Clear Sky Science · en
Discover the maze-like network for glabridin biosynthesis
Why licorice holds more than just sweet memories
Licorice root has long been prized in traditional remedies and luxury skincare for a natural compound called glabridin, known for its antioxidant, anti-inflammatory, and skin-brightening effects. Yet getting glabridin today mostly means stripping it from wild licorice plants, a slow and wasteful process that can damage fragile dryland ecosystems. This study uncovers in molecular detail how licorice makes glabridin and shows how that intricate chemistry can be rebuilt inside baker’s yeast, pointing the way toward more sustainable production of valuable plant-based ingredients.
From simple building blocks to a chemical maze
Plants produce an astonishing variety of flavonoids—over 9,000 different molecules—from just a handful of basic starting compounds. Much of this diversity comes from “tailoring” steps that tack on or remove small chemical groups after the core scaffold is built. For glabridin, the authors first used computational tools to work backward from its structure and search through known enzyme reactions. They mapped all plausible routes from a common amino acid, L‑phenylalanine, to glabridin, then trimmed this huge network using real metabolic data from licorice roots. The result was a maze of 13 possible pathways with many branch points, hinting that licoridin’s biosynthesis is not a simple linear assembly line but a flexible network with multiple routes to the same end product. 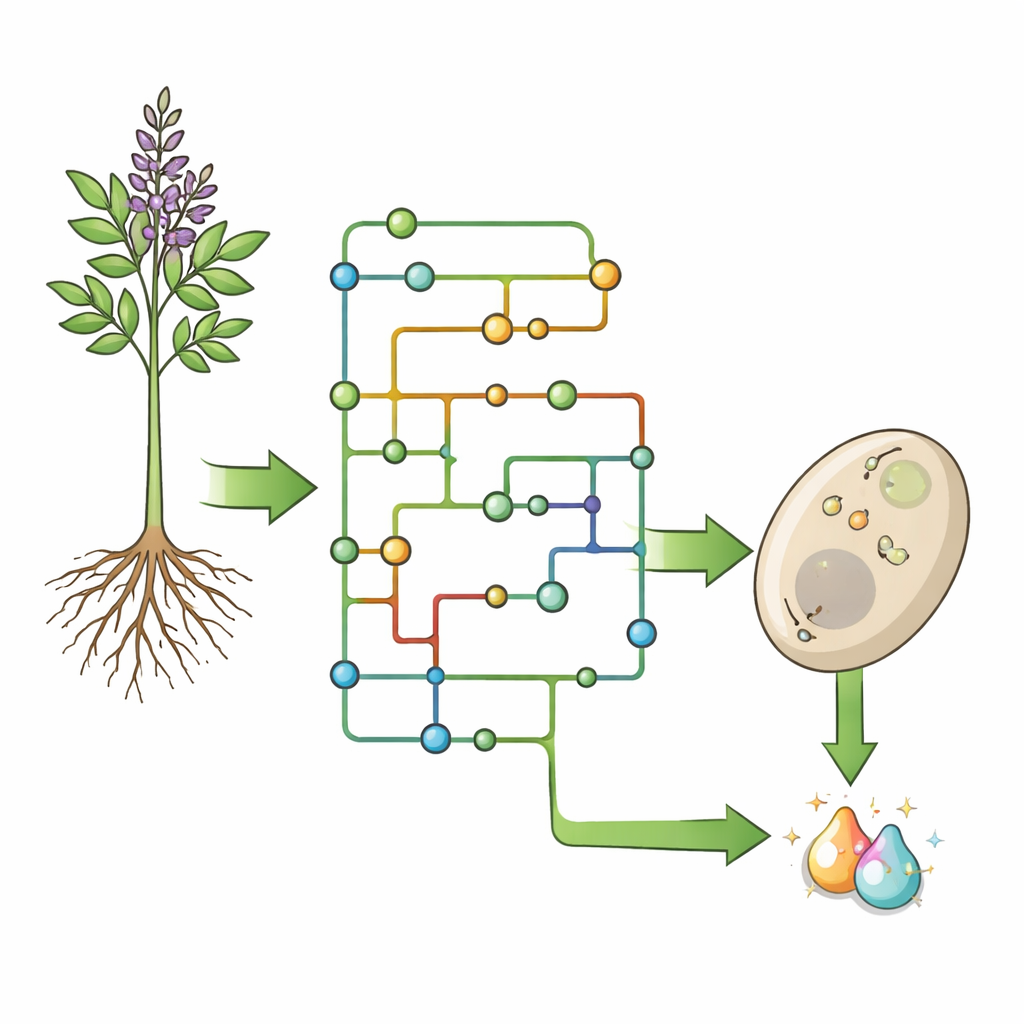
Hunting down the key molecular workers in licorice
To find which enzymes actually build glabridin in the plant, the team assembled a chromosome-level genome for Glycyrrhiza glabra and paired it with 183 transcriptomes—snapshots of which genes are active in different organs, species, seasons, and growth stages. By combining sequence similarity, evolutionary relationships, and co-expression patterns, they narrowed thousands of genes down to a focused toolkit: seven candidate reductases, eighteen prenyltransferases, thirty-nine oxidative cyclases, and six demethylases. Many of these genes cluster on specific chromosomes and are most active in roots, where glabridin accumulates. Comparing three related licorice species showed that G. glabra, the main natural source of glabridin, tends to express these key enzymes at higher levels, matching the much higher glabridin content seen in its roots.
