Clear Sky Science · en
Succinate receptor 1 restricts hematopoiesis and prevents acute myeloid leukemia progression
When Cell Fuel Goes Rogue
Our blood is constantly renewed by a small population of stem cells hidden in the bone marrow. This study explores how a common metabolic by-product, succinate, and its sensor on the cell surface act as a brake on this renewal system. When that brake fails, the balance between healthy blood production and cancerous growth can tip toward acute myeloid leukemia (AML), an aggressive blood cancer. Understanding this hidden control switch could lead to more precise treatments that tame the disease by restoring the body’s own safeguards.
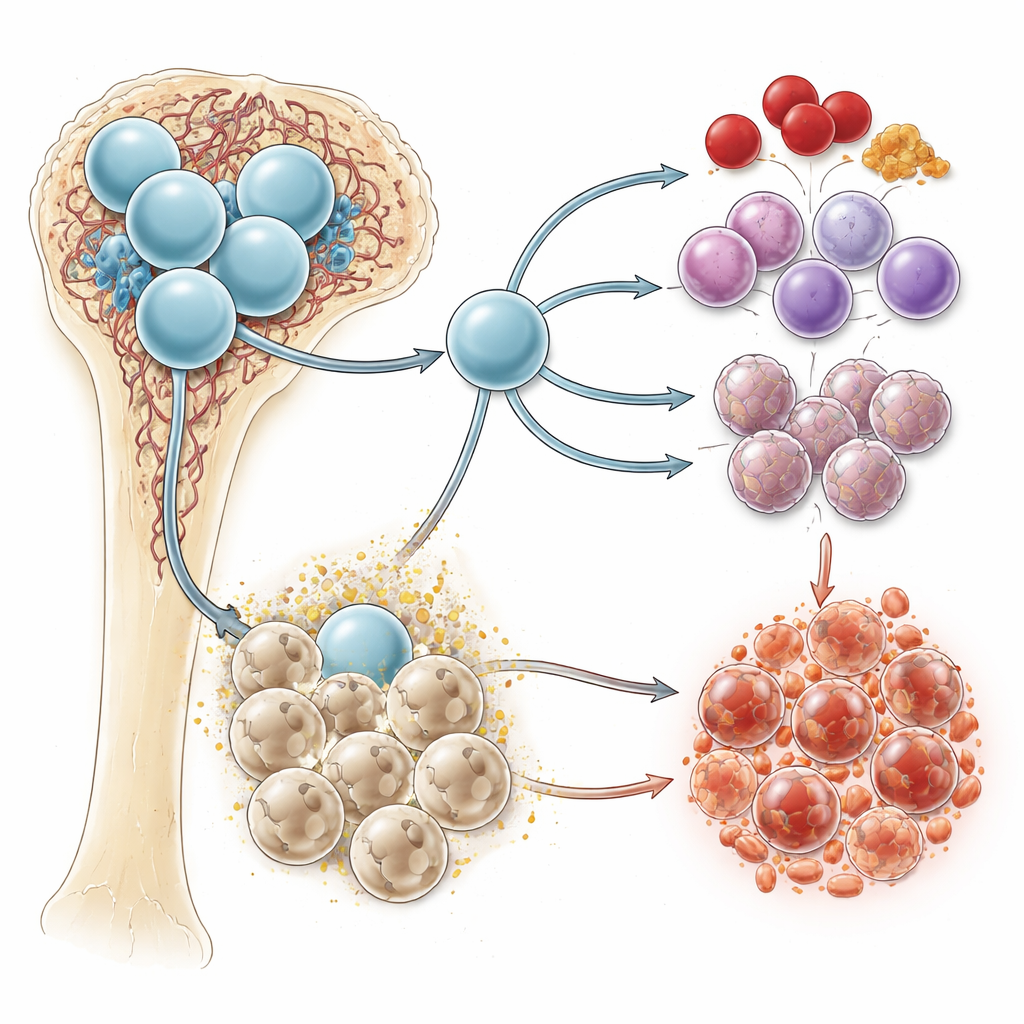
A Chemical Signal with a Double Life
Succinate is a routine by-product of how cells burn fuel, especially inside mitochondria, the cell’s power plants. Under low-oxygen conditions—as found in the bone marrow—succinate tends to build up. It can act inside cells, altering metabolism and inflammatory responses, and it can also slip outside cells to act as a signal. On the cell surface, a receptor called SUCNR1 detects succinate and relays its presence inward. Previous work linked succinate and SUCNR1 to inflammation and cancer-friendly environments, but their direct role in blood formation and leukemia was unclear.
