Clear Sky Science · en
Precision estimates of longitudinal brain aging capture unexpected individual differences in one year
Why tiny brain changes matter
Our brains are always changing as we age, but those changes are usually so small from year to year that standard brain scans struggle to see them. This study shows that by taking many quick MRI scans in tightly packed “clusters,” scientists can measure how an individual person’s brain is aging over just one year. The results reveal that people of the same age—even people who appear cognitively healthy—can have remarkably different brain aging patterns, ranging from near-youthful stability to rapid decline.
Watching the brain grow older
Before zooming in on individuals, the researchers first used data from the UK Biobank, a huge long-term health study with tens of thousands of brain scans, to chart typical brain aging. They focused on the hippocampus, a structure crucial for memory that gradually shrinks with age and more quickly in Alzheimer’s disease. The charts confirmed that hippocampal loss speeds up in later life, and they showed enormous spread between people of the same age. That spread reflects a mix of true individual differences and plain measurement noise—one reason why it has been so hard to say how any single person’s brain is changing over short periods.
Taking many quick looks instead of one long one
To tackle the noise problem, the team tested a new strategy they call cluster scanning. Instead of relying on a single long MRI scan at each visit, they collected eight very rapid scans, each lasting just over a minute, on six separate days spread across a year. That gave 48 structural scans per participant. By pooling information across these repeated snapshots, they could mathematically cancel out much of the random fluctuation that plagues single scans. They also included closely spaced “test” and “retest” sessions at each of three timepoints so they could directly measure how much of the apparent change was real versus measurement error.
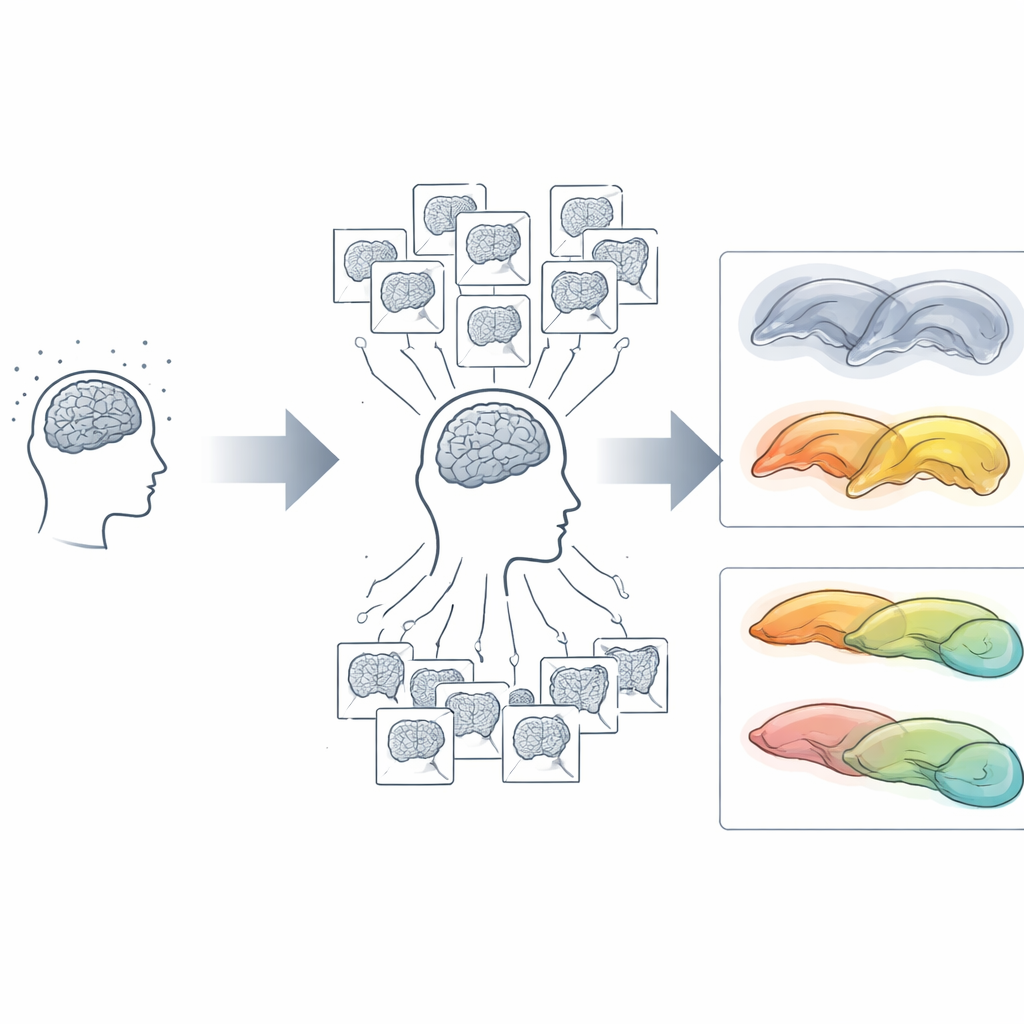
Sharper vision reveals hidden differences
With standard scanning, year-to-year hippocampal changes in most individuals were essentially unreadable: the noise was as large as, or larger than, the true change. Cluster scanning cut that error by about a factor of three and turned blurry estimates into stable, repeatable trajectories. As expected, younger adults in their 20s and 30s showed almost no hippocampal shrinkage over a year, while older adults with mild cognitive impairment, Alzheimer’s disease, or frontotemporal dementia showed the fastest loss. But among cognitively unimpaired older adults, the picture was surprisingly diverse. Some had modest, “typical” declines; others showed strikingly rapid or asymmetrical shrinkage; and a few seemed to maintain brain volume as if they were decades younger.
Stories hidden in individual brains
Detailed case studies made these differences vivid. One woman in her late 70s, rated cognitively normal at the start of the study, showed the steepest hippocampal loss of anyone in the sample, along with widespread brain shrinkage and enlarged fluid-filled spaces. During the study she underwent intensive cancer treatment, and within a year she was diagnosed with mild cognitive impairment, suggesting that cluster scanning had captured an early, rapid downturn in brain health. Another participant, initially diagnosed with mild cognitive impairment, showed almost no brain shrinkage and even a reduction in ventricle size, aligning with later biomarker tests that cast doubt on Alzheimer’s as the cause of her symptoms. Other individuals showed strongly one-sided (right greater than left) decline suggestive of early, localized disease processes, while one man in his early 70s displayed remarkably preserved structure across measures, with brain changes resembling those of a much younger adult.
New tools for personalizing brain aging
By turning many fast scans into precise year-long trajectories, cluster scanning opens the door to studying brain aging at the level of the individual rather than the average. The method could make clinical trials more sensitive, allowing researchers to detect whether a treatment is slowing brain atrophy in smaller groups and shorter timeframes. It may also help clinicians monitor how a patient’s brain responds to illness, medication, or lifestyle changes. The central message for lay readers is that brain aging is far from uniform: with better measurement, we can see that some people’s brains decline quickly, others remain stable, and these paths can change rapidly within a year. Cluster scanning provides a powerful new lens for understanding—and eventually guiding—those personal brain aging journeys.
Citation: Elliott, M.L., Du, J., Nielsen, J.A. et al. Precision estimates of longitudinal brain aging capture unexpected individual differences in one year. Nat Commun 17, 2401 (2026). https://doi.org/10.1038/s41467-026-68886-3
Keywords: brain aging, MRI, Alzheimer’s disease, longitudinal study, neurodegeneration