Clear Sky Science · en
Early detection of aberrant cell fate and repair using circulating progenitor cells in patients with heterotopic ossification
When Bone Grows Where It Shouldn’t
Sometimes, after a bad injury or joint surgery, bone starts to grow inside muscles and other soft tissues where it does not belong. This condition, called heterotopic ossification, can lock joints, cause severe pain, and make everyday movements nearly impossible. Doctors currently find it only after the rogue bone has already taken hold, when it is too late to prevent it. This study asks a simple but powerful question: could a routine blood draw warn us weeks in advance that this hidden bone is on its way?
A Hidden Threat After Injury and Surgery
Heterotopic ossification is surprisingly common after severe burns, combat blast injuries, fractures, and especially hip replacement surgery. Yet today’s tools—X‑rays, CT scans, and bone scans—only pick it up after six to eight weeks, when new bone is already mature. Protective treatments such as radiation or strong anti-inflammatory drugs do exist, but they have serious side effects and are often given broadly because we cannot tell who truly needs them or when to start and stop. As a result, many patients either receive risky medications unnecessarily or miss the narrow window when those drugs could actually prevent extra bone from forming.
Following Rare Repair Cells in the Blood
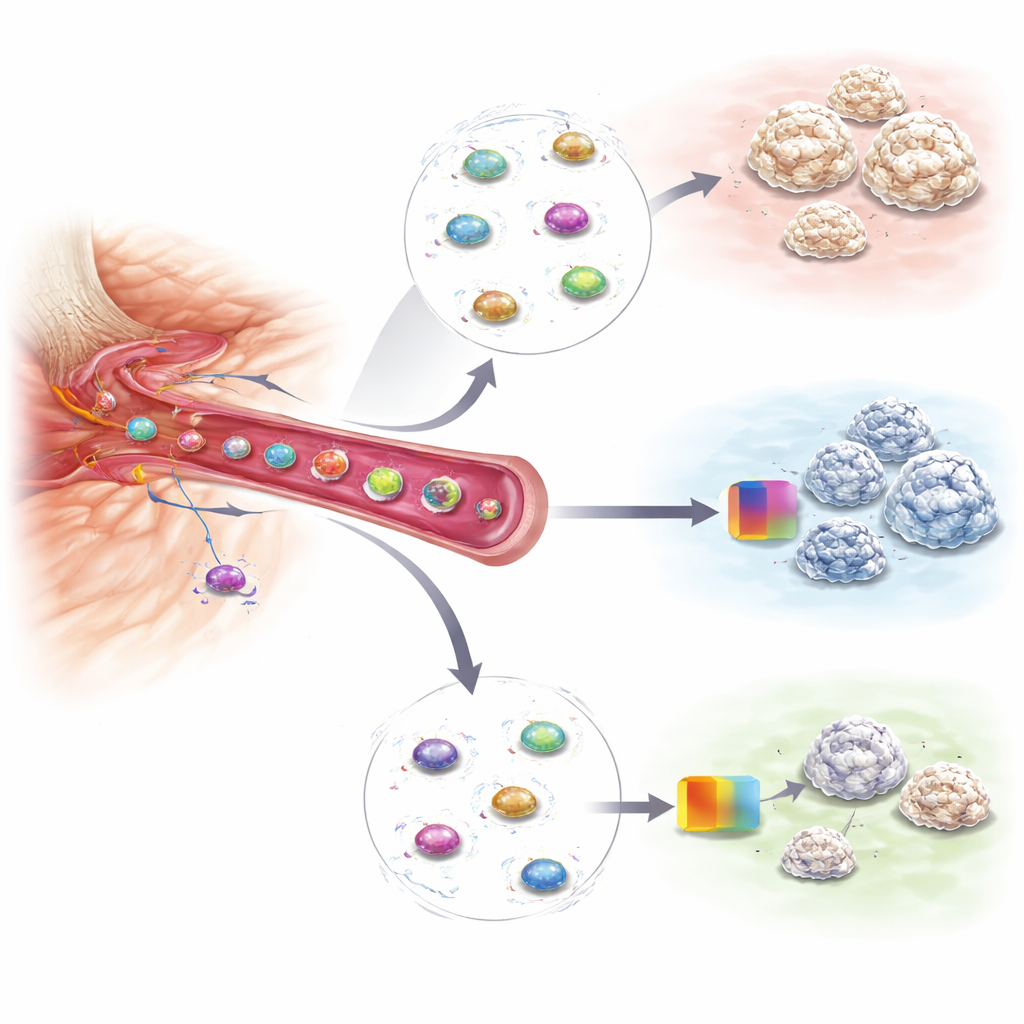
The research team focused on a special group of repair cells called mesenchymal progenitor cells, which normally help rebuild bone and connective tissue. After certain injuries, a tiny number of these cells slip into the bloodstream. Using a microfluidic device—a chip that can sort cells flowing through narrow channels—the scientists fished these rare circulating mesenchymal progenitor cells out of blood samples from mice and from patients undergoing hip replacement. They discovered that, just hours after an injury that will later cause heterotopic bone, these circulating cells carry a distinctive pattern of active genes that differs from both normal blood cells and from blood taken after injuries that do not lead to extra bone.
Turning Gene Patterns Into an Early Warning Test
By comparing blood from patients who did and did not go on to develop heterotopic ossification, and by matching those findings to detailed studies in mice, the team identified a set of 32 genes that together act as a molecular “footprint” of harmful bone formation. They then used machine-learning algorithms to train a prediction model that looks only at the activity levels of these genes in the circulating progenitor cells. In the human samples, this blood-based test correctly identified up to 90 percent of future cases while avoiding false alarms in all patients who stayed healthy. Strikingly, the gene signature appeared as soon as six hours after surgery or trauma—more than a month before standard imaging can detect new bone.
Seeing Whether Treatments Are Actually Working
The same blood-based approach also proved useful for monitoring therapy. In a mouse model, the researchers treated injured animals with a drug that blocks an enzyme involved in stiffening the tissue matrix and is known to reduce heterotopic bone. When treatment was effective, the gene signature in the circulating progenitor cells dropped sharply, and the animals later developed much smaller patches of extra bone. Other types of injuries that do not cause heterotopic ossification, such as muscle damage or head trauma, did not show the same surge in these cells or their gene pattern, underscoring the test’s specificity. This suggests that repeated blood draws could both flag high‑risk patients and reveal, in near real time, whether a preventive treatment is doing its job.
What This Could Mean for Patients
Taken together, the findings point to a future in which a simple blood test could guide highly personalized care after major injuries or joint surgery. Instead of treating everyone the same, clinicians could identify people whose repair cells are veering toward making bone in the wrong place, start preventive therapies early, and stop them once the dangerous gene signature fades. Beyond heterotopic ossification, similar “liquid biopsies” of circulating repair cells may help doctors track other conditions in which tissue healing goes awry, such as arthritis or fibrosis. For patients, that could translate into fewer complications, shorter courses of risky medications, and a better chance of staying mobile and pain-free.
Citation: Nunez, J., Holtz, M., Korlakunta, S. et al. Early detection of aberrant cell fate and repair using circulating progenitor cells in patients with heterotopic ossification. Nat Commun 17, 2231 (2026). https://doi.org/10.1038/s41467-026-68857-8
Keywords: heterotopic ossification, liquid biopsy, mesenchymal progenitor cells, joint replacement, early diagnosis