A Warning Sign in Leukemia Patients
The researchers analyzed gene-expression data from hundreds of AML patients and discovered that SUCNR1 levels vary widely between individuals. Patients whose leukemia cells produced little SUCNR1 had shorter overall and progression-free survival, even after adjusting for age, sex, and standard clinical subtypes. Certain genetic subgroups of AML tended to show particularly low SUCNR1. When the team transplanted human AML cells lacking SUCNR1 into immune-deficient mice, then injected the animals with succinic acid (which becomes succinate in the body), the leukemic burden in bone marrow increased. This suggested that in cancers with low SUCNR1, extra succinate may actually fuel disease progression rather than restrain it.
The Hidden Brake on Blood Stem Cells
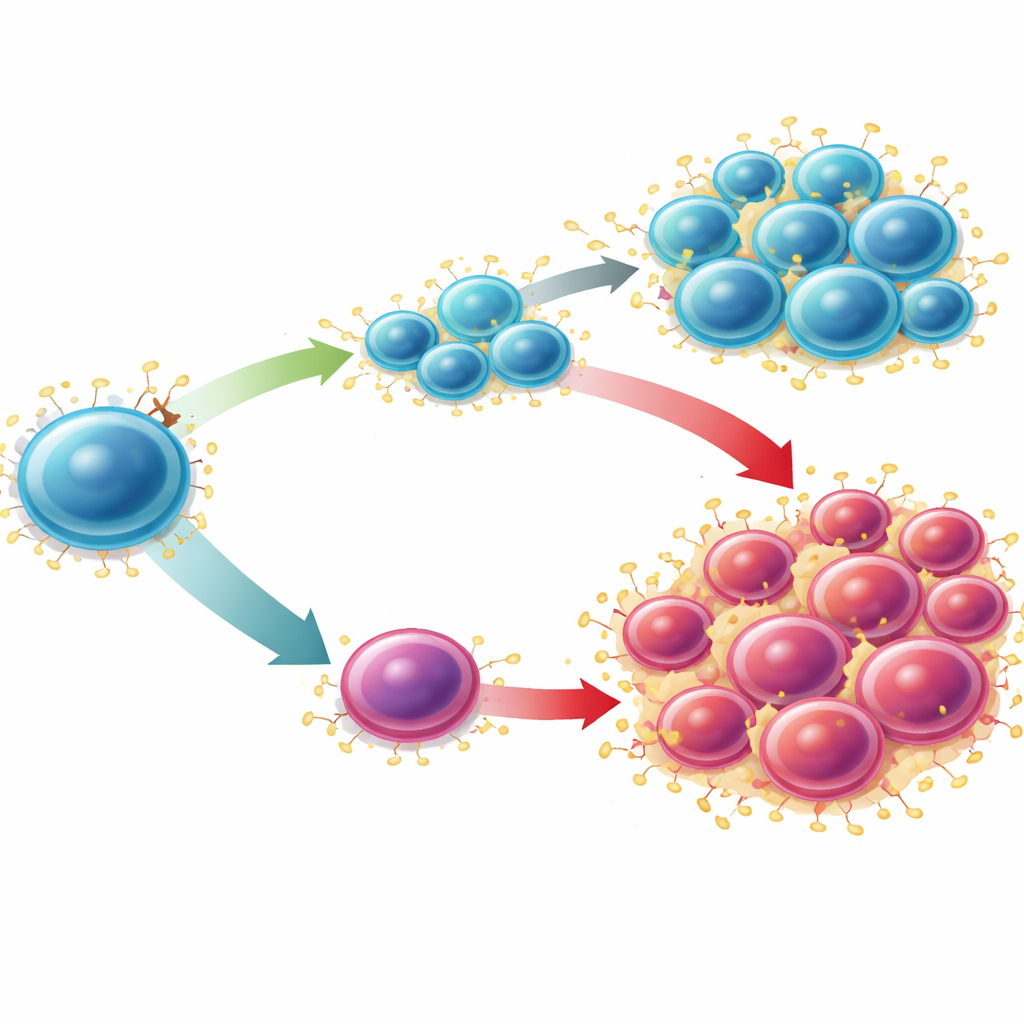
To understand how SUCNR1 acts in normal blood formation, the scientists turned to genetically engineered mice. When SUCNR1 was deleted either in the whole animal or only in blood-forming cells, the bone marrow became overactive. Stem and progenitor cells expanded, and both myeloid cells (such as monocytes and granulocytes) and B cells increased in blood and spleen. These stem-like cells survived better and formed more colonies in lab dishes, although they were slightly less capable of re-establishing blood production when transplanted in competition with normal cells. A special reporter mouse line showed that only a subset of stem and progenitor cells actually expresses SUCNR1; these SUCNR1-positive cells had particularly limited engraftment potential. Overall, the receptor acted as a restraint on the size and activity of the stem-cell pool.
