Clear Sky Science · en
Exploiting a high-performance magnesium-fluoride battery prototype enabled by anion-receptor-mediated electrolyte
Safer, Cheaper Batteries for a Power-Hungry World
As our homes, cars, and grids demand ever more electricity, today’s lithium-ion batteries face tough questions about cost, safety, and raw materials. This study explores a promising alternative: batteries built around magnesium, a common, inexpensive metal. By cleverly redesigning the liquid inside the battery—the electrolyte—the researchers show how to unlock a new, high-energy magnesium–fluoride battery that works efficiently, lasts for hundreds of cycles, and even keeps going at sub-zero temperatures.
Why Magnesium Deserves a Closer Look
Magnesium batteries are attractive because magnesium metal is abundant in the Earth’s crust and can store a great deal of charge in a small volume. Unlike lithium, magnesium is less prone to forming needle-like growths that can pierce the separator and cause short circuits, improving safety. Yet magnesium technology has stalled, mainly because it is hard to find a suitable positive electrode (the “cathode”) that can deliver both high energy and long life. Traditional materials such as sulfides and oxides either run at low voltages, limiting how much energy the battery can deliver, or they move magnesium ions so slowly that power and lifetime suffer. Metal fluorides, especially iron fluoride and iron oxyfluoride, offer far higher energy, but they are notoriously difficult to run efficiently with magnesium.
A Smart Additive that Tames a Difficult Electrolyte
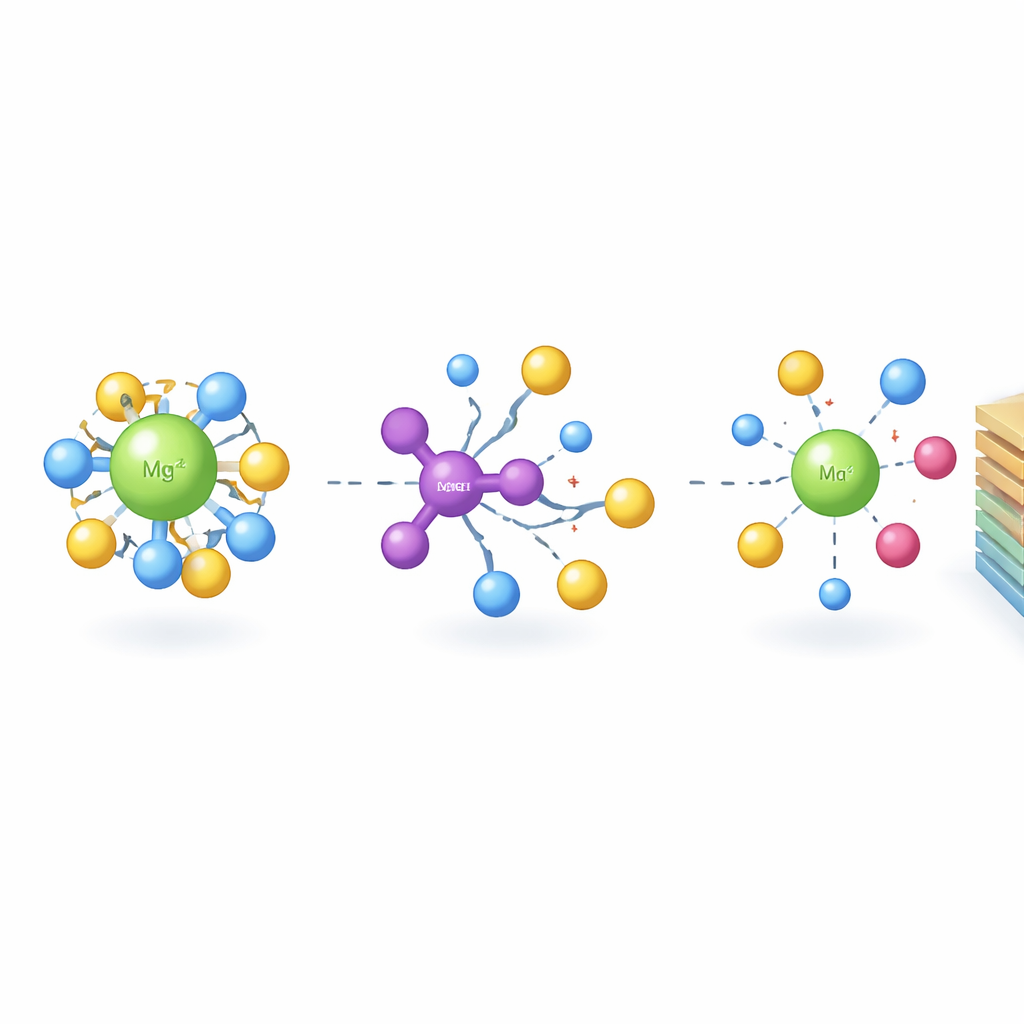
The heart of the problem lies in the electrolyte, the liquid that carries charge between the battery’s two electrodes. A popular magnesium electrolyte, known as an all-phenyl-complex solution, conducts ions well and is compatible with magnesium metal, but it contains chloride-based clusters that aggressively corrode metal parts and decompose at high voltages. The team introduces a special molecule, tris(pentafluorophenyl)borane, that acts as an “anion receptor” in this liquid. Using computer simulations, nuclear magnetic resonance, and Raman spectroscopy, they show that this additive selectively grabs chloride-containing species and interacts with the solvent. This breaks up the most corrosive magnesium–chloride clusters, spreads out negative charge, and weakens how tightly the solvent and chloride cling to magnesium and lithium ions.
Making Ions Move Faster and Surfaces Last Longer
