Clear Sky Science · en
Antiparallel stacking of Csu pili drives Acinetobacter baumannii 3D biofilm assembly
How hospital germs build shielded cities
Some of the toughest hospital germs survive antibiotics by hiding in slimy, three-dimensional communities called biofilms. This study asks a simple but crucial question: how do individual bacteria knit themselves into such sturdy, layered structures? By zooming in with advanced electron microscopes, the researchers uncovered a microscopic “Velcro” made of hair-like filaments that let the dangerous pathogen Acinetobacter baumannii weave itself into protective 3D clumps. Understanding this hidden scaffolding could open new ways to pull biofilms apart and make existing drugs work better.
Tiny hairs that hold bacteria together
A. baumannii, a major cause of hard-to-treat hospital infections, bristles with thin surface hairs called Csu pili. Earlier work showed these pili are essential for forming rugged, dome-like biofilms, but no one knew how they actually knit cells into a 3D structure. Using several forms of electron microscopy, the authors first confirmed that in liquid culture the pili mostly stick out separately from each bacterium, like spikes on a burr. In dense surface-grown colonies, however, the picture changes dramatically: many pili pack tightly together, forming broad, flat bands that run between neighboring cells, creating a delicate-looking but extensive meshwork in three dimensions. 
Flat ladders built from paired filaments
To work out how these bands form, the team purified Csu pili and watched what they did on their own. Over days to weeks, single filaments began to pair up and then merge into multi-filament “superstacks”, eventually forming a gel-like network that closely mimicked the structures seen in real biofilms. High-resolution cryo-electron microscopy revealed the underlying design. Each pilus is not a smooth tube, but a zigzagging rod. When two rods lie side by side in opposite directions, they touch at repeating corner points, forming stable lateral junctions. Many such antiparallel pairs can then line up into a very thin, sheet-like stack—essentially a microscopic ladder or ribbon that is only one filament thick but many filaments wide.
A built-in blueprint for 3D growth
The detailed structures show that this stacking ability is hard-wired into the geometry of the pili themselves. The zigzag pattern repeats over a very short distance, creating many potential contact points along each filament. As a result, once two pili meet in the right orientation, a chain of junctions can form like a zipper, quickly extending the sheet. Importantly, the way the rods face ensures that contacts usually form between pili from different cells, not from the same one, which naturally promotes cell–cell linking. The stacks remain flexible and stretchy, so the growing biofilm can absorb physical stress without breaking apart. The researchers saw that during cell division, new daughter cells often end up with their pili positioned face-to-face, which encourages stack formation exactly where it is needed to tie cells into a 3D cluster. 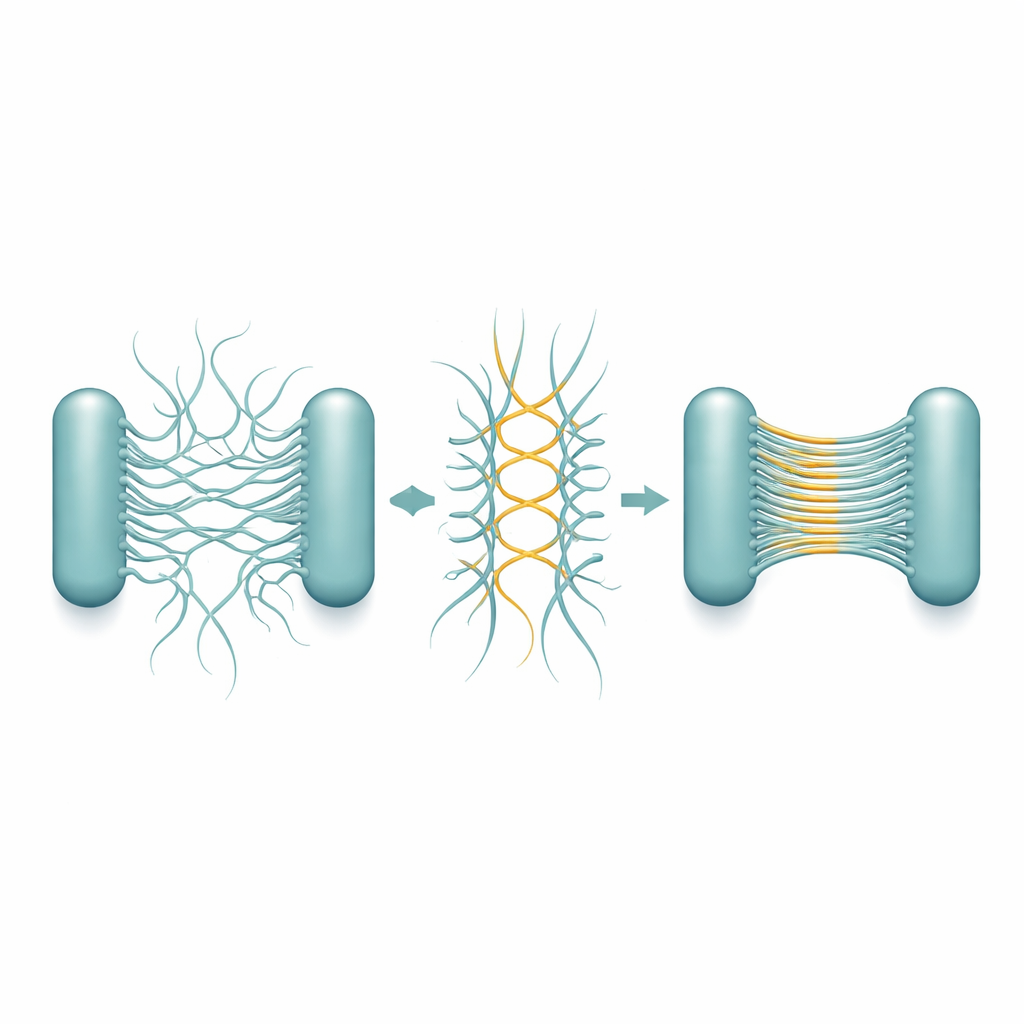
Glue, filler, and early scaffolding
The study also examined how other components of the biofilm contribute. A sugary polymer called PNAG and loose DNA from broken cells are known to accumulate in mature biofilms. Microscopy revealed these substances filling the narrow gaps between bacteria and wrapping around the pilus stacks, much like concrete poured around steel rebar. Yet when the team removed PNAG and DNA, bacteria still formed organized rafts held together by Csu pilus stacks alone. This shows that the pili build the primary scaffold, while the surrounding material later cements and stabilizes the structure.
New weak points in bacterial fortresses
In simple terms, this work explains how A. baumannii uses its surface hairs to lace itself into tough, multi-layered clumps that drugs and immune cells struggle to penetrate. The pili act first as hooks to grab onto surfaces and then as flexible straps that stack into flat sheets, tying neighboring cells together in all directions. Sugary polymers and DNA then fill in the gaps, turning this strap network into a solid fortress. By pinpointing the precise way pili stick to each other, the study highlights a new target: drugs or molecules that block pilus–pilus contacts could weaken biofilm scaffolds from within, making long-lasting bacterial infections easier to clear.
Citation: Malmi, H., Pakharukova, N., Paul, B. et al. Antiparallel stacking of Csu pili drives Acinetobacter baumannii 3D biofilm assembly. Nat Commun 17, 2508 (2026). https://doi.org/10.1038/s41467-026-68860-z
Keywords: biofilms, Acinetobacter baumannii, pili, antibiotic resistance, cryo-electron microscopy