Clear Sky Science · en
Counteracting FOX proteins epigenetically control the herpesvirus lytic-latent balance
Why hidden herpes viruses matter
Many people carry herpes simplex viruses for life, often without knowing it. These viruses can sit quietly inside nerve cells for years and then suddenly flare up, causing cold sores, eye disease, or more serious problems in people with weak immune systems. What keeps the virus asleep most of the time, and what flips it back on, has been a long‑standing mystery. This study uncovers a key part of that control system: a tug‑of‑war between two opposing groups of human proteins that reshape viral DNA and determine whether herpes stays silent or starts making new viruses.
Two cellular teams pulling in opposite directions
The authors focused on a large family of human DNA‑binding proteins called FOX proteins, which normally help control our own genes in development and metabolism. By testing many FOX family members in nerve‑like cells, they found two clear camps. One group (including proteins like FOXF1) strongly boosted the replication of herpes simplex virus type 1 (HSV‑1) and several related “alpha” herpesviruses. Another group (FO XK1 and FOXK2) did the opposite, powerfully suppressing viral growth. This balance was not the same in every cell type: non‑neuronal cells and stressed neurons tended to make more of the activating FOX proteins, while resting sensory neurons made very little of them but high levels of the repressive FOXK proteins.
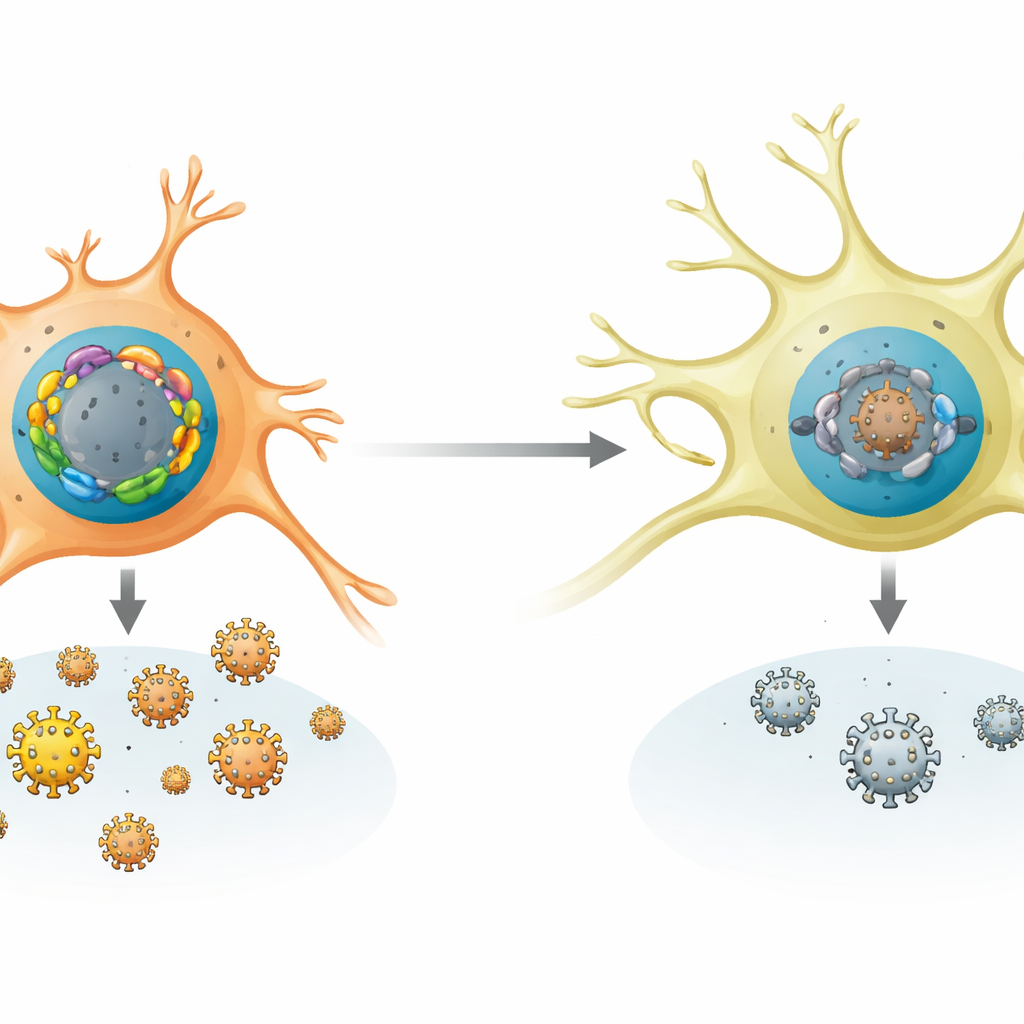
How nerve cells favor the quiet state
The team compared FOX gene activity across many human and mouse cell types and in infected animals. Sensory neurons, the natural home for latent HSV‑1, showed a pattern that favors silence: strong expression of FOXK1 and FOXK2, and weak expression of most activating FOX genes. In mice, early infection and later stress signals such as heat or tissue injury selectively raised the levels of several activating Fox genes without reducing the repressive ones. In cultured mouse neurons and in living mice, deleting or knocking down FOXK proteins made it easier for HSV‑1 to replicate and to spring back from latency. Conversely, forcing neurons to produce extra activating FOX proteins was enough to trigger reactivation even without chemical triggers, showing how sensitive the viral state is to this FOX balance.
