Clear Sky Science · en
Inhibition of TGF-β signaling in microglia stimulates hippocampal adult neurogenesis and reduces anxiety-like behavior in adult mice
Why brain immune cells matter for mood and memory
Most of us think of immune cells as patrolling the body, not shaping our thoughts and feelings. This study challenges that view by showing that microglia—immune cells that live in the brain—can powerfully influence the birth of new neurons in the hippocampus, a region important for memory and emotion. By turning off a specific signaling pathway in microglia in adult mice, the researchers boosted the production and survival of new neurons and saw measurable changes in anxiety-like behavior.
Brain’s quiet nursery for new neurons
In adult mammals, new neurons are still born in a few specialized “nurseries.” One of the most important is the subgranular zone of the hippocampus, which helps separate similar memories, supports learning, and contributes to mood regulation. Under normal conditions, many of these newborn cells die before they fully mature. Their numbers can rise or fall in response to exercise, enriched environments, stress, stroke, or epilepsy. Microglia constantly survey this environment, but whether their reactive, “inflamed” state helps or harms this adult neurogenesis has been unclear.
Switching off a key brake in microglia
The team focused on a signaling molecule called TGF-beta, which usually keeps microglia in a calm, homeostatic state. Using genetically engineered mice, they selectively disabled TGF-beta signaling in microglia during adulthood—either by removing the TGF-beta ligand itself or the receptors (ALK5 or TβRII) that sense it. This pushed microglia into a reactive, pro‑inflammatory profile without causing widespread neuron loss. In this state, microglia increased in number and changed shape, indicating activation, but neighboring support cells called astrocytes remained largely unaffected.
More new neurons, better survival, and altered anxiety
When TGF-beta signaling was shut down in microglia, the hippocampal nursery produced a surge of immature neurons marked by the protein DCX. This spike appeared about three weeks after the genetic switch and led to a lasting increase in fully mature neurons weeks later. Careful tracking of dividing cells showed that the main effect was improved survival of newborn neurons, not simply faster cell division. Removing microglia entirely with a drug did not mimic this effect, suggesting that it is not the absence of microglia that matters, but the specific reactive profile created when TGF-beta is silenced. Behaviorally, mice with TGF-beta–deficient microglia spent more time in the open arms of elevated mazes, a common sign of reduced anxiety-like behavior or disinhibition. These changes faded when microglia later returned to a more normal state in some models, but persisted in others where microglia stayed reactive.
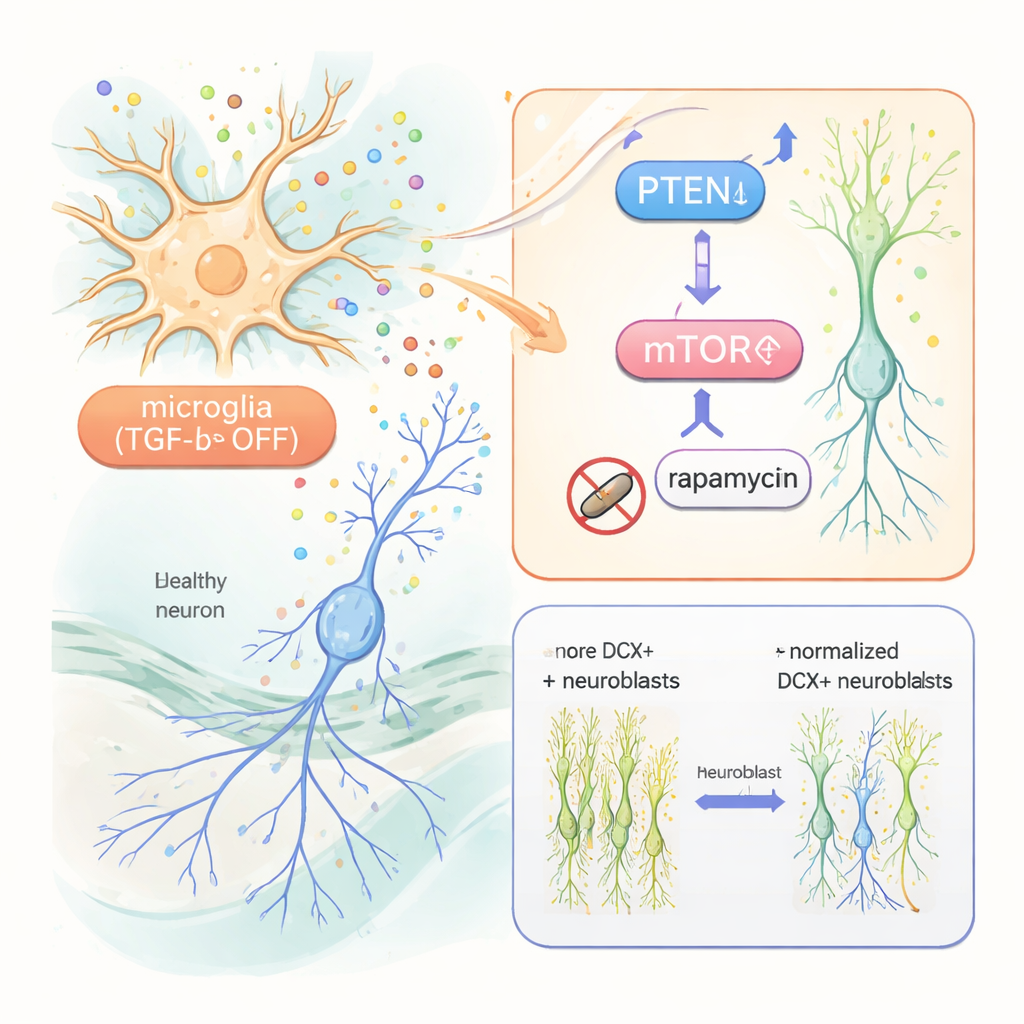
Uncovering the pathway inside newborn neurons
To understand how altered microglia talk to newborn neurons, the researchers used single-cell RNA sequencing on hippocampal cells. They found broad changes in gene activity not only in microglia but also in immature neurons. Key among these was a reduction in PTEN, a protein that normally restrains cell growth, and signs of increased activity in the mTOR pathway, which promotes cell survival and growth. Interestingly, well-known growth factors such as IGF‑1 and TNF‑alpha, though elevated in reactive microglia, turned out not to be required: even when these were genetically removed, the boost in neurogenesis remained. In contrast, when the team treated mice with rapamycin, a drug that blocks mTOR, the excess of immature neurons and their exuberant branching returned to normal. Newborn neurons also showed higher levels of a downstream mTOR marker, which rapamycin reduced.
What this could mean for brain health
Taken together, the findings suggest that when TGF-beta signaling in microglia is turned off, these cells adopt a reactive state that encourages the survival and integration of newborn hippocampal neurons via a PTEN–mTOR pathway inside those neurons. This, in turn, is linked to lower anxiety-like behavior in mice, though it may also come with changes in learning and memory. For a layperson, the key message is that the brain’s own immune cells can fine‑tune how many new neurons join our memory circuits—and that carefully nudging this system could one day help boost beneficial neurogenesis or rein in harmful, excessively sprouting neurons in conditions like epilepsy or after brain injury.
Citation: Ware, K., Peter, J., Yazell, J. et al. Inhibition of TGF-β signaling in microglia stimulates hippocampal adult neurogenesis and reduces anxiety-like behavior in adult mice. Nat Commun 17, 1440 (2026). https://doi.org/10.1038/s41467-026-68885-4
Keywords: adult neurogenesis, microglia, hippocampus, anxiety behavior, TGF-beta signaling