Clear Sky Science · en
Potential-dependent polaron formation activates TiO2 for the hydrogen evolution reaction
Turning Rust-Like Materials into Clean Fuel Makers
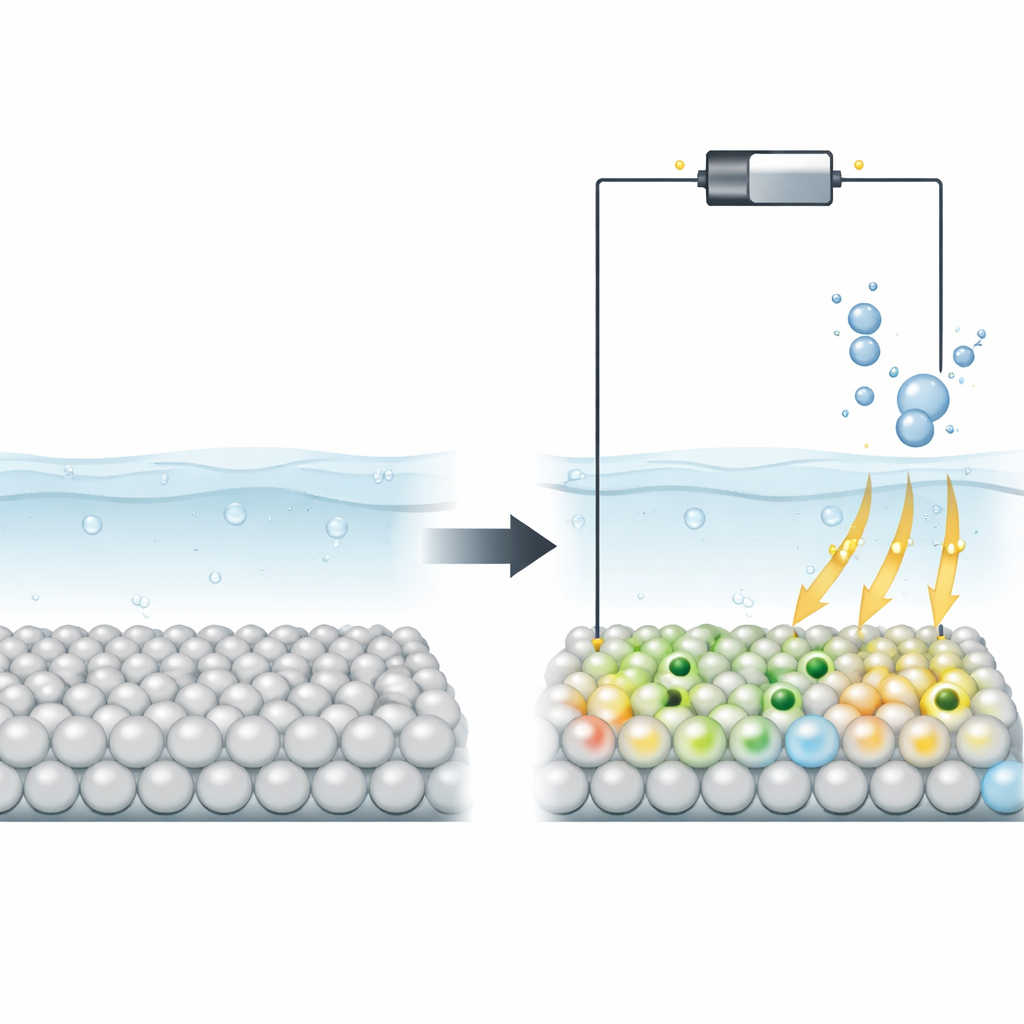
Splitting water into hydrogen fuel usually relies on expensive precious metals. This study shows that a common, stable oxide called titanium dioxide (TiO2) can be switched from sluggish to highly active simply by changing the electrical potential applied to it. The key is the creation of tiny, reversible charge pockets called polarons on the surface, which open up new, efficient pathways for making hydrogen gas.
Why Semiconductors Struggle and How Defects Help
Semiconductors such as TiO2 are attractive for clean energy because they are cheap, abundant, and already widely used in solar and photocatalytic technologies. Yet in their pristine form, they conduct electricity poorly and often bind reaction intermediates either too strongly or too weakly, making them mediocre catalysts. Researchers have long tried to fix this by baking in defects—missing atoms or distortions—during synthesis. These permanent changes can improve performance but are hard to control precisely, and it has remained unclear, at the atomic level, how such defects change the surface to speed up reactions like hydrogen evolution.