From Balanced Defense to Inflammatory Overdrive
Detailed gene-expression profiling of stem and progenitor cells lacking SUCNR1 revealed a striking shift. Signatures of quiet, primitive stem cells were dampened, while genes linked to inflammation, reactive oxygen species, and mature blood cells were boosted. Two molecules stood out: S100A8 and S100A9, a pair of “alarmins” that amplify inflammatory signaling. These factors were increased both inside the stem-cell compartment and in the surrounding bone-marrow fluid. When the team treated SUCNR1-deficient mice with tasquinimod, a drug that blocks S100A9 signaling, the excess expansion of stem and progenitor cells, as well as the overproduction of myeloid and B cells, was largely reversed. In other words, much of the damage from losing SUCNR1 could be undone by cutting the S100A8/S100A9 alarm loop.
Turning a Vulnerability into a Treatment Strategy
The scientists then asked whether this pathway could be exploited therapeutically in AML. In aggressive mouse leukemia driven by the MLL-AF9 mutation—where Sucnr1 levels are very low—blocking S100A9 with tasquinimod, especially when combined with a potent SUCNR1-activating compound, reduced leukemia stem-like cells and lowered disease burden in bone marrow and spleen. Analyses of single-cell RNA data from human AML models treated with the chemotherapy drug cytarabine showed that resistant cell clusters were rich in S100A8 and S100A9, while SUCNR1 was sparse and associated with a different, metabolically active subset that shrank after treatment. In human leukemia cell lines, activating SUCNR1 increased chemotherapy-induced cell death and could trigger apoptosis on its own when SUCNR1 was overproduced. Together, these findings position low SUCNR1 not only as a marker of poor prognosis but also as a weakness: leukemia cells that have shed this brake may depend heavily on the S100A8/S100A9 axis and be particularly sensitive to strategies that reactivate SUCNR1 signaling or block the downstream alarm.
A New Lever to Tame Leukemia
For a lay reader, the central message is that a seemingly mundane metabolic by-product, succinate, and its receptor SUCNR1 form an unexpected safety system in blood formation. When SUCNR1 is present and active, it keeps stem cells in check and prevents runaway growth. When SUCNR1 is lost or low—common in some AML patients—succinate and inflammatory alarmins such as S100A8 and S100A9 can push stem and progenitor cells toward overexpansion and leukemia. By combining drugs that dampen this inflammatory alarm with agents that restore or mimic SUCNR1 signaling, it may be possible to steer the system back toward balanced blood production and improve the effectiveness of existing chemotherapy.
Citation: Cuminetti, V., Boet, E., Heugel, M. et al. Succinate receptor 1 restricts hematopoiesis and prevents acute myeloid leukemia progression. Nat Commun 17, 2403 (2026). https://doi.org/10.1038/s41467-026-68906-2
Keywords: acute myeloid leukemia, succinate receptor, hematopoietic stem cells, metabolism and cancer, inflammatory signaling