Grabbing viral DNA and opening or closing it
To understand the underlying mechanism, the researchers mapped where FOX proteins sit on the viral genome. They found that both activator FOXF1 and repressor FOXK1 bind widely across HSV‑1 DNA, not just at a few specific sequence motifs. This binding relies on a conserved part of the FOX proteins that grips the DNA backbone rather than individual letters, allowing them to attach almost anywhere. Once in place, the two protein camps recruit different molecular partners that remodel chromatin, the combination of DNA and its packaging proteins. Activating FOX proteins, especially FOXF1, associate with CBP and P300, enzymes that add acetyl groups to histones and loosen chromatin. This makes viral DNA more accessible and boosts early, middle, and late viral gene activity. FOXK1, in contrast, works with factors like SIN3A and MAX that are linked to histone deacetylation and gene repression, tightening viral chromatin and keeping genes off.
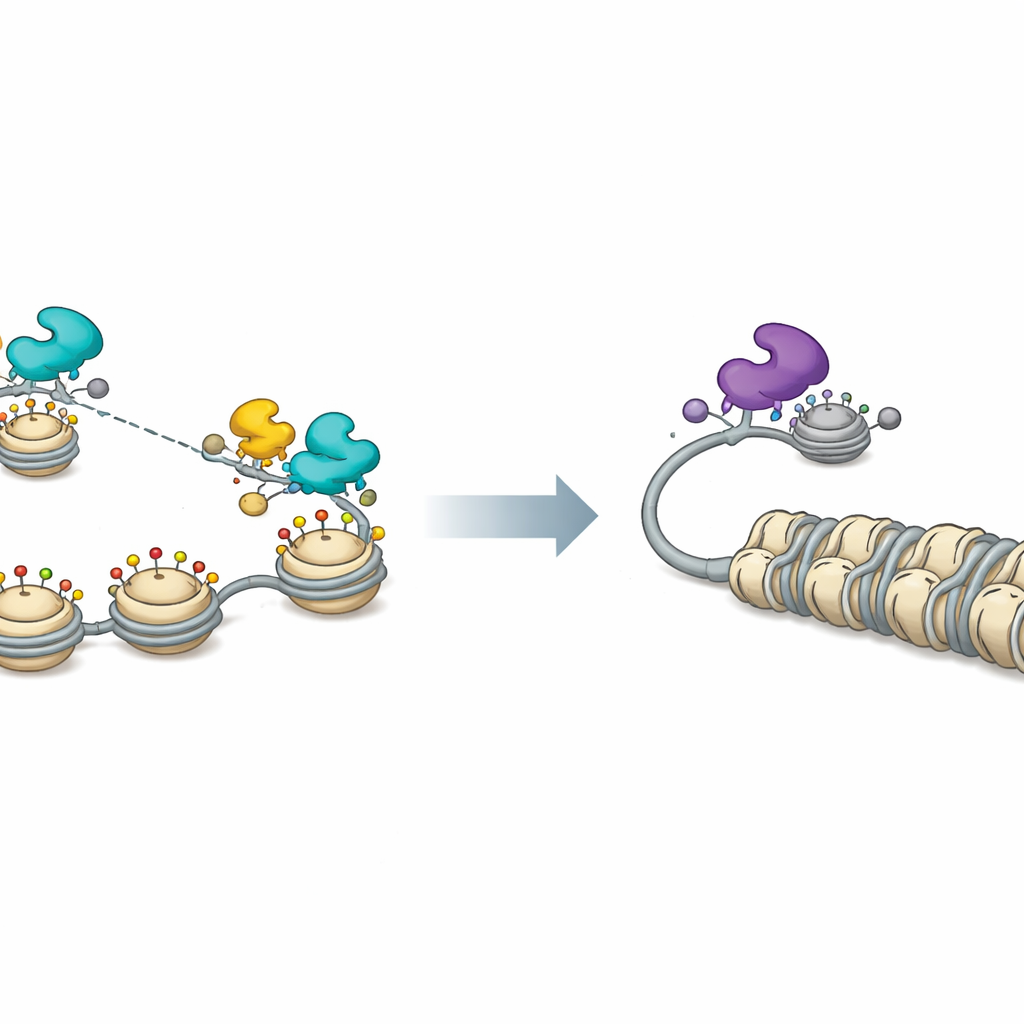
A chromatin switch for viral sleep and wakefulness
Measurements of viral chromatin confirmed this picture. When FOXF1 was present, histones on viral DNA were stripped back or chemically marked in ways associated with open, active chromatin, and the overall accessibility of viral promoters increased across the genome. FOXF1 expression also dispersed small nuclear structures called PML nuclear bodies that normally help keep viral DNA repressed. Blocking CBP and P300 erased many of these activating effects and reduced FOXF1‑driven viral gene expression. On the repressive side, the researchers showed that FOXK1‑associated partners and histone deacetylase activity were important for keeping HSV‑1 replication low; chemical inhibition of deacetylases both boosted virus output and weakened FOXK1‑mediated suppression.
What this means for controlling lifelong infection
Taken together, the work suggests that whether HSV‑1 is active or dormant depends heavily on the mix of FOX proteins in its host neuron. Resting sensory neurons naturally favor the repressive FOXK‑dominated state, pushing the virus into a deeply silent, chromatin‑compacted form. Stresses or other conditions that raise activating FOX proteins can tip this balance, opening viral chromatin and allowing the lytic cycle to restart. By revealing this epigenetic tug‑of‑war, the study highlights new ways scientists might one day push herpes viruses more firmly into harmless latency or, alternatively, force them out of hiding under controlled conditions so they can be eliminated.
Citation: Xiang, Y., Yang, X., Zhang, J. et al. Counteracting FOX proteins epigenetically control the herpesvirus lytic-latent balance. Nat Commun 17, 2256 (2026). https://doi.org/10.1038/s41467-026-68915-1
Keywords: herpes simplex virus latency, FOX transcription factors, viral chromatin, epigenetic regulation, viral reactivation