Creating Switchable Charge Pockets with Voltage
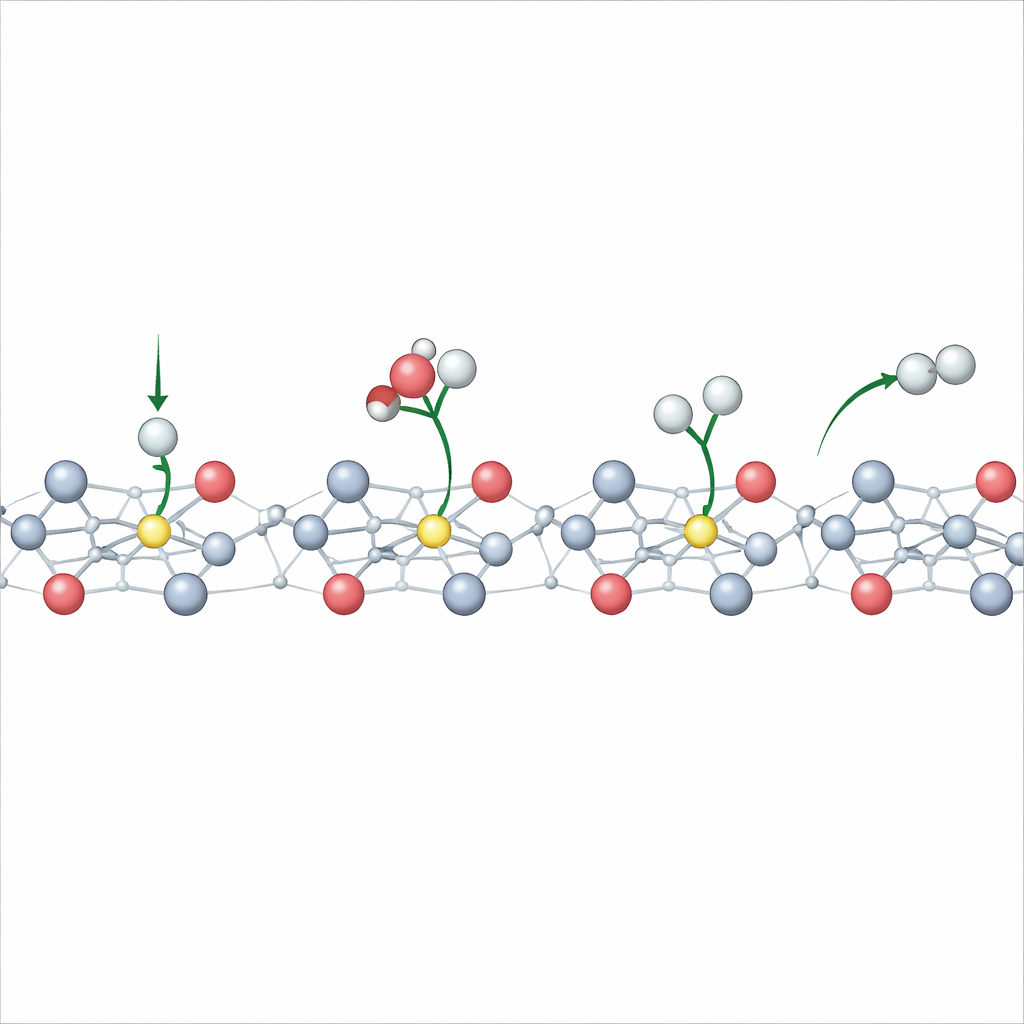
The authors propose a different strategy: use the operating voltage itself to sculpt the electronic structure of TiO2 in real time. When a sufficiently negative potential is applied, some titanium ions at the surface change from a higher charge state to a lower one and trap extra electrons in localized regions known as polarons. Using advanced constant-potential quantum calculations alongside in situ spectroscopic measurements, the team shows that these polarons form only under reducing conditions, are confined to the topmost atomic layer, and appear and disappear reversibly as the potential is cycled. This means the catalyst’s active surface can be dynamically tuned during operation, rather than fixed during fabrication.
Defects, Moving Charges, and Faster Hydrogen Release
The study goes further by examining what happens when the TiO2 surface already contains oxygen vacancies—missing oxygen atoms that are common in real materials. These vacancies encourage extra electrons to reside near specific titanium atoms, making it easier for polarons to form at less negative voltages. Simulations reveal that multiple polarons can align into chains and hop between neighboring atoms, greatly boosting surface conductivity. Experiments that track magnetic signals and charge transfer confirm that defective TiO2 accumulates more of these charge pockets and transports electrons more readily than pristine TiO2. As a result, electrodes with oxygen vacancies drive the hydrogen evolution reaction at much lower overpotentials and with much higher currents.
Rethinking Simple Rules for Reaction Energies
On metal electrodes, chemists often rely on tidy linear rules that connect reaction energies, activation barriers, and applied voltage. The authors show that these rules begin to break down on TiO2 once polarons enter the picture. The energy to bind hydrogen on the surface no longer changes smoothly with potential; instead, it kinks and bends as new polaron states switch on. Surprisingly, even though this simple voltage–energy link fails, a more general relationship that ties reaction barriers to reaction energies still holds. This means that, with careful accounting of when and where polarons appear, one can still predict how fast hydrogen will form on these semiconductor surfaces.
Designing Smarter, Tunable Catalysts
Taken together, the results paint a picture of TiO2 as a catalyst whose performance is not fixed by its composition alone but can be actively tuned by the operating potential. By combining built-in defects such as oxygen vacancies with voltage-controlled polaron formation, the surface can be turned into a dense network of highly active, conductive sites for hydrogen evolution. For a lay reader, the main message is that inexpensive semiconductor materials can be made to rival noble metals by learning how to "switch on" and steer these tiny charge pockets during operation, opening new routes to efficient, scalable hydrogen production and other clean electrochemical technologies.
Citation: Wu, T., Guo, X., Zhang, G. et al. Potential-dependent polaron formation activates TiO2 for the hydrogen evolution reaction. Nat Commun 17, 2104 (2026). https://doi.org/10.1038/s41467-026-68892-5
Keywords: hydrogen evolution reaction, titanium dioxide, polarons, semiconductor electrocatalysis, oxygen vacancies