Clear Sky Science · en
A lattice oxygen-free design for efficient and stable photothermal methane dry reforming
Turning Greenhouse Gases into Useful Fuel
Methane and carbon dioxide are two of the most powerful greenhouse gases warming our planet, but they are also rich sources of chemical energy. This study shows how carefully engineered tiny metal particles can use light and heat together to convert these gases into syngas—a versatile mixture of hydrogen and carbon monoxide—while avoiding the usual problems that make such processes inefficient and short‑lived.
Why Cleaning Up Methane Is So Hard
Dry reforming of methane is a reaction that turns methane and carbon dioxide into syngas. In industry, it normally demands furnace‑like temperatures of 700–1000 °C to proceed fast enough. At those temperatures, common nickel and cobalt catalysts tend to clump together and accumulate carbon deposits, losing activity over time. Recent “photothermal” approaches aim to use concentrated light to heat catalysts more gently and drive extra electronic effects, but current materials waste most of the incoming light and still suffer from carbon buildup and catalyst damage.
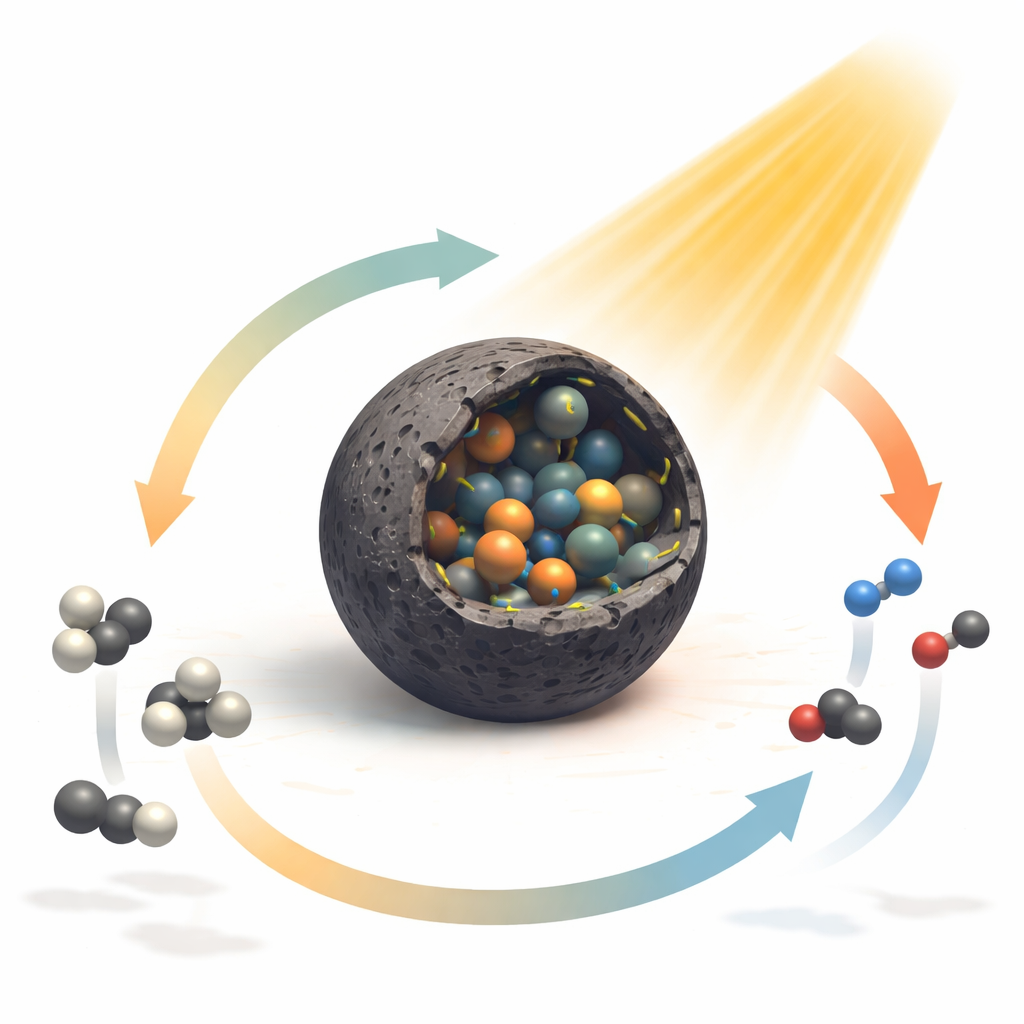
Designing a New Kind of Catalyst Shell
The researchers tackled this challenge by building a catalyst from a metal–organic framework—a crystalline scaffold that arranges metal atoms and organic linkers in a regular pattern. After a tailored heat treatment, this framework turns into spherical particles coated with a thin layer of graphitic carbon, inside of which sit very small nickel–cobalt alloy nanoparticles. Crucially, nitrogen atoms are woven into the carbon shell and linked to nickel, forming what the authors call C–N–Ni sites. These nitrogen‑nickel connections reshape how electrons are shared between nickel and cobalt and between the metals and the carbon layer, subtly straining the crystal lattice and turning the surface into a more responsive playground for incoming molecules.
Letting Reactive Oxygen Do the Heavy Lifting
In traditional catalysts for this reaction, oxygen built into the solid lattice plays a key role in breaking methane’s strong C–H bonds and cleaning up carbon fragments. But lattice oxygen is hard to move, and using too much of it eventually damages the catalyst. Here, the team engineered a completely different route: instead of relying on built‑in oxygen, they harness highly reactive oxygen and hydroxyl species generated directly from carbon dioxide during the reaction. Experiments and computer simulations show that the nitrogen‑modified nickel–cobalt surface strongly grabs both methane and carbon dioxide, but steers them to different metal atoms—nickel specializes in splitting methane, while cobalt focuses on activating carbon dioxide. The reactive oxygen species formed from carbon dioxide then rapidly oxidize carbon‑rich fragments from methane into intermediates like formaldehyde and finally into carbon monoxide and carbon dioxide, preventing solid carbon from ever piling up.
