Clear Sky Science · en
A DNA-based nanodevice senses purinergic signaling and drives an immune switch for resolving inflammation
Turning Down the Body’s Alarm System
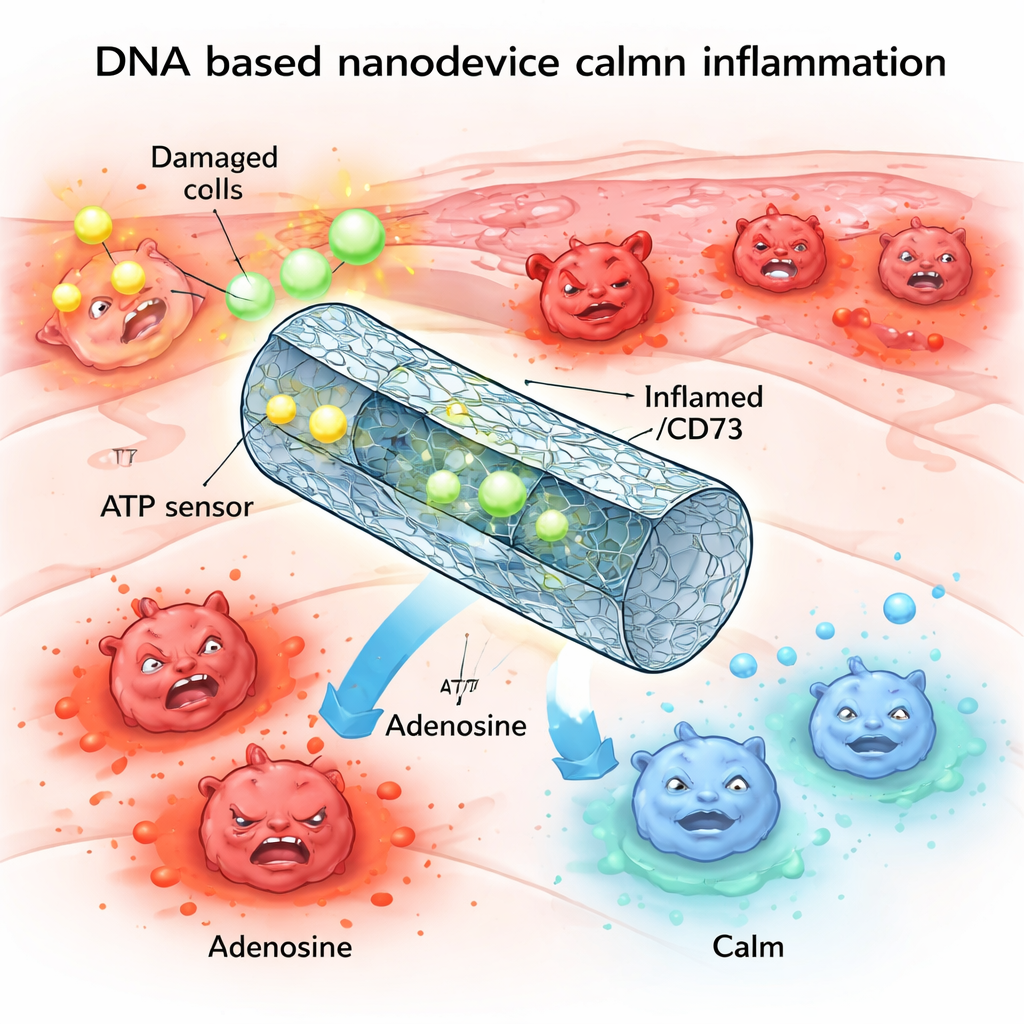
When our tissues are injured, cells spill a burst of chemical “alarm signals” that call in immune cells to fight infection and clear debris. This response is vital, but if the alarm blares too loudly or for too long, it can drive damaging inflammation in the lungs, kidneys, and other organs. This study describes a tiny, programmable DNA-based device that can sense when the alarm signal is too strong and quietly dial it back, helping the immune system switch from attack mode to healing mode.
A Chemical Alarm That Can Go Too Far
One of the body’s key alarm molecules is ATP, best known as the “energy currency” inside cells. During injury or stress, large amounts of ATP leak out of damaged cells into the surrounding space. There, this extracellular ATP acts as a distress signal, binding to receptors on immune cells and pushing them toward a highly inflammatory state. Under healthy conditions, enzymes on cell surfaces rapidly break down excess ATP into adenosine, a related molecule that has a calming, anti-inflammatory effect. But in many chronic or severe inflammatory diseases, this breakdown machinery is impaired, leaving ATP levels high, adenosine low, and tissues stuck in a cycle of damage.
Building a Smart DNA Tube
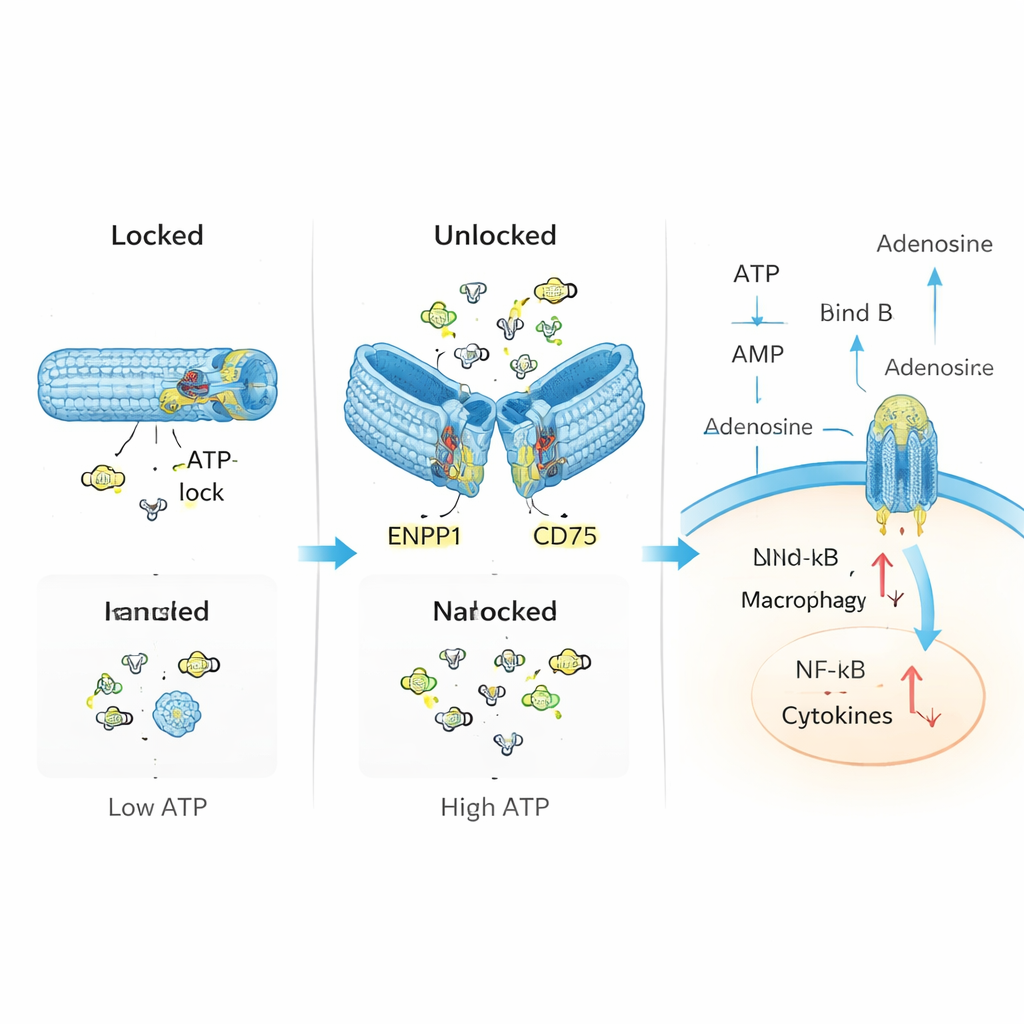
The researchers set out to imitate and repair this natural control system using DNA nanotechnology. They built a hollow tube from DNA, folded much like origami, and equipped it with two main components: “sensors” that recognize ATP, and enzymes that convert ATP into adenosine. In its resting, locked state, the tube is held closed by short DNA segments that double as ATP sensors. When ATP levels rise above a certain threshold typical of injured tissues, ATP binds to these segments, causing the tube to open and expose the enzymes ENPP1 and CD73 inside. These then chew up the excess ATP and produce adenosine, gently shifting the local chemical balance from danger to resolution.
Proving the Switch Works
In controlled lab tests, the team confirmed that their DNA tube stays closed at low ATP levels but reliably opens when ATP becomes abundant, much like an emergency valve. Once unlocked, the device steadily converts ATP into intermediate products and finally into adenosine, without noticeably damaging the enzymes’ activity. In immune cells exposed to high ATP, this nanodevice reduced toxic by-products in mitochondria, lowered activation of inflammatory pathways such as NF-κB and the NLRP3 inflammasome, and boosted signals linked to adenosine’s calming actions. In other words, the device did more than mop up ATP; it actively rewired the cells’ behavior from inflammatory to regulatory.
Hitchhiking to Sites of Injury
Designing a smart switch is one thing; getting it to the right place in the body is another. To solve this delivery problem, the researchers attached their DNA tubes to the surface of circulating monocytes, a type of white blood cell that naturally homes to inflamed tissues. Using a cholesterol-linked DNA anchor, they “parked” large numbers of nanodevices on each cell’s membrane, where the devices remained mostly on the surface rather than being swallowed by the cell. In mouse models of acute lung and kidney injury, these decorated monocytes accumulated in damaged organs far more efficiently than free-floating devices, and the nanodevices still responded specifically to high ATP in injured tissue.
Helping Tissues Heal
In mice with chemically induced lung or kidney injury, treatment with the DNA device reduced tissue damage, immune cell buildup, and inflammatory messenger molecules. The monocyte-carried version worked best, offering stronger protection than either the free device or the enzymes alone. Deeper genetic and metabolic analyses of lung tissue showed that this approach not only dampened inflammatory genes but also helped restore disturbed energy metabolism in cells. Importantly, the devices showed low toxicity and did not trigger harmful immune reactions in healthy animals.
Why This Matters for Future Therapies
For a non-specialist, the key message is that this work demonstrates a programmable, molecular “thermostat” for inflammation. Built from DNA and natural enzymes, the nanodevice senses when a danger signal is too strong and automatically converts it into a calming signal, helping immune cells stand down and tissues repair. While still at an early, experimental stage, this strategy suggests a new class of treatments that do not simply block immune pathways everywhere, but instead locally rebalance chemical signals at sites of injury, potentially offering precise control over harmful inflammation in many diseases.
Citation: Li, W., Liu, S., Zhou, X. et al. A DNA-based nanodevice senses purinergic signaling and drives an immune switch for resolving inflammation. Nat Commun 17, 2030 (2026). https://doi.org/10.1038/s41467-026-68879-2
Keywords: DNA nanodevice, extracellular ATP, inflammation, adenosine signaling, nanomedicine