Rebuilding the pathway one reaction at a time
The researchers then tested each candidate enzyme in yeast and in purified form to see what it actually does. They identified a powerful reductase (GgPTR1) that opens a ring in an isoflavan precursor, a specialized prenyltransferase (GgPT1) that adds a greasy side chain, an oxidative cyclase (GgOC1) that closes a new ring, and a versatile plant demethylase (GgDMT1) that can remove methyl groups from several intermediates. Together, these four steps convert the pterocarpan medicarpin into glabridin through multiple interlinked routes. A striking feature of the network is a repeated “protection–deprotection” cycle: methylation helps steer reactive intermediates down efficient paths and improves how they fit into enzymes, and later demethylation restores the final active form. Spatial separation within the cell—some enzymes in the endoplasmic reticulum, others in the cytoplasm—and shifts in gene activity over the season further tune when and where each step occurs.
Turning yeast into a mini licorice factory
Armed with this enzyme set, the team engineered baker’s yeast to make glabridin from simple sugar. First they built a “core module” of fourteen enzymes that converts glucose into medicarpin, the central scaffold. They then added a “tailoring module” with the licorice reductase, prenyltransferase, and oxidase, plus either the plant demethylase GgDMT1 or a fungal demethylase NhPDA1. Instead of forcing a single rigid route, they allowed enzyme promiscuity—the ability to act on multiple intermediates—to create a ladder-like network of parallel branches. Experiments and computer modeling both showed that this multi-route design is more robust and productive than a streamlined single-route pathway, in part because it reduces the loss of intermediates that would otherwise leak out of the cell.
What this means for skincare and sustainable chemistry
By fully charting glabridin’s biosynthetic maze and rebuilding it in yeast, the authors provide a blueprint for making this high-value cosmetic ingredient without uprooting large quantities of wild licorice. Their work also reveals a broader principle: plant pathways for specialized molecules may rely on reversible “on–off” chemical decorations and redundant branches to stay flexible and resilient. Harnessing these maze-like networks in microbes could make it easier to produce not only glabridin, but also many other complex plant natural products, supporting greener manufacturing and reducing pressure on vulnerable plant species. 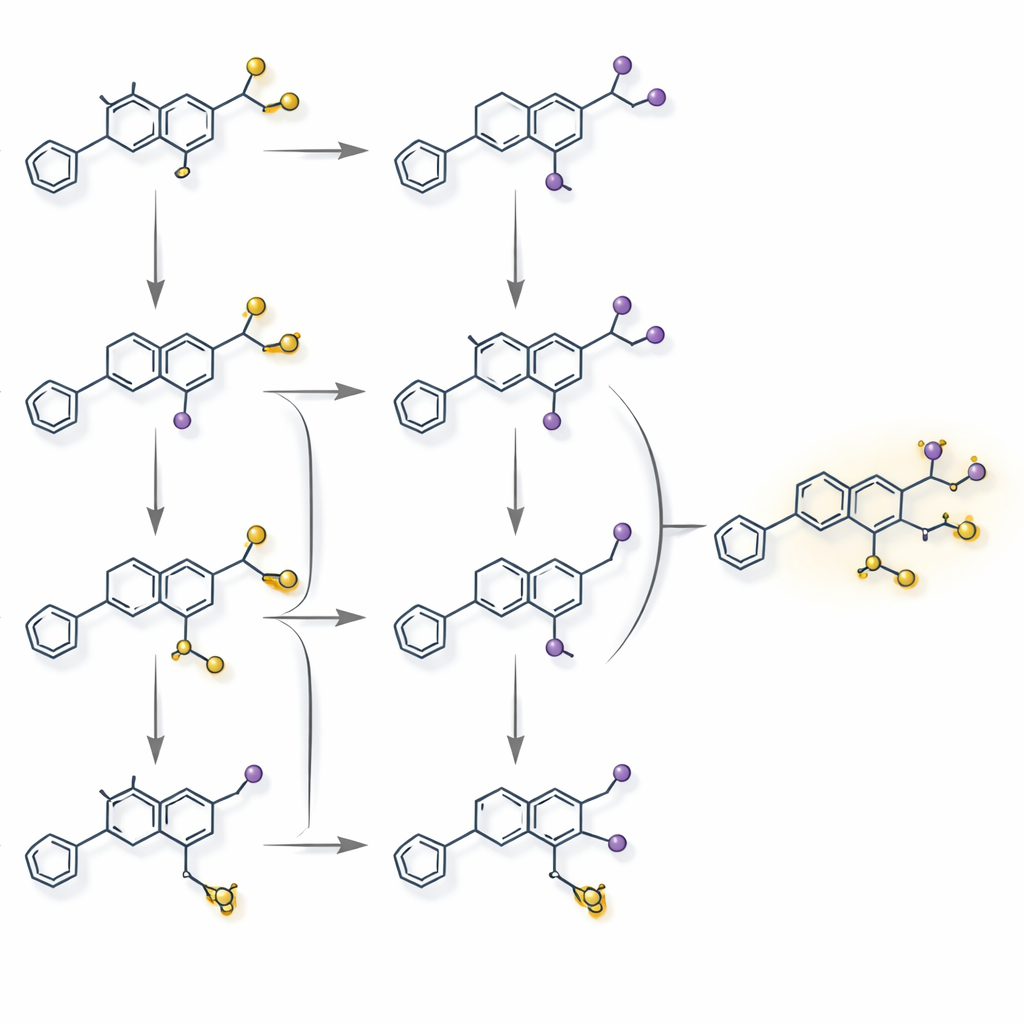
Citation: Zhang, Z., Li, W., Meng, F. et al. Discover the maze-like network for glabridin biosynthesis. Nat Commun 17, 2215 (2026). https://doi.org/10.1038/s41467-026-68881-8
Keywords: glabridin, licorice, microbial biosynthesis, metabolic engineering, flavonoids