Clear Sky Science · en
PRMT3-mediated post-translational adaptation to fasting regulates metabolic flexibility
Why meal timing and body fuel matter
Many people try fasting or time-restricted eating to lose weight or improve blood sugar, but the results can be hit-or-miss. This study looks under the hood of our metabolism to find out why. Focusing on belly fat in mice and on human samples, the researchers uncover a molecular switch that helps fat tissue decide whether to burn or store fuel, and show how both meal timing and a drug can flip that switch toward better health.
A flexible engine inside our fat
Our bodies work best when they can smoothly switch between burning fat during fasting and burning sugar after meals. This “metabolic flexibility” is often lost in obesity, making it harder to handle swings in energy supply and demand. The team studied visceral white fat—the deep belly fat strongly linked to diabetes and heart disease—and found that specific chemical tags on proteins, called methyl groups, change with feeding and fasting. In both mice and humans, two such tags, MMA and ADMA, build up in visceral fat as body weight and body mass index rise, suggesting they are markers of a sluggish, less adaptable metabolism.
A feeding signal that stiffens metabolism
The researchers homed in on an enzyme in fat cells called PRMT3, which adds these methyl tags. In mice, PRMT3 levels in visceral fat rise during the night when animals eat and fall during the day when they fast, closely tracking the pattern of MMA and ADMA. This rhythm depends on insulin and a key signaling protein, AKT: when food or injected insulin activates AKT, PRMT3 becomes switched on and more heavily modified, which in turn boosts the amount of methylated proteins. Blocking AKT or directly inhibiting PRMT3 quickly flattens this methylation signal, even mimicking the effect of a much longer fast.
Rewiring fat cells from storage to burning
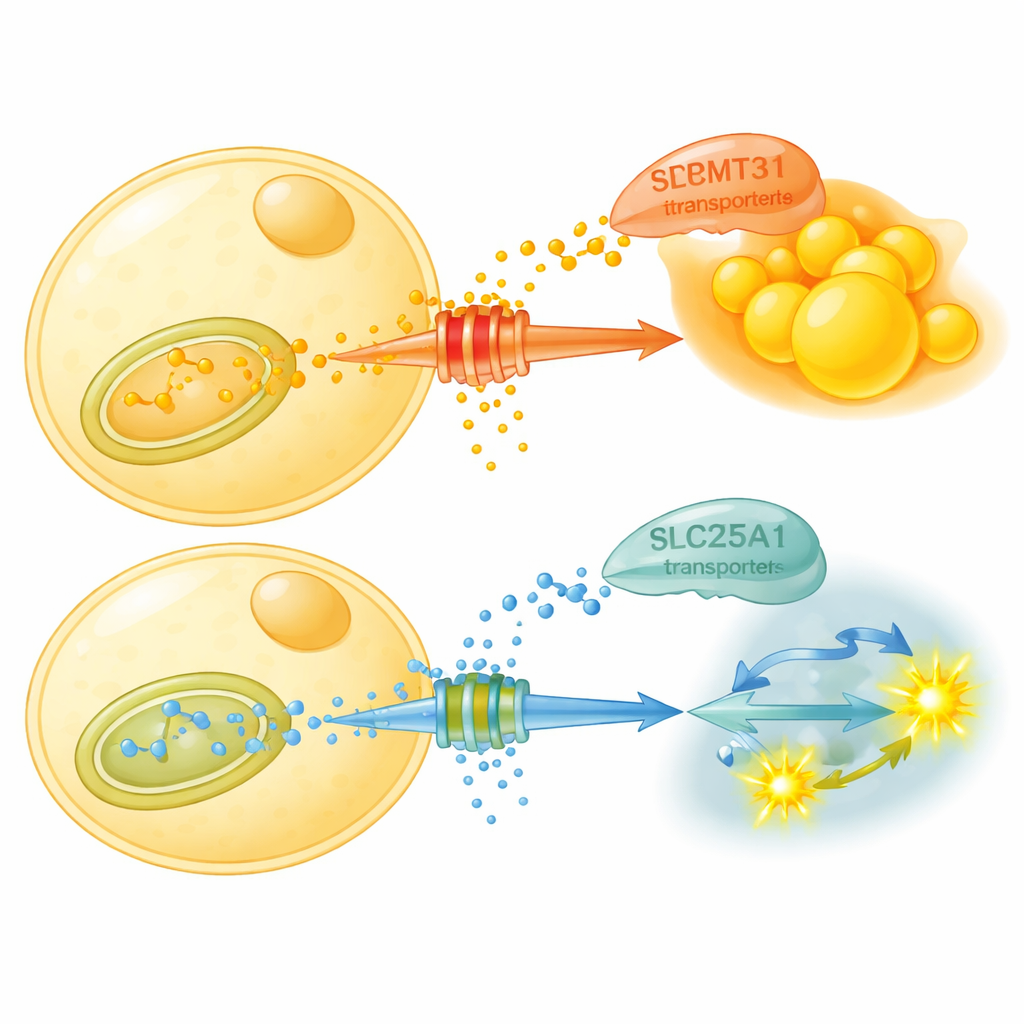
What does this chemical tagging actually do? The study shows that PRMT3 stabilizes a transporter protein called SLC25A1 in the mitochondria—the cell’s energy factories. SLC25A1 moves citrate, a key metabolic intermediate, out of mitochondria into the cell fluid where it feeds new fat production. PRMT3 methylates two specific arginine sites on SLC25A1, making the transporter more stable and encouraging citrate export and fat building. When PRMT3 is blocked, SLC25A1 levels drop, citrate export falls, and fat cells shift toward breaking down sugar instead of making more fat. In mice on a high-fat diet, a PRMT3-blocking drug reduces body fat, improves blood sugar control, and increases a measure called the respiratory exchange ratio, showing that the animals more easily switch to burning carbohydrates.
Fasting schedules and gene tweaks point to the same pathway
The researchers then asked whether common fasting patterns use this same switch. Under a 16:8 time-restricted feeding schedule—16 hours of fasting and 8 hours of eating—mice lost weight and handled glucose better without eating less overall. Their visceral fat showed lower levels of PRMT3, SLC25A1, and methylated proteins, and their fuel use became more flexible, especially at night when they normally eat. Remarkably, giving the PRMT3-blocking drug at specific times of day produced many of the same benefits as the 16:8 schedule. Genetically removing SLC25A1 only in fat cells also protected mice from diet-induced high blood sugar and fatty liver, again by boosting sugar breakdown and energy use in fat, even without shrinking fat mass.
What this means for future therapies
Taken together, the findings suggest that deep belly fat contains a timing-sensitive switch, controlled by insulin, AKT, PRMT3, and SLC25A1, that determines whether our bodies stay metabolically nimble or become locked into a storage mode. Time-restricted eating appears to nudge this switch toward flexibility by dialing down PRMT3 and its downstream effects. A drug that inhibits PRMT3 can reproduce many of these advantages in obese mice, hinting at a future medicine that makes the metabolic benefits of fasting more accessible. For everyday readers, the message is that when we eat, not just what we eat, can reshape how our fat tissue handles fuel—with important consequences for weight, blood sugar, and long-term health.
Citation: Huang, Z., Liu, X., Chen, X. et al. PRMT3-mediated post-translational adaptation to fasting regulates metabolic flexibility. Nat Commun 17, 2264 (2026). https://doi.org/10.1038/s41467-026-68883-6
Keywords: metabolic flexibility, time-restricted feeding, visceral fat, protein methylation, glucose metabolism