Clear Sky Science · en
NKG2D upregulation sensitizes tumors to combined anti-PD1 and anti-VEGF therapy and prevents hearing loss
Why this research matters for people with hearing loss
Some people inherit a condition called NF2-related schwannomatosis, which causes benign tumors to grow on the nerves of the inner ear and often leads to gradual, permanent hearing loss. Current treatments can shrink tumors but may damage hearing or stop working over time. This study explores a new drug combination that not only slows tumor growth in laboratory models but also helps protect hearing, pointing toward gentler and longer-lasting options for patients.
Understanding the tumors behind the silence
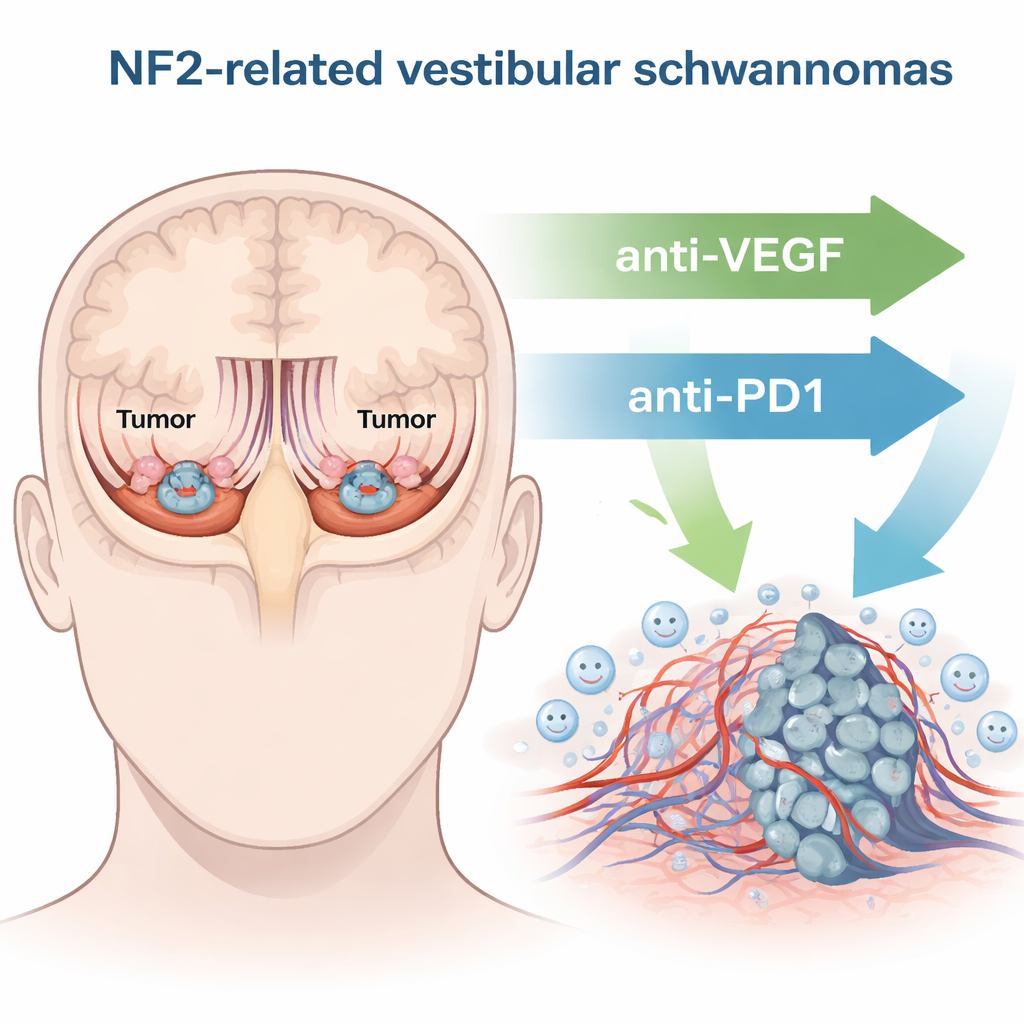
In NF2-related schwannomatosis, tumors called vestibular schwannomas form on the balance and hearing nerve inside the skull. As these tumors enlarge, they can damage the delicate nerve fibers that carry sound signals to the brain and, in severe cases, press on the brainstem. Surgery and radiation can save lives and remove or control tumors, but both carry real risks: worsening hearing, dizziness, and facial nerve weakness. A drug called bevacizumab, which blocks a growth signal for blood vessels (VEGF), is sometimes used to shrink these tumors and improve hearing, yet only about a third of patients benefit and the effect often fades. This has driven the search for treatments that are both safer and more durable.
