Clear Sky Science · en
Green chemical process for continuous production of high-purity 2,5-furandicarboxylic acid in anion exchange membrane flow electrolyzer
Turning Plants into Cleaner Plastics
Many of the plastics and everyday materials we rely on still come from oil, bringing a heavy carbon footprint. This study explores a different path: starting from plant-based ingredients and using electricity to make a key building block for next‑generation plastics. The work shows how a carefully engineered electrochemical device can continuously produce this ingredient at high purity and competitive cost, while also generating clean hydrogen gas as a bonus.
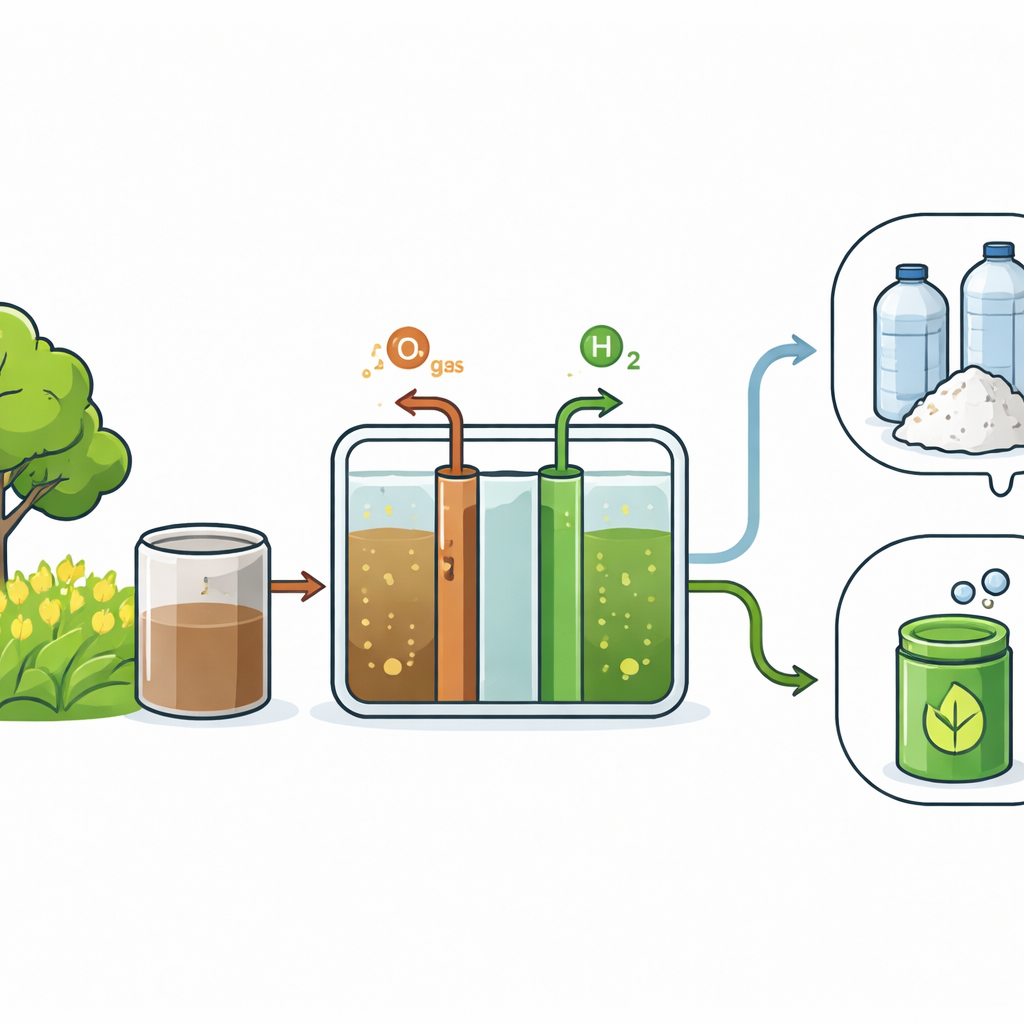
Why a New Plastic Ingredient Matters
Instead of relying on fossil fuels, chemists can now make an important plastic precursor called FDCA from sugars found in biomass such as agricultural waste and wood. FDCA can replace the fossil‑based component in familiar plastics like polyethylene terephthalate, leading to bio‑based materials such as polyethylene furanoate (PEF). These new plastics can have better barrier properties for bottles and packaging, and because their carbon ultimately comes from plants, they help close the carbon loop. The challenge has been making FDCA efficiently, cleanly, and at a scale that makes economic sense.
Using Electricity to Drive Green Chemistry
The authors focus on an electrochemical route, where a biomass‑derived liquid called HMF is transformed into FDCA inside a compact device similar to a fuel cell. In this setup, HMF flows past a metal‑based catalyst on one side of a thin plastic membrane, while water is split on the other side to make hydrogen gas. Electrons from the external circuit do double duty: they help convert HMF into FDCA and, at the same time, generate hydrogen that can be used as a clean fuel or chemical feedstock. Because the power source can be solar, wind, or other renewable electricity, the whole process can dramatically cut emissions compared with traditional high‑temperature, high‑pressure chemical plants.
Engineering a Powerful Flowing Reactor
To move from lab demonstrations to meaningful production, the team had to tackle several engineering hurdles. They designed a highly active nickel–cobalt catalyst grown as thin nanosheets on a porous metal foam, giving the reaction abundant surface area. Just as important, they reshaped the tiny channels that carry liquid through the device, finding that slightly wider flow paths greatly improve how fast reactants and bubbles are transported. These optimized flow channels reduce resistance, keep gas from clogging the system, and allow the HMF solution to be converted almost completely in a single pass, instead of being recycled many times.
