Clear Sky Science · en
Twist engineering induced spin-orbit coupling for photosynthesis of ethane from carbon dioxide and water
Turning Air and Water into a Useful Fuel
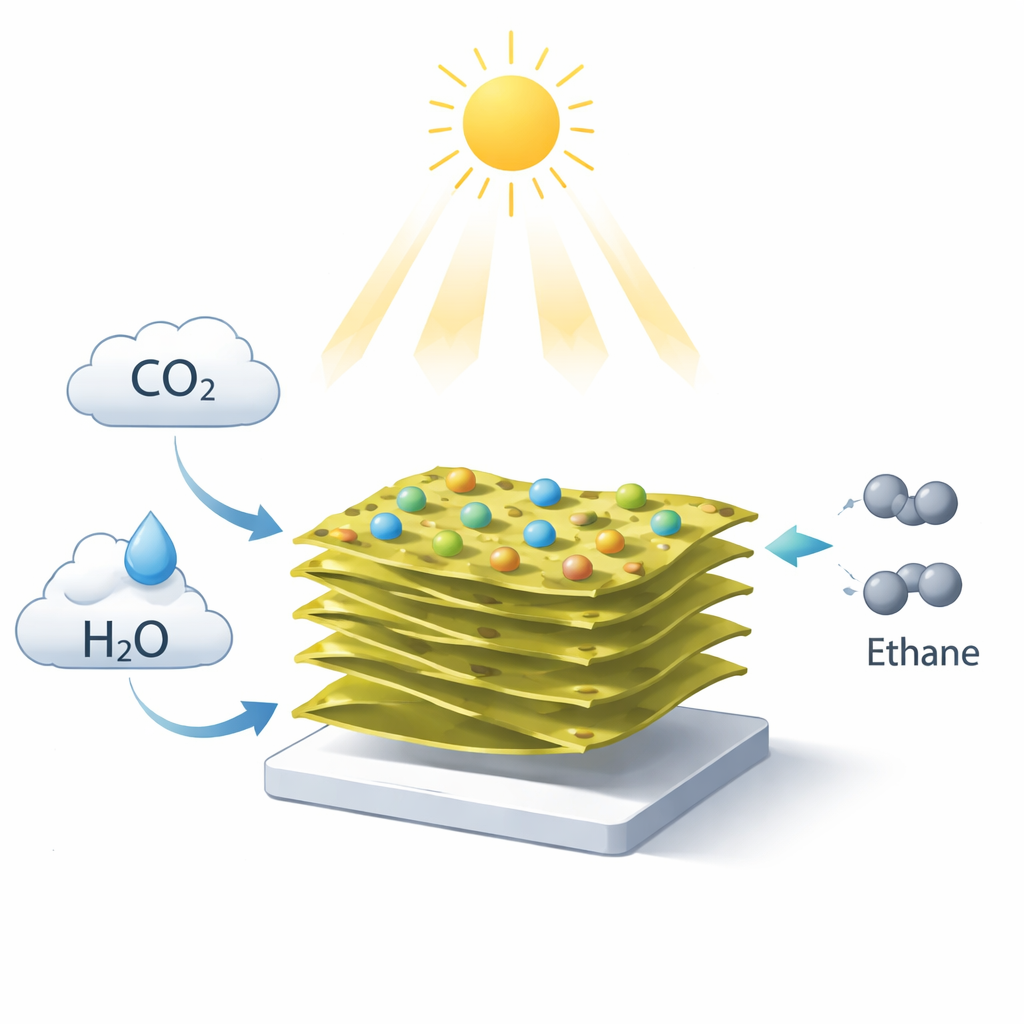
Imagine using nothing more than sunlight, the carbon dioxide in the air, and ordinary water to make a clean fuel. That is the vision behind this research, which explores a new material that can "photosynthesize" ethane, an energy-rich two‑carbon molecule used as a fuel and industrial building block. By carefully arranging atoms in ultra‑thin layers, the scientists found a way to steer the tiny magnetic properties of electrons so that this artificial leaf works faster and wastes far less energy.
A New Way to Build an Artificial Leaf
At the heart of the study is a specially engineered catalyst made from sheets of a tin and sulfur compound (SnS2) that are gently twisted with respect to each other and decorated with isolated nickel atoms. This material, called Ni‑TSnS2, forms a delicate "moiré" pattern, similar to what you see when two window screens are overlaid at an angle. That pattern creates a regular landscape of slight strains and distortions in the crystal, and those distortions subtly change how electrons move. The nickel atoms sit at carefully chosen spots in this pattern, acting as individual reaction hot‑spots that help break down carbon dioxide and rebuild it into more complex molecules.