Recruiting the immune system as an ally
Over the past decade, cancer therapy has been transformed by immune checkpoint inhibitors—drugs that release the “brakes” on immune cells so they can better attack tumors. One such brake, PD-1, sits on the surface of immune cells and can shut them down when engaged. The authors asked whether blocking PD-1 (using an anti-PD1 antibody) could help the immune system control vestibular schwannomas, and whether combining this with VEGF-blocking therapy would work even better. Using mouse models that closely mimic human inner-ear and peripheral nerve tumors, they treated animals with anti-VEGF alone, anti-PD1 alone, or the combination, then followed tumor growth, survival, and hearing.
Making tumor blood vessels work better, not disappear
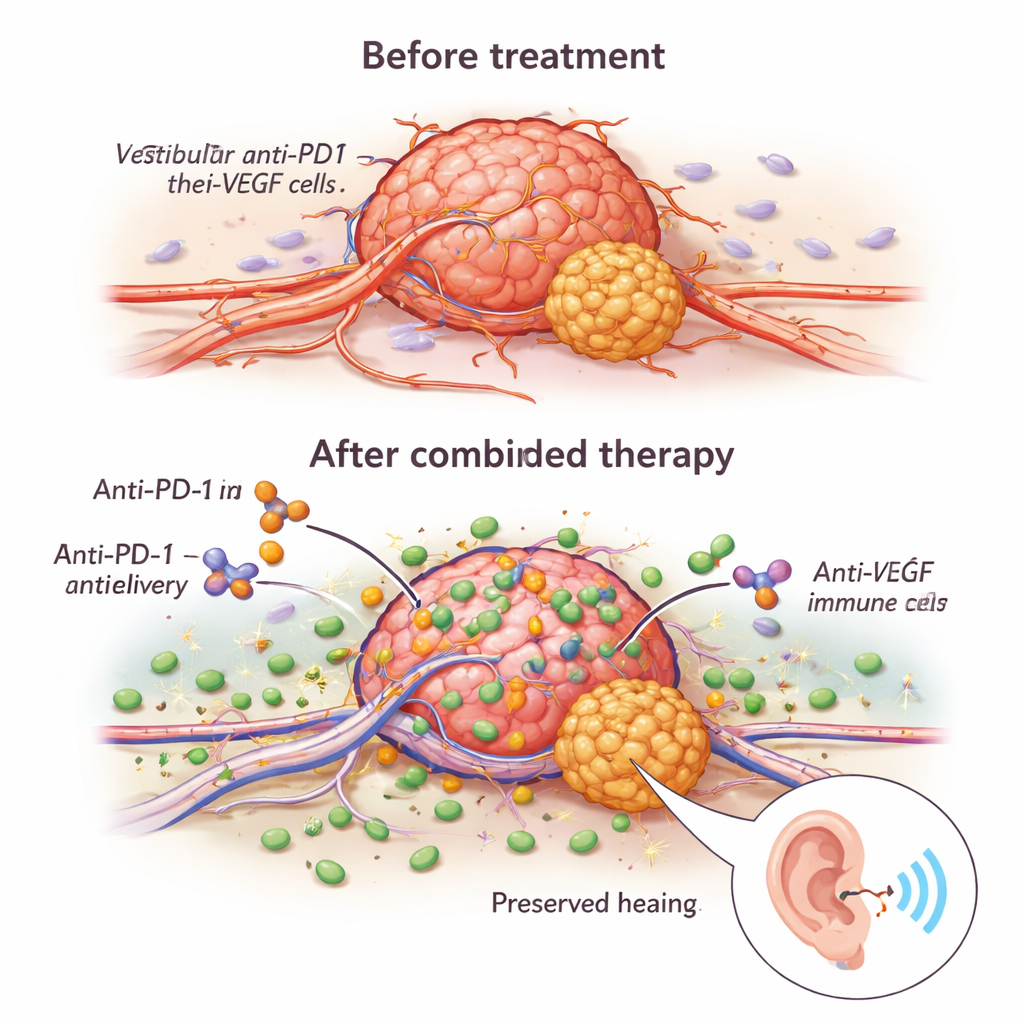
The team found that anti-VEGF did more than just starve tumors. It remodeled their abnormal, leaky blood vessels into more stable, better-functioning channels. Under the microscope, treated tumors had more vessels covered by supporting cells and a higher fraction of vessels that were actually carrying blood. This “normalization” improved the delivery of the anti-PD1 antibody into the tumor and allowed more cancer-fighting immune cells—especially CD8 T cells and natural killer (NK) cells—to enter. In mice given the combination therapy, tumors grew more slowly, had fewer dividing cells, and more cells undergoing programmed death, and the animals lived longer than with either drug alone.
Turning up the killing power of immune cells
The benefits of the combination were not just about getting more immune cells into the tumor; those cells were also more aggressive once they arrived. Anti-VEGF treatment increased the presence of molecules like granzyme B and perforin, which are the immune system’s molecular “bullets” for punching holes in tumor cells. A key switch in this process was a receptor called NKG2D, found on both T cells and NK cells. Anti-VEGF boosted NKG2D and its partner molecules on tumor cells, making immune cells more effective killers in lab tests. When researchers blocked NKG2D, the anti-tumor effects of the therapies dropped sharply, showing that this pathway is central to the drug synergy. Importantly, small samples from human tumors treated long-term with bevacizumab showed gene activity patterns consistent with more activated, less “exhausted” T cells and NK cells, suggesting that similar immune boosting may occur in patients.