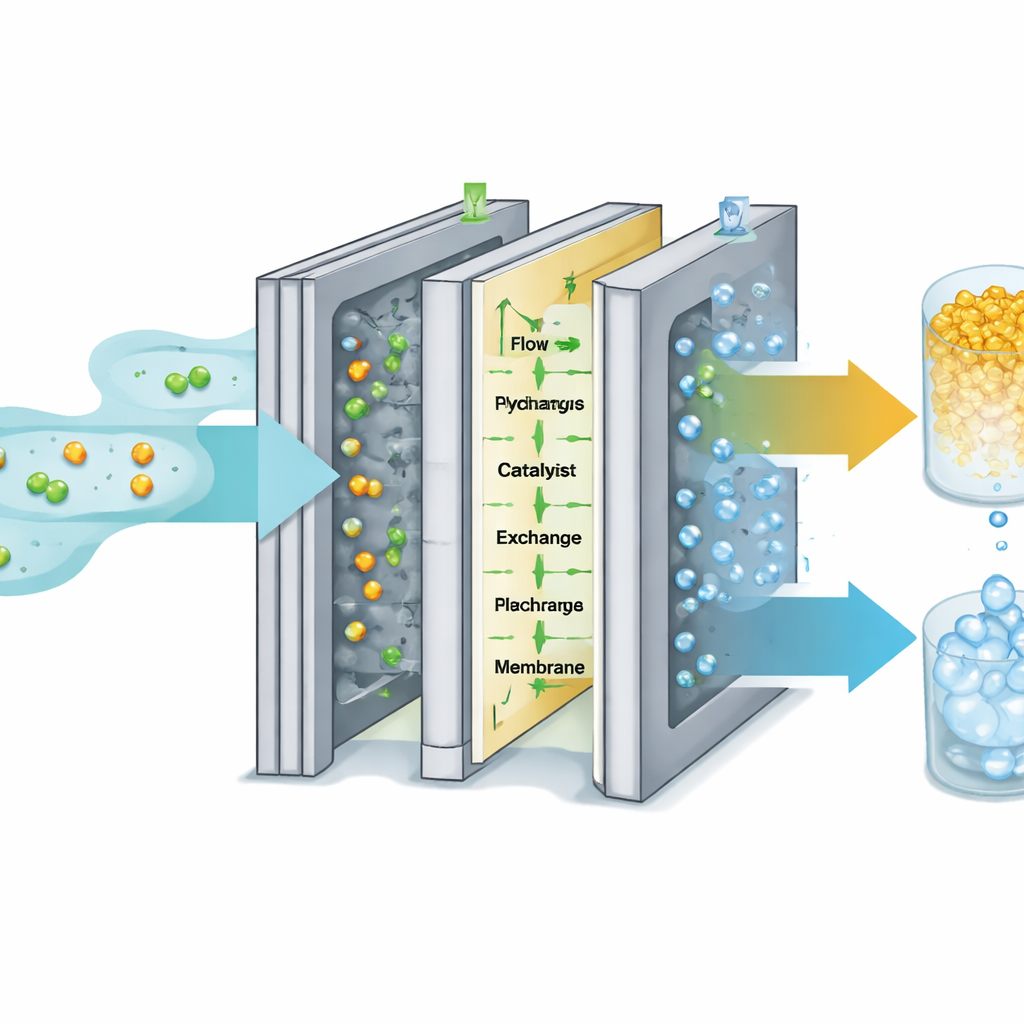
From Bench Setup to Industrial‑Style Stack
Building on these design choices, the researchers assembled stacks of multiple electrochemical cells connected in parallel, similar to the way battery modules are combined to power an electric car. Their hundred‑watt‑scale stack operates at industrially relevant conditions: high HMF concentrations, high current, and steady operation over more than 100 hours. Under these conditions, the system converts essentially all of the incoming HMF in one pass, achieving both high yield and high selectivity for FDCA while sustaining strong production rates. The same stack produces hydrogen with nearly perfect efficiency, adding value to the process.
Cleaning Up the Product and Counting the Impacts
High‑end plastics demand extremely pure ingredients, so the team integrated a water‑based purification train that uses modern membranes instead of harsh solvents. After neutralizing the alkaline reaction mixture, FDCA is concentrated and separated from impurities using nanofiltration and reverse osmosis, and then isolated as a bright white powder with 99.8% purity. When used to make PEF, this ultra‑pure FDCA yields clearer, higher‑quality plastic than material purified by simpler methods. The authors also performed detailed economic and environmental assessments. Their analysis suggests that, at realistic electricity prices and feedstock costs, the electrochemical process can undercut conventional fossil‑based routes, especially once the value of the hydrogen and salt by‑products is included. Life‑cycle modeling shows that pairing the system with renewable electricity can slash climate impacts by more than half compared with standard separation techniques, and by even more when cleaner power sources like wind are used.
What This Means for Everyday Materials
At its core, this work shows that it is possible to join plant‑derived ingredients, smart reactor design, and renewable electricity into a single, continuous process that turns biomass into a high‑purity plastic building block and clean hydrogen. While further scaling and industrial integration are still needed, the approach points toward future factories where bottles, fibers, and coatings are made from carbon that plants recently pulled from the air, powered by the sun and wind rather than oil and gas.
Citation: Liu, J., Chen, D., Tang, T. et al. Green chemical process for continuous production of high-purity 2,5-furandicarboxylic acid in anion exchange membrane flow electrolyzer. Nat Commun 17, 2099 (2026). https://doi.org/10.1038/s41467-026-68894-3
Keywords: biobased plastics, electrochemical synthesis, green hydrogen, flow electrolyzer, sustainable chemistry