By loosening these bindings, the tailored electrolyte lowers the energy cost for ions to shed their solvent and chloride “shells” before entering or leaving the electrode—a step that often slows batteries down. Calculations reveal that the additive significantly reduces the barrier for magnesium–chloride bond breaking, the slowest step in the process. Experiments confirm that this chemistry broadens the safe operating voltage of the electrolyte and sharply reduces corrosion of common metal current collectors. At the same time, magnesium can still be plated and stripped reversibly at the negative electrode. Overall, the electrolyte maintains similar bulk conductivity to the original solution but dramatically improves interfacial stability and charge-transfer kinetics.
A High-Energy Magnesium–Fluoride Battery in Action
Armed with this improved electrolyte, the researchers build a full magnesium battery using an iron oxyfluoride positive electrode. The design cleverly combines lithium and magnesium ions: lithium ions help the iron oxyfluoride react quickly and reversibly, while magnesium metal at the negative side provides high energy and safety. In tests at room temperature, the battery delivers a high reversible capacity of about 354 milliampere-hours per gram and maintains useful capacity even at a tenfold higher current. At –20 °C, it still supplies 177 milliampere-hours per gram over 200 cycles. When the reaction is limited to gentler “intercalation” processes, the cells cycle more than 500 times with only tiny capacity losses per cycle and an average voltage around 1.77 volts, indicating long-term durability.
What This Means for Future Energy Storage
To an everyday user, the take-home message is that smarter chemistry inside the electrolyte can turn a promising but problematic material set into a practical, high-performance battery. By using an anion receptor to neutralize corrosive species and speed up ion motion, the team opens the door for high-energy magnesium–fluoride batteries that are safer, cheaper, and more tolerant of cold conditions than many current technologies. While further work is needed to cut initial losses and scale up, this anion-receptor strategy offers a powerful tool for designing next-generation batteries that move beyond lithium while still delivering the performance modern energy systems require.
Citation: Chen, K., Lei, M., Wang, T. et al. Exploiting a high-performance magnesium-fluoride battery prototype enabled by anion-receptor-mediated electrolyte. Nat Commun 17, 2143 (2026). https://doi.org/10.1038/s41467-026-68903-5
Keywords: magnesium batteries, electrolyte design, iron oxyfluoride cathode, anion receptor, energy storage