Protecting hearing while controlling tumors
A crucial question was whether these immune-based treatments would harm or help hearing. When healthy mice received anti-PD1 alone, their hearing thresholds remained normal, suggesting the drug does not cause short-term damage to the auditory system. In tumor-bearing mice, anti-VEGF alone improved hearing compared with no treatment, echoing clinical experience. Anti-PD1 alone and the combination of anti-PD1 with anti-VEGF both restored hearing thresholds to near-normal levels. When the researchers mimicked a real-world scenario—first treating tumors with anti-VEGF and then either continuing, stopping, or adding anti-PD1—they found that switching to anti-PD1 could still slow tumor growth, but stopping anti-VEGF removed its hearing benefit. Keeping anti-VEGF and adding anti-PD1 gave the best results for both survival and hearing preservation.
What this could mean for patients
For people living with NF2-related schwannomatosis, this work suggests a future in which treatment does more than simply hold tumors at bay—it may also preserve the ability to hear. In carefully designed mouse models, combining a VEGF blocker with a PD-1 blocker created a friendlier landscape for immune cells, amplified their tumor-killing power through NKG2D, and protected hearing better than either approach alone. While clinical trials are still needed to confirm safety and benefit in humans, the study lays out a clear roadmap for testing this drug pairing as a new, potentially more durable strategy for managing vestibular schwannomas and preventing hearing loss.
Citation: Lu, S., Yin, Z., Wu, L. et al. NKG2D upregulation sensitizes tumors to combined anti-PD1 and anti-VEGF therapy and prevents hearing loss. Nat Commun 17, 1148 (2026). https://doi.org/10.1038/s41467-026-68865-8
Keywords: vestibular schwannoma, NF2, immunotherapy, bevacizumab, hearing preservation