Why Electron Spin Matters
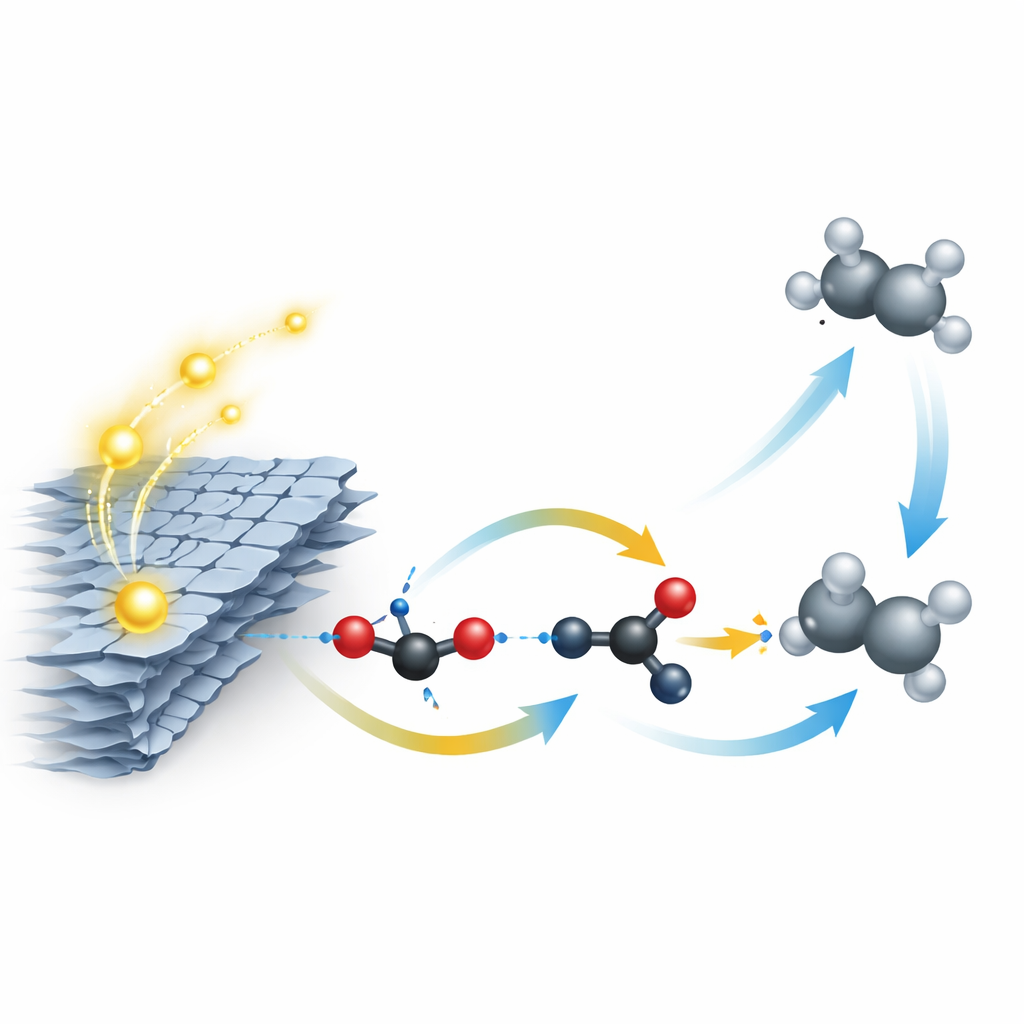
Electrons do not just carry charge; they also behave like tiny bar magnets with a property called spin. When light strikes the catalyst, electrons are excited and can either drive chemical reactions or fall back and waste their energy as heat or light. In this material, the twisted layers and low‑symmetry nickel sites combine to create a strong interaction between an electron’s motion and its spin. That interaction, known in physics as spin–orbit coupling, locks the direction of spin to the way electrons travel through the material. Because electrons and their positively charged partners (holes) with opposite spins have trouble recombining, charges live longer and are more available to power the reaction that turns carbon dioxide and water into fuel.
Guiding Reactions Toward Ethane
Converting carbon dioxide into two‑carbon products like ethane is usually very difficult. It requires many electrons and a high‑energy step where two carbon‑containing fragments join on the surface of a catalyst. Instead of relying on that slow step, the Ni‑TSnS2 material takes a different route. Experiments that watch reaction intermediates in real time, along with computer simulations, show that carbon dioxide is reduced step‑by‑step to a surface‑bound methyl group (CH3). Thanks to the special spin behavior at the nickel sites, an extra electron can hop onto this group, turning it into a highly reactive methyl radical. These radicals then couple with each other in a rapid chain reaction in the surrounding solution, forming ethane without needing to overcome the usual energy barrier on the surface.
A Highly Efficient and Stable System
The result of this design is a remarkable boost in performance. Compared with simpler versions of the material, the twisted, nickel‑decorated sheets dramatically increase how long photogenerated charges survive and how well they are separated. Measurements show a more than 30‑fold increase in surface photovoltage, over 40‑fold longer lifetimes for reactive charges, and a strong match between spin–orbit strength and catalytic activity. Under simulated sunlight, Ni‑TSnS2 produces ethane at a high rate while directing nearly 90 percent of the available electrons into making this single product. The catalyst maintains its structure and activity over many hours of operation, suggesting that the spin‑organized state is both robust and practical.
From Fundamental Physics to Cleaner Carbon Cycles
In plain terms, this study shows that carefully twisting and decorating atom‑thin layers can give engineers a new dial to tune: the spin of moving electrons. By harnessing that hidden degree of freedom, the researchers created a photocatalyst that turns carbon dioxide and water into an energy‑rich fuel more efficiently and selectively than before, while avoiding the usual chemical bottlenecks. If such strategies can be scaled and adapted to other materials, they could become powerful tools for recycling greenhouse gases into useful products, nudging our energy and chemical systems toward a more sustainable carbon cycle.
Citation: Liu, Z., Gao, Y., Chen, L. et al. Twist engineering induced spin-orbit coupling for photosynthesis of ethane from carbon dioxide and water. Nat Commun 17, 2195 (2026). https://doi.org/10.1038/s41467-026-68901-7
Keywords: photocatalytic CO2 reduction, ethane photosynthesis, spin orbit coupling, single atom catalysts, twisted 2D materials