Clear Sky Science · en
Expanding the DNA damaging potential of artificial metallo-nucleases with click chemistry
Building Smarter Molecular Scissors
Chemotherapy often works by damaging the DNA of cancer cells, but today’s drugs can be blunt instruments that also harm healthy tissue. This study explores a new class of highly programmable “molecular scissors” made from copper and small organic pieces snapped together with click chemistry. By tuning their shape and how they grip DNA, the researchers aim to create agents that cut genetic material more precisely, opening paths toward future anticancer and antibacterial treatments.
Snapping Together a Three-Armed Cutter
The team uses copper-catalyzed click chemistry, a Nobel Prize–winning method known for its reliability, to assemble a family of molecules called Tri-Click (TC) ligands. Each TC molecule is a three-armed hub that can hold three copper ions at once, forming a compact cluster that can attack DNA. By swapping different chemical “donor” groups at the end of each arm—based on nitrogen, oxygen, or sulfur—the scientists systematically tested how subtle structural changes affect copper binding, DNA recognition, and biological activity. Among several new designs, versions bearing flat, ring-shaped nitrogen donors stood out as especially promising for gripping DNA tightly.
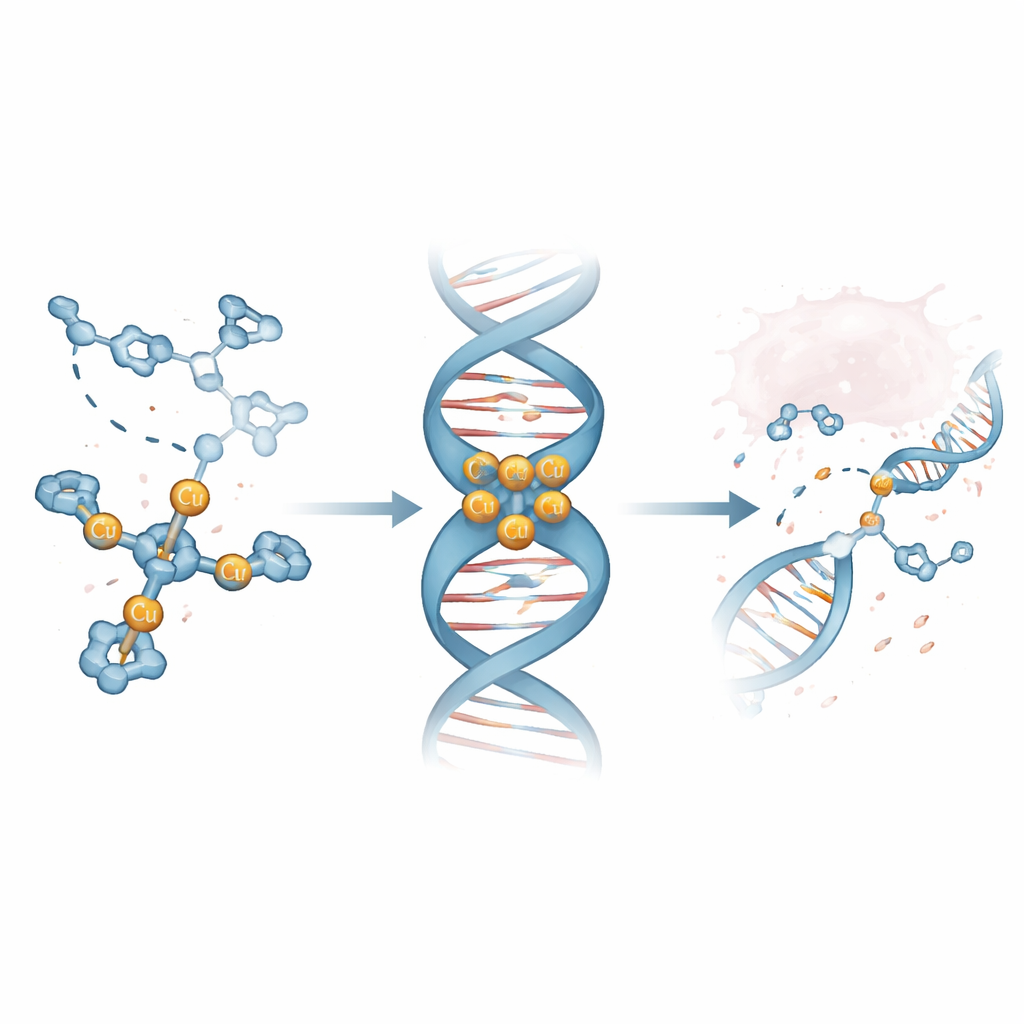
Finding a Lead Molecule That Loves DNA Grooves
Using mass spectrometry and optical methods, the researchers showed that one complex, called Cu3-TC-Py, reliably forms a three-copper cluster that is stable in solution. Competition experiments with fluorescent dyes bound to calf thymus DNA revealed that Cu3-TC-Py displaces these dyes at very low concentrations, indicating strong DNA binding. Follow-up tests on short, hairpin-shaped DNA pieces showed that this complex prefers to nestle into the minor groove—the narrower valley that winds around the DNA double helix—and that it particularly favors stretches rich in G and C base pairs. High-resolution computer simulations supported this picture, showing the three-armed complex sitting in the groove, its positive charges clasping the negatively charged DNA and subtly compacting the helix.
