical conductance without much changing lipid scrambling, whereas mutating the lipid gate caused lipids to flip more readily and even made cells display “eat me” signals on their surface without any added stimulus. Another key regulator was cholesterol, a stiffening component of cell membranes. Adding cholesterol to model membranes markedly slowed lipid scrambling and stabilized the closed state of TMEM63A, both in simulations and in cryo–electron microscopy structures where cholesterol nestled into specific sites on the protein.
Mechanical Force as the On-Switch
If cholesterol normally keeps scrambling in check inside cells, what turns it on at the right moment? The team used ring-shaped sugar molecules called cyclodextrins to selectively pull lipids from membranes and thus increase tension without removing cholesterol. In cell-sized vesicles, adding cyclodextrin activated lipid scrambling by TMEM63A only when the protein was present, proving that mechanical stress alone can trigger the flipping. Similar approaches in living cells showed that mild cholesterol removal by itself was not enough; a subsequent mechanical challenge was required to see strong scrambling. When cells engineered to express TMEM63A were stretched repeatedly, they rapidly exposed certain lipids on their outer surface, a hallmark of scrambling, while cells lacking these proteins showed far less change.
Protecting Cells from Rupture
Beyond flipping lipids, mechanically activated scrambling changed the physical behavior of membranes under extreme stress. In artificial vesicles exposed to high force, those lacking scramblases tended to burst, whereas vesicles containing TMEM63A or a known scramblase shrank, formed thin tubules and largely remained intact. In a human brain cancer cell line that naturally expresses TMEM63A and TMEM63B, reducing the levels of either protein made cells more likely to rupture when challenged with strong mechanical stress. Together, these findings support a model in which OSCA/TMEM63 proteins act as valves that open under tension and temporarily blur the usual difference between the inner and outer faces of the membrane. By allowing lipids to rapidly redistribute, they help equalize stress and prevent catastrophic tearing.
What This Means for Health and Disease
In straightforward terms, this work shows that some stretch-sensitive channels do more than conduct electrical signals: they also reorganize the cell’s outer skin to help it survive being squeezed. TMEM63 proteins emerge as mechanically activated lipid scramblases whose activity is finely tuned by membrane composition, especially cholesterol. This mechano-resilience mechanism may be important wherever cells are exposed to large forces, from insulating nerve fibers and sound-sensing cells in the ear to cancer cells navigating dense tissues. Understanding and eventually manipulating this dual ion channel–scramblase function could open new paths for protecting vulnerable tissues—or, conversely, for making hard-to-kill cancer cells more fragile.
Citation: Lin, Y., Zhou, Z., Han, Y. et al. TMEM63 proteins act as mechanically activated cholesterol modulated lipid scramblases contributing to membrane mechano-resilience. Nat Commun 17, 2717 (2026). https://doi.org/10.1038/s41467-026-68919-x
Keywords: mechanosensitive ion channels, lipid scrambling, cell membrane mechanics, cholesterol regulation, TMEM63 OSCA proteins