Clear Sky Science · en
Engineered alcohol oxidases catalyse transesterification in aqueous media without competing hydrolysis
Why this enzyme story matters
Many everyday products, from medicines and food flavorings to biodiesel, depend on a simple type of chemical bond called an ester. Making and tweaking these esters efficiently, cheaply, and in environmentally friendly ways is a major goal in green chemistry. The challenge is that water, the ideal solvent from a safety and sustainability standpoint, usually sabotages these reactions by breaking esters apart as fast as they are formed. This study uncovers an engineered enzyme that can build esters in water while almost completely ignoring the water around it, opening a route to cleaner industrial chemistry.
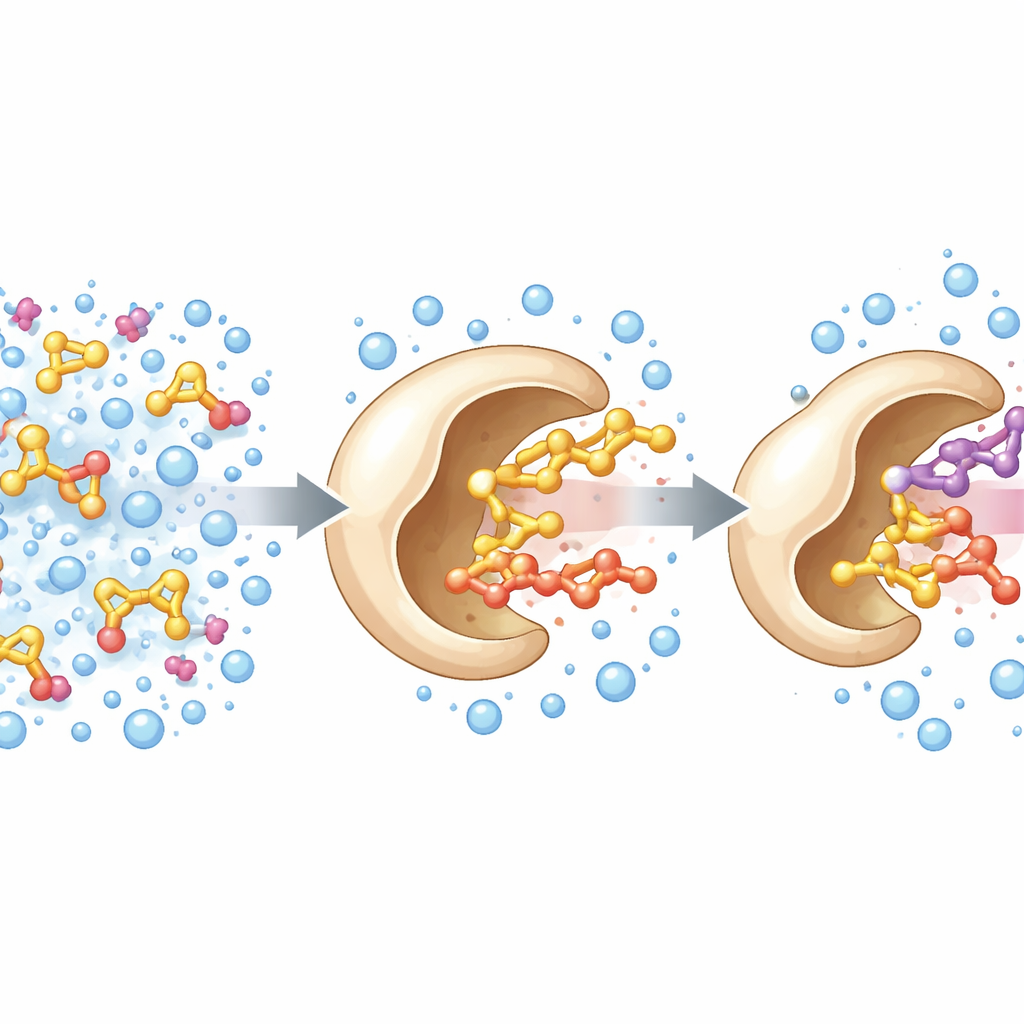
Turning a known enzyme to a new job
The researchers started from a known enzyme, an alcohol oxidase from a wood‑rotting fungus. In nature, this enzyme uses a flavin cofactor to help strip electrons from small alcohols such as methanol. The team set out to broaden its reach so that it could handle bulkier alcohols, which are more relevant for fine chemicals and flavors. Using the enzyme’s three‑dimensional structure as a roadmap, they focused on three amino acids that form a sort of gateway into the active site. By randomizing these positions and screening the resulting variants, they discovered a triple mutant—named PcAOx‑VPN—that could efficiently process larger alcohols like benzyl alcohol while losing much of its original activity toward tiny ones such as methanol.
Unexpected talent: building esters in water
While testing PcAOx‑VPN in mixtures designed to dissolve oily substrates, the team noticed extra peaks in their analytical readouts. These turned out to be acetylated products: the enzyme was transferring an acyl group from an activated ester donor to alcohols, a reaction known as transesterification. Remarkably, this took place in water‑based buffer, something that usually leads to strong competing hydrolysis, where water attacks and destroys the ester. Here, PcAOx‑VPN efficiently joined alcohols and acyl donors such as vinyl acetate to form new esters, often with yields above 80%, and with only modest side reactions. The same enzyme could even act on a wide variety of alcohols, including straight‑chain, aromatic, chiral, and sulfur‑containing examples, and showed a marked preference for one mirror‑image form over the other in several cases.