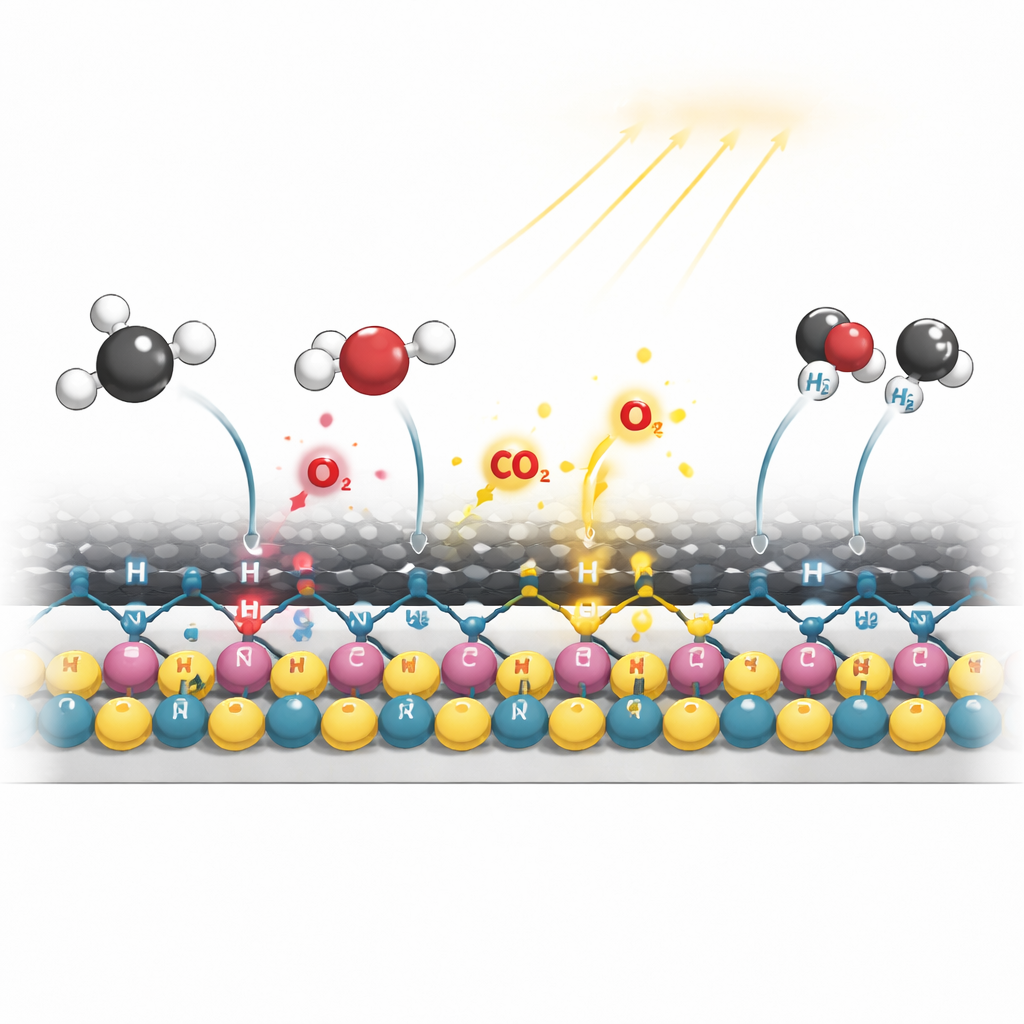
How Light Makes the Catalyst Smarter
Using in situ spectroscopy, the authors observed what happens to the catalyst while it is working under both dark and illuminated conditions. Without light, the nickel and cobalt surfaces tend to oxidize, and water‑forming side reactions become more prominent, gradually weakening performance. Under light, however, electrons excited in the carbon coating are funneled along the C–N–Ni pathways toward the metal sites. This extra electron density helps keep nickel and cobalt in their metallic, active state, suppresses unwanted side reactions, and strengthens the formation of key intermediates such as a surface‑bound COOH species that breaks into carbon monoxide and hydroxyl radicals without attacking the metal. Detailed quantum‑chemical calculations confirm that this light‑assisted pathway lowers the energy barriers for methane dehydrogenation and carbon fragment oxidation while raising the barrier for the step that would otherwise leave behind stubborn carbon deposits.
Efficiency and Stability Under Milder Conditions
The optimized nitrogen‑doped catalyst, labeled N1, delivered syngas with a near‑ideal hydrogen‑to‑carbon monoxide ratio and achieved a light‑to‑chemical energy efficiency of about 52 percent—competitive with or better than many reported solar‑driven systems—at a relatively modest operating temperature of 540 °C. It maintained its performance for 200 hours of continuous operation with almost no sign of catalyst restructuring or added amorphous carbon. By designing a lattice‑oxygen‑free route that uses reactive oxygen drawn directly from carbon dioxide and guiding electrons along precise nitrogen‑nickel pathways, this work points to a new family of durable, light‑assisted catalysts that could both recycle greenhouse gases and produce valuable fuels more efficiently.
Citation: Pan, T., Xu, W., Deng, H. et al. A lattice oxygen-free design for efficient and stable photothermal methane dry reforming. Nat Commun 17, 2151 (2026). https://doi.org/10.1038/s41467-026-68898-z
Keywords: methane dry reforming, photothermal catalysis, NiCo catalyst, syngas production, greenhouse gas conversion