From Tight Binding to DNA Collapse and Breakage
At the single-molecule level, the team watched long DNA strands confined in nano-sized channels as they interacted with Cu3-TC-Py. At low doses, the complex competed with a stretching dye, displacing it as the complex bound in the grooves. At higher doses, the DNA began to shrink and then fully condense, consistent with strong electrostatic attraction between the copper cluster and the DNA backbone. When plasmid DNA was exposed to the complex in the presence of a reducing agent, the strands converted from their native coiled form to relaxed and then fully linear forms, a pattern signaling the formation of both single- and double-strand breaks. Further tests with radical scavengers implicated short-lived oxygen species—such as superoxide, singlet oxygen, and hydroxyl radicals—as the true cutting tools generated near the bound complex.
Attacking DNA in Cancer and Bacterial Cells
Moving beyond purified DNA, the researchers examined how Cu3-TC-Py behaves inside living cells. In cancer cell panels, the free ligands containing nitrogen-rich aromatic groups showed much stronger growth inhibition than earlier designs, and the fully formed copper complex was more potent still, killing several cancer cell lines at micromolar concentrations. Measurements of intracellular copper revealed that Cu3-TC-Py carries copper efficiently into cells and triggers a buildup of damaged DNA, as detected by a repair-assisted imaging method that marks lesions along stretched genomic strands. In bacteria, microscopy showed that treatment rapidly disrupted the compact bacterial chromosome, dispersing DNA throughout the cell and mimicking the severe fragmentation seen with a strong DNA-damaging antibiotic, consistent with widespread genetic destruction.
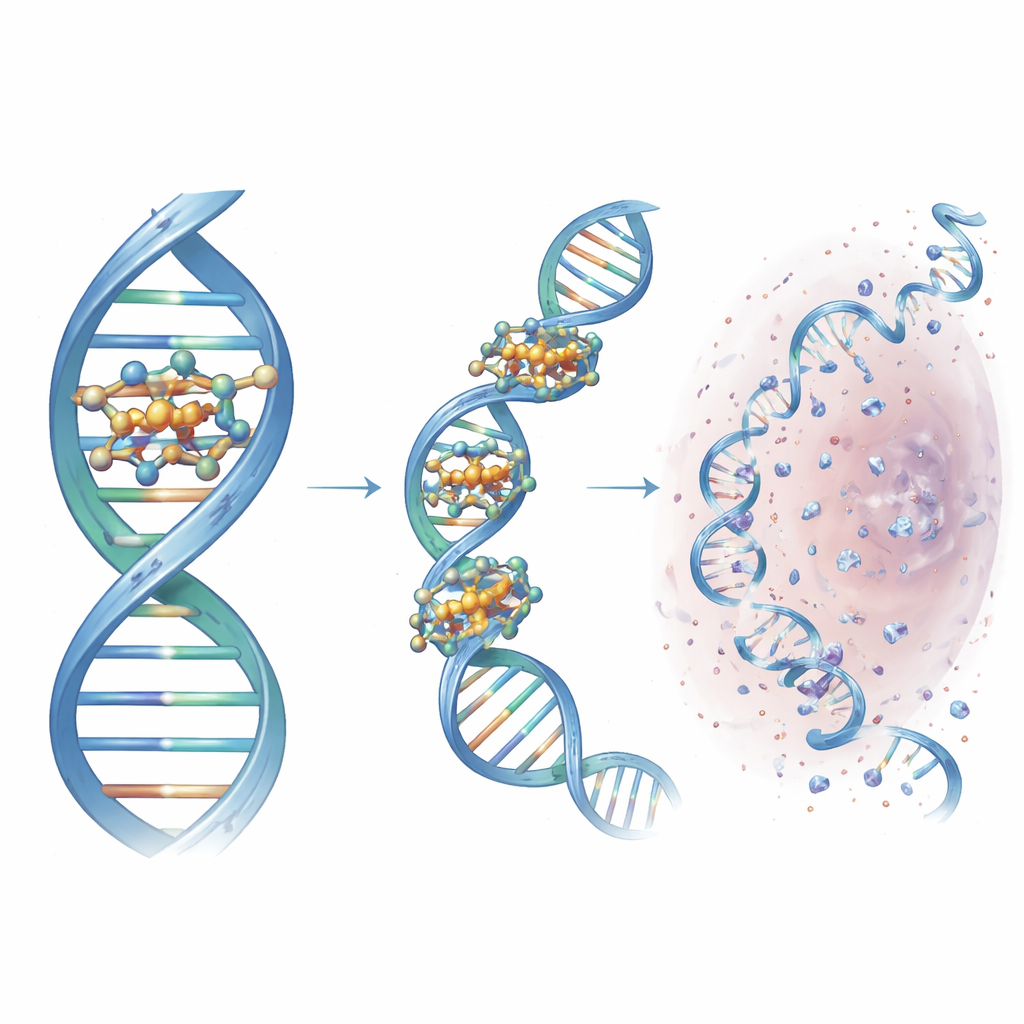
Why These Designer Scissors Matter
To a non-specialist, the key message is that the researchers have turned a simple, modular click-chemistry scaffold into a finely tuned DNA-cutting machine. By choosing the right ring-shaped nitrogen groups, they created a compact copper cluster that docks into a specific groove in DNA, pulls the strands together, and locally generates reactive oxygen species that slice the double helix. This lead compound, Cu3-TC-Py, damages DNA efficiently in both cancer and bacterial cells and shows how rational design can sharpen the action of metal-based drugs. While much work remains before clinical use, the study maps out design rules for next-generation molecular scissors that could one day offer more targeted cancer therapies and novel antimicrobial strategies.
Citation: Gibney, A., Sidarta, M., Delahunt, E. et al. Expanding the DNA damaging potential of artificial metallo-nucleases with click chemistry. Nat Commun 17, 2309 (2026). https://doi.org/10.1038/s41467-026-68911-5
Keywords: click chemistry, DNA damage, copper complexes, anticancer agents, artificial nucleases