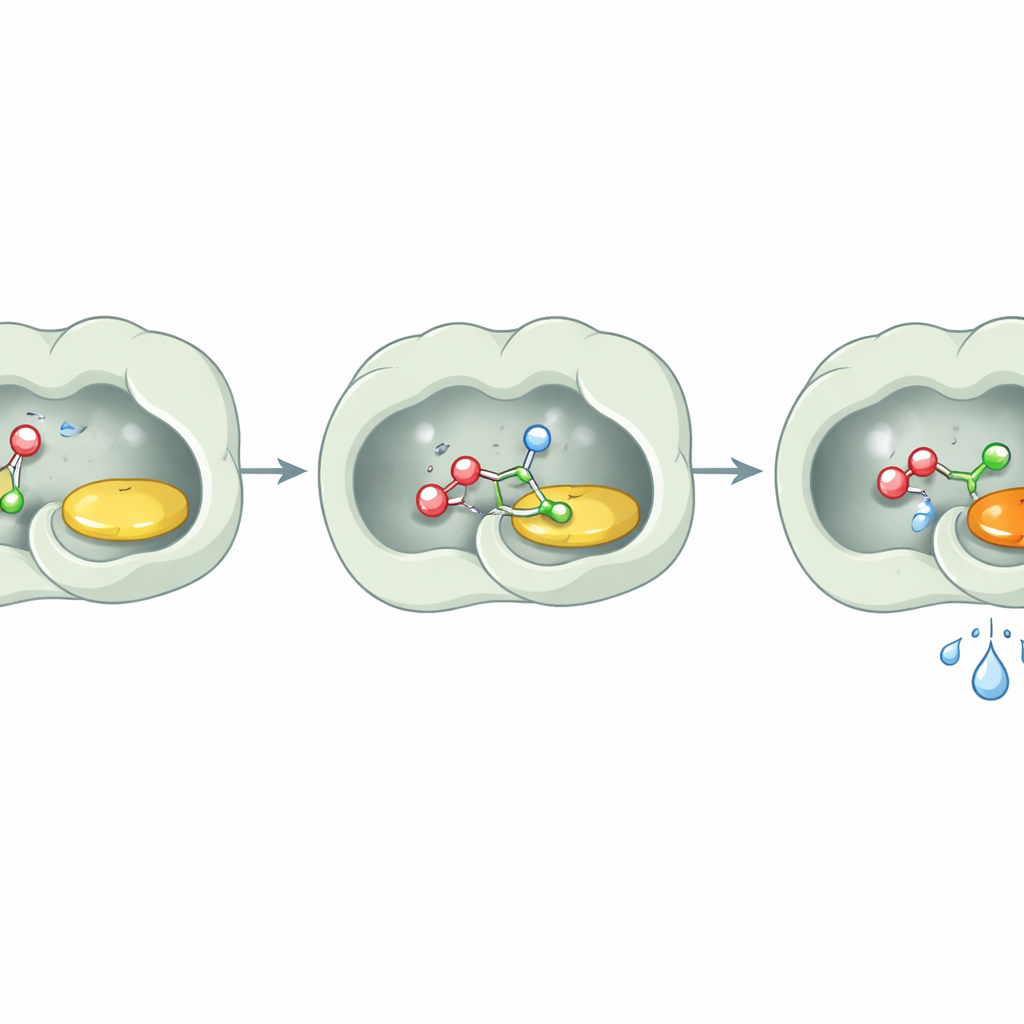
Keeping water out of the way
A key surprise was what the enzyme did not do: it did not noticeably hydrolyze esters. Even after long incubations with ester substrates and water, PcAOx‑VPN left them essentially intact unless an additional acyl donor was provided. Structural studies offer a simple explanation. The active site is lined with largely oily, aromatic amino acids that create a highly hydrophobic pocket. Detailed analysis showed no water molecules close to the flavin cofactor, and computational tools confirmed that the channel leading into the active site is also water‑unfriendly. In essence, the enzyme builds a tiny dry room inside a watery world. Alcohols and activated esters can enter and react, but water molecules are kept at bay, so they never get a chance to break the product apart.
How the reaction likely works inside
Mechanistic experiments and mutational analysis revealed that two amino acids, a histidine and an asparagine, are central to both the enzyme’s natural oxidation role and its new transesterification talent. The histidine acts as a base, helping to pull a proton off the incoming alcohol and make it more reactive, while the asparagine stabilizes the charged state that results. Together, they promote the alcohol’s attack on the acyl donor to form a short‑lived intermediate that collapses into the desired ester. The oxidized state of the flavin cofactor is also required: under oxygen‑free conditions, when the flavin becomes reduced, the transesterification reaction stops and resumes only when air is reintroduced. Notably, when the same trio of mutations was installed in related oxidases from other fungi, those enzymes also gained robust transesterification activity, suggesting that this behavior can be generalized.
What this means for greener chemistry
For non‑specialists, the central message is that the authors have taught a known enzyme to do a new trick: build useful esters directly in water, while effectively ignoring the water’s tendency to undo the job. By carving out a dry, oil‑friendly pocket inside a protein and tuning a few key amino acids, they created a catalyst that favors alcohols over water as reaction partners. Because the same design principle works in related enzymes, this approach could be extended to a family of catalysts for making flavors, fragrances, fine chemicals, and possibly biofuels in safer, more sustainable processes that rely less on harsh organic solvents and more on water.
Citation: Wu, B., Ma, Y., Feng, C. et al. Engineered alcohol oxidases catalyse transesterification in aqueous media without competing hydrolysis. Nat Commun 17, 2183 (2026). https://doi.org/10.1038/s41467-026-68899-y
Keywords: enzyme engineering, biocatalysis, green chemistry, ester synthesis, flavoproteins