Clear Sky Science · en
Anti-TLR2 immunotherapy modulates neuron-to-oligodendrocyte propagation of α-synuclein in mouse and human models
Why this research matters
Multiple system atrophy (MSA) is a rare but fast-moving brain disorder that combines Parkinson-like movement problems with balance and autonomic failures, such as blood-pressure drops. Doctors can ease symptoms, but they currently cannot slow the disease itself. This study uncovers how a misfolded protein spreads between brain cells to damage the “wiring insulation” of the brain, and shows that a targeted antibody treatment can interrupt this process in animal and cell models. The work points to a concrete, drug-like strategy that might one day change the course of MSA rather than just treating its symptoms.
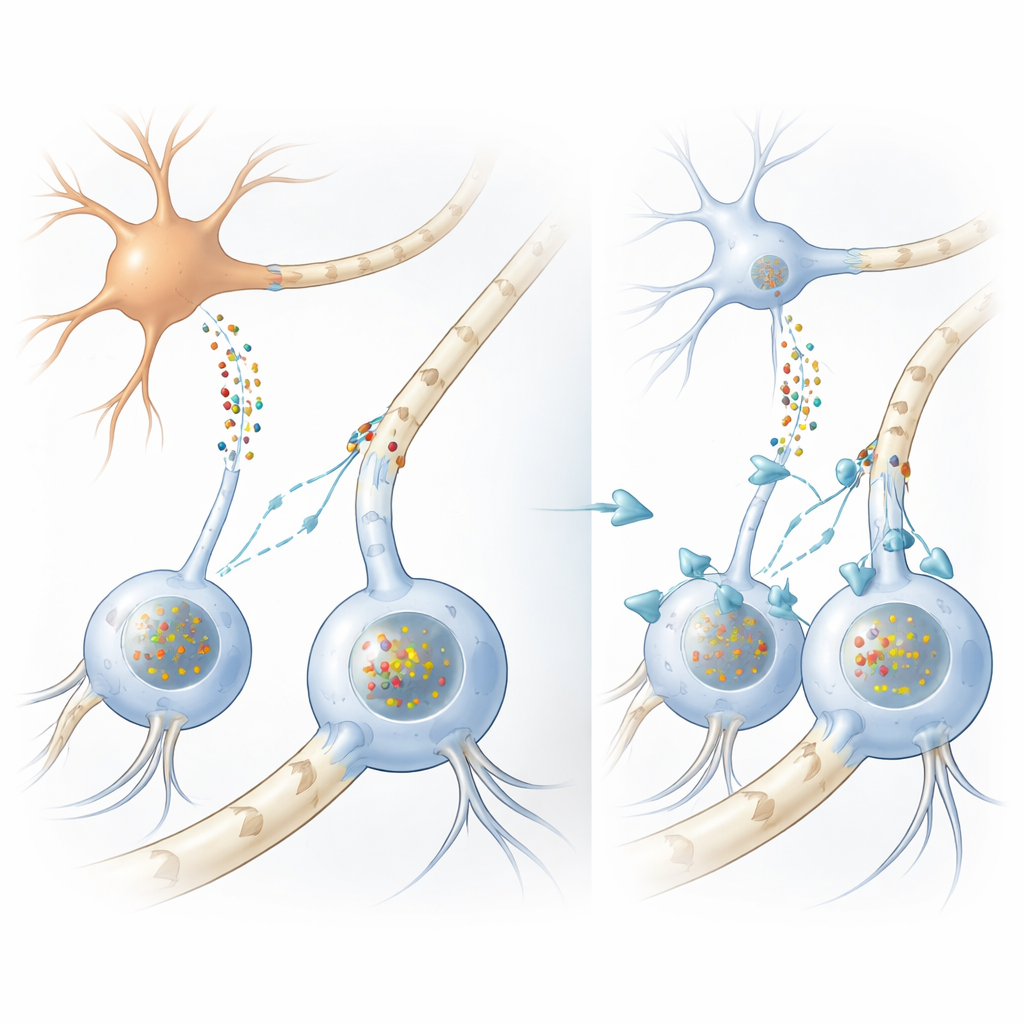
How brain wiring goes wrong
In many movement disorders, including Parkinson’s disease, clumps of a protein called alpha-synuclein build up inside nerve cells. In MSA, however, the most striking clumps form inside support cells called oligodendrocytes, which normally wrap nerve fibers with fatty myelin sheaths that speed electrical signals. Oddly, oligodendrocytes make very little alpha-synuclein themselves, leaving a long-standing puzzle: where do the massive protein deposits in these cells come from? The authors first confirmed, using human brain samples and large-scale RNA analyses, that oligodendrocytes indeed produce far less alpha-synuclein than neurons, strengthening the idea that the harmful protein must arrive from outside.
Protein passed from neuron to support cell
To test this, the team built several complementary models. In dishes, they grew human oligodendrocyte-like cells from neural stem cells and bathed them in liquid collected from neuron-like cells that were engineered to release large amounts of alpha-synuclein. The support cells took up this protein and developed clumps closely resembling the glial inclusions seen in MSA brains, complete with the same chemical tags and helper proteins. When the researchers looked in a mouse strain that produces human mutant alpha-synuclein only in neurons, they again found human protein clumps inside oligodendrocytes in white matter, even though those cells did not express the human gene. Together, these experiments show that alpha-synuclein can move from neurons into oligodendrocytes and form disease-like inclusions there.
The gate on the cell surface
Next, the scientists asked how the protein gains entry into oligodendrocytes. Prior work had identified the immune sensor Toll-like receptor 2 (TLR2) on cell surfaces as a docking site for alpha-synuclein in neurons and microglia. Mining gene-expression datasets from MSA brains, the team found that oligodendrocytes in patients carried unusually high levels of TLR2 compared with controls, and that higher TLR2 was linked to lower levels of myelin-related genes such as myelin basic protein. This relationship did not appear in several independent Parkinson’s datasets, hinting that oligodendrocyte sensitivity to alpha-synuclein via TLR2 is a distinctive feature of MSA rather than a general trait of all synuclein disorders.
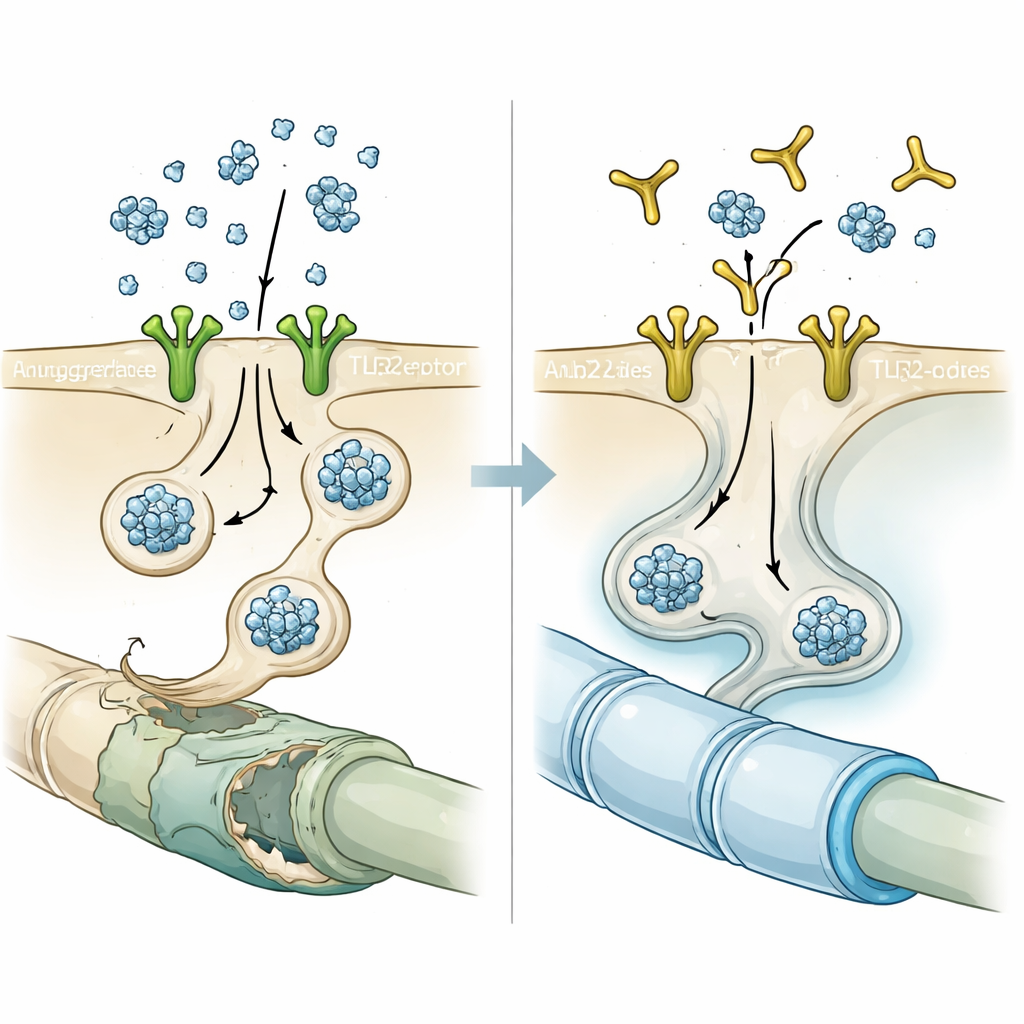
Blocking the gate with an antibody
Armed with this clue, the researchers tested NM-101, an antibody designed to latch onto TLR2 and prevent its activation. In cell cultures, briefly pre-treating oligodendrocytes with NM-101 before adding neuron-derived alpha-synuclein sharply reduced the number and intensity of inclusion-like clumps. In mice that either overproduced neuronal alpha-synuclein or received injections of pre-formed alpha-synuclein fibrils, weekly NM-101 infusions lowered aggregated protein in white matter, calmed inflammatory responses from microglia and astrocytes, and reduced activation of an inflammatory enzyme called caspase-1 inside oligodendrocytes. Treated animals lived longer and performed better on motor tests, suggesting that the antibody’s protective effects were functionally meaningful, not just microscopic curiosities.
Rescuing damaged insulation
Because oligodendrocytes are the brain’s myelin makers, the team examined whether alpha-synuclein transfer harmed myelin, and whether blocking TLR2 could help. Single-cell RNA sequencing of human-derived oligodendrocytes exposed to neuron-conditioned alpha-synuclein revealed broad shifts away from a mature, myelin-producing state and toward a more immature, progenitor-like profile, with many key myelin genes turned down. Parallel gene-expression studies of laser-captured oligodendrocytes from MSA patients and from the mouse model showed a shared signature: reduced expression of genes involved in forming and maintaining myelin. Under the electron microscope, white matter in alpha-synuclein mice displayed thinner, disorganized myelin sheaths. NM-101 treatment reversed many of these changes, thickening myelin, restoring levels of myelin proteins, and normalizing the expression of genes needed for oligodendrocyte maturation.
What it all means for future treatments
The study supports a clear storyline: in MSA, alpha-synuclein produced by neurons can spread into oligodendrocytes through TLR2 on their surface, where it builds up, triggers inflammation, derails the cells’ developmental program, and erodes the myelin coating of brain wiring. By blocking TLR2 with a targeted antibody, the researchers were able to interrupt this chain of events in mice and human cell models, reducing toxic inclusions, calming inflammation, repairing myelin, and improving survival and movement. While NM-101 itself still needs rigorous testing in people, the work establishes TLR2-dependent protein transfer as a central driver of MSA-like pathology and highlights anti-TLR2 immunotherapy as a promising strategy to slow or prevent this devastating disease.
Citation: Bae, EJ., Ham, S., Jeong, Y.W. et al. Anti-TLR2 immunotherapy modulates neuron-to-oligodendrocyte propagation of α-synuclein in mouse and human models. Nat Commun 17, 2175 (2026). https://doi.org/10.1038/s41467-026-68870-x
Keywords: multiple system atrophy, alpha-synuclein, oligodendrocytes, myelin damage, immunotherapy