Clear Sky Science · en
Dynamic stretching beyond electron transfer in a homointerpenetrated metal‒organic framework for enhanced Fenton-like reactions
Why bendable catalysts matter for clean water
Many of today’s medicines and chemicals slip through conventional wastewater treatment and end up in rivers and drinking water. The study described here explores a new kind of solid catalyst whose internal framework can flex and stretch as it works. This “springy” material dramatically speeds up the breakdown of stubborn pollutants, pointing toward more efficient and safer water purification technologies.
A sponge-like solid built to move
The researchers created a metal–organic framework, or MOF, named BUC-95. MOFs are crystalline materials made of metal atoms connected by organic linkers, forming porous, sponge-like networks. BUC-95 is special because it contains two identical, interwoven networks that are not rigidly locked together. Instead, these intertwined frameworks can shift slightly relative to each other, giving the material a built-in ability to stretch and relax as the environment changes. Microscopy and diffraction techniques confirmed this interpenetrated architecture and showed that the iron atoms sit in similar local environments to those in a closely related, but more rigid, material called BUC-96.
Turning a common oxidant into a powerful cleaner
To test how well BUC-95 cleans water, the team focused on a widely used disinfectant and oxidant called peroxydisulfate. On its own, this chemical is sluggish, but when “activated” by a catalyst, it can generate short-lived, highly reactive species that attack pollutants. Using ofloxacin, a common antibiotic, as a test contaminant, BUC-95 combined with peroxydisulfate removed more than 99.99% of the drug in just 10 minutes—far faster than traditional iron salts and faster than a range of other iron-based MOFs. The same system also rapidly degraded several other pharmaceuticals, showing broad effectiveness and good stability over many cycles, with only trace amounts of iron leaching into the water.
A different kind of oxidizing power
Most advanced oxidation processes rely on free radicals, such as hydroxyl and sulfate radicals, which are extremely reactive but nonselective. By adding various “scavenger” molecules that selectively quench these radicals, and by using spin-resonance probes, the researchers showed that these species play only a minor role in BUC-95’s performance. Instead, the dominant player is a high-valent iron–oxo species, essentially an iron center double-bonded to oxygen. This species behaves as a strong yet more targeted oxidant, preferring pollutants with electron-rich regions—like many antibiotics and anti-inflammatory drugs—while reacting less with more resistant compounds. Calculations and spectroscopic measurements revealed that surface hydroxyl groups and the flexible framework help iron reach this powerful state more easily by lowering the energy barrier to form the iron–oxo unit.
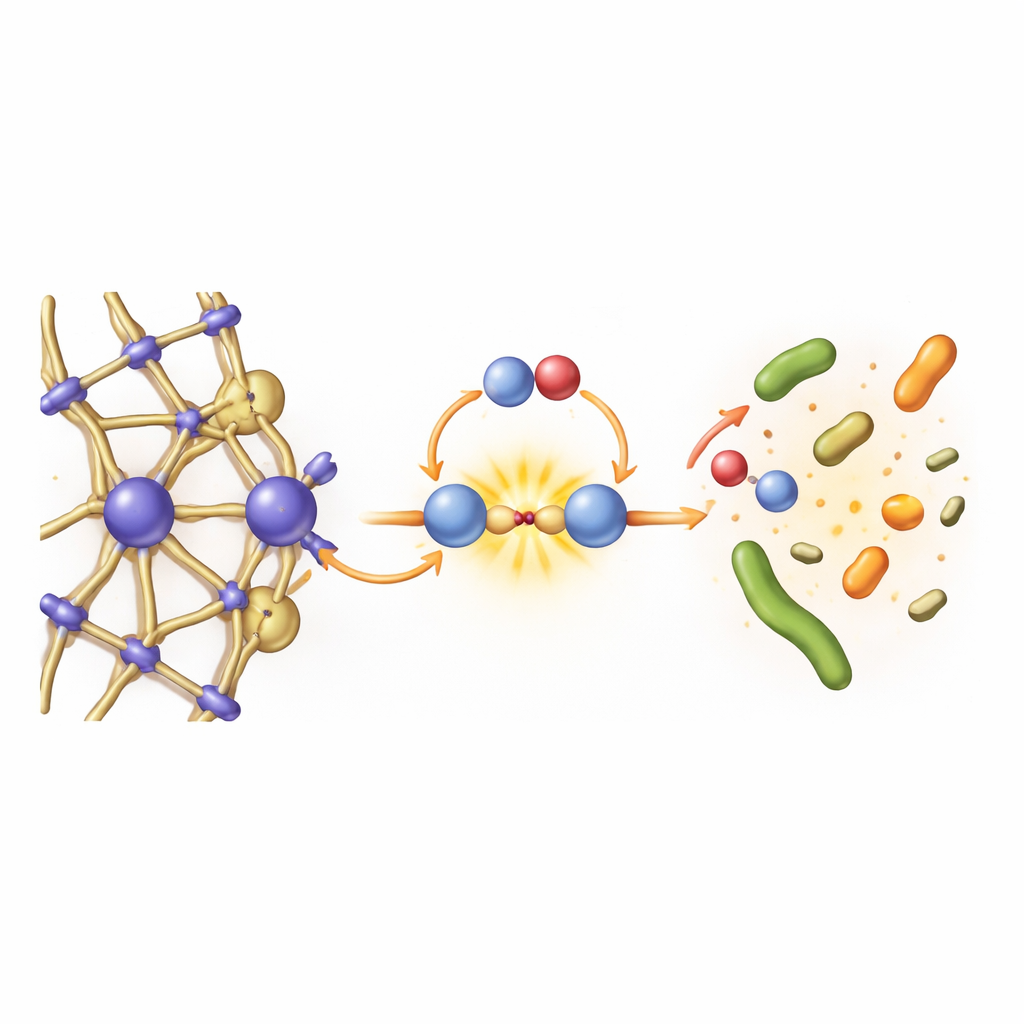
How stretching boosts the reaction
BUC-95’s true novelty lies in how its framework dynamics influence chemistry. When water and peroxydisulfate interact with the material, in situ X-ray, infrared, and Raman measurements show that the atomic lattice shifts slightly—evidence of dynamic stretching. Computer simulations and electrochemical tests compared BUC-95 with its rigid cousin BUC-96. Surprisingly, the rigid material actually transfers electrons to the oxidant more efficiently, yet it performs far worse in pollutant removal. The key difference is that BUC-95’s stretchable, dual-iron sites can adapt their spacing and electronic structure during the reaction. This flexibility fine-tunes the way peroxydisulfate binds and splits, making it easier to generate the iron–oxo species that drive efficient, nonradical oxidation.
Toward practical water cleanup
To move beyond lab beakers, the team mounted BUC-95 onto a porous sponge and built a small continuous-flow reactor. Polluted water containing ofloxacin and peroxydisulfate flowed through this reactor for over 100 hours, during which the system maintained nearly complete removal of the drug and kept iron release below drinking-water safety limits. Tests with mung bean seedlings and several bacteria showed that the treated water lost most of its toxicity, confirming that harmful drugs were not simply transformed into equally dangerous byproducts. These results demonstrate that a carefully designed, flexing solid framework can harness oxidants in a more controlled and effective way, offering a promising route to safer, more sustainable treatment of emerging pollutants in water.
What this means for the future
The study shows that the mechanical adaptability of a catalyst—its ability to stretch and subtly reorganize at the atomic level—can be as important as its composition. By designing MOFs like BUC-95 that deliberately use dynamic stretching to favor powerful iron–oxo species over short-lived radicals, researchers can build more selective, robust systems for cleaning complex wastewaters. This design principle could guide the next generation of advanced materials that help keep our water supplies free from persistent pharmaceuticals and other microscopic contaminants.
Citation: Wang, F., Li, YH., Wang, FX. et al. Dynamic stretching beyond electron transfer in a homointerpenetrated metal‒organic framework for enhanced Fenton-like reactions. Nat Commun 17, 2185 (2026). https://doi.org/10.1038/s41467-026-68917-z
Keywords: water purification, metal-organic frameworks, advanced oxidation, peroxydisulfate activation